Navigating the long and winding road to the lymph node and optimal vaccine responses
Published in Bioengineering & Biotechnology
The rapid development of effective vaccines has provided an invaluable tool for addressing the global COVID-19 pandemic. However, as viral variants rapidly develop new tricks to evade vaccine-directed immune responses, vaccine-developers face an ever-mounting challenge in the rush to design newly effective shots. In the arms race between the virus and the immune response, prevention of infection is the highest goal, providing substantial individual benefit, while also breaking the cycle of transmission amongst the community.
Unfortunately, the most effective tools in the immune system’s arsenal for this mission, neutralizing antibodies, are easily fooled by the shifting appearance of the virus as it evolves to disguise itself from the immune response. T cells, on the other hand, are more reliably able to broadly identify viral variants through recognition of key features that are less likely to be changed during viral evolution. Though their ability to prevent infection at the earliest stages is limited, as sentinels in the body, T cells possess remarkable potential for long-lived memory. Rapid expansion of T cells at the first sign of viral exposure can interrupt an infection and prevent the most dangerous outcomes of disease. As we look to the next stage of vaccine development, shots designed to give the body an edge in this battle should ideally equip the immune response with both highly effective neutralizing antibodies and broadly-specific anti-viral T cells. Such a response could provide improved protection against the emergence of new viral variants and offer longer-lived individual and societal benefits.
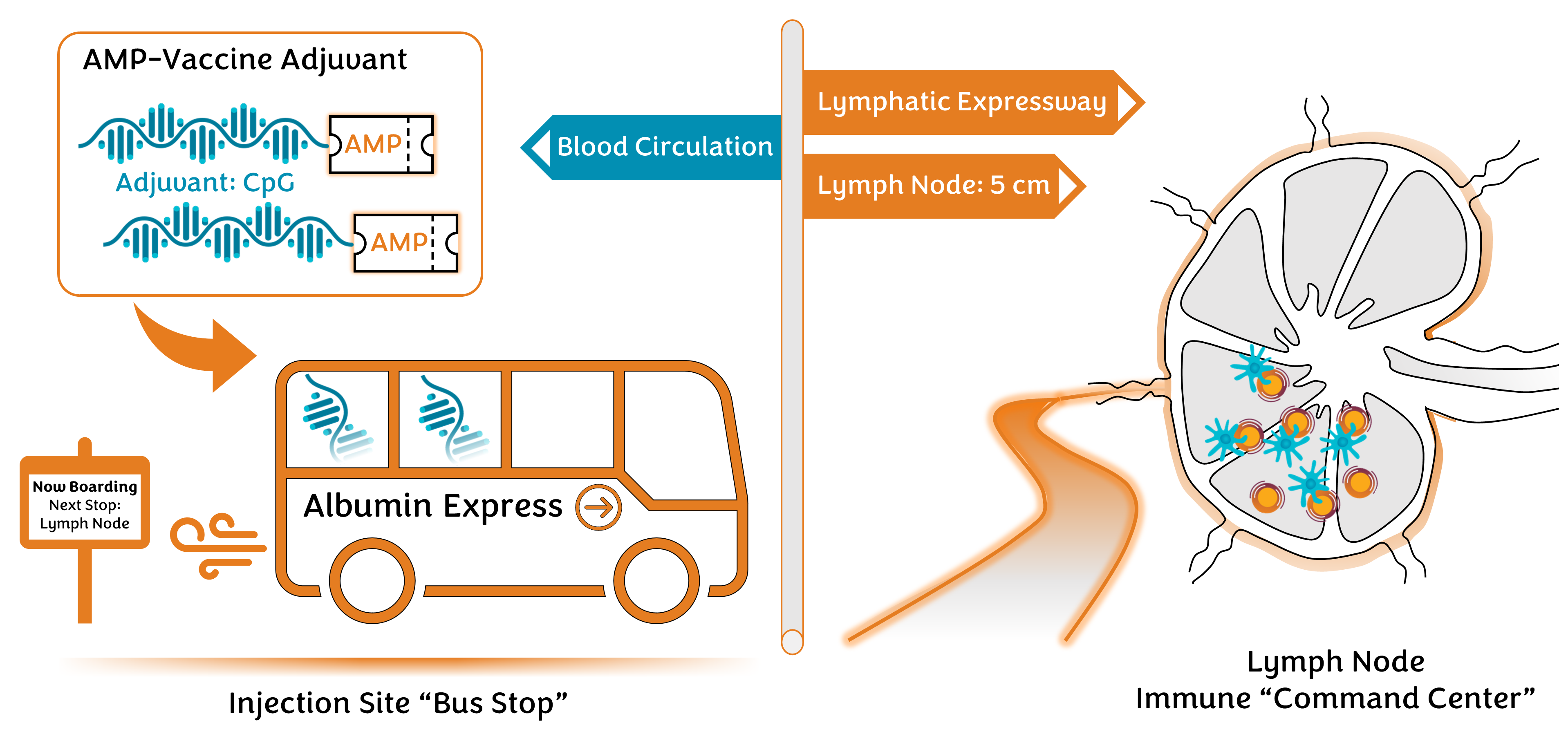
Effective orchestration of the immune response relies heavily upon coordination of immune biology in the lymph nodes. These sites serve as the immune system’s “command center” responsible for collecting cues from the body to assess threats to our health. This information is synthesized in the lymph nodes through highly organized interactions between millions of individual immune cells and used to coordinate the development of protective immune responses finely tuned to combat the identified danger. Vaccines are designed to program this response through administration of target antigen and immune-activating danger signals in the form of vaccine adjuvants. Access to the lymph nodes following injection, however, is far from certain. Transit from the injected tissue to lymph nodes is only efficient for relatively large molecules, leaving many smaller vaccine components hopelessly wandering the body and unable to effectively reach the immune cells which orchestrate immunity in the lymph nodes. Often, the result is a failure to realize the full potential of the desired immune response, or worse yet, development of immune inhibition or dangerous toxicities at other sites in the body.
Fortunately, the body is full of protein chaperones programmed to transport molecules from the tissues directly into lymph nodes. One such protein, albumin, is highly concentrated in the tissues, forming a ready fleet of shuttle buses offering express service directly into the lymph node – the immune system’s “command center.” Vaccines unable to reach lymph nodes on their own may board these shuttles by presenting the equivalent of a “molecular bus pass”, which in this case is a fatty acid structure designed to fit perfectly into one of several binding pockets normally utilized by albumin as “seats” to transport fatty-acid passengers throughout the body. The discovery of this process and the development of the Amphiphile (AMP) platform for vaccine and immunotherapy delivery was the seminal work of Dr. Haipeng Liu, and Professor Darrell Irvine at MIT in 2014 (Liu H., et al. Nature 2014). It has since been developed as a strategy to enhance immune responses to infectious disease and cancer at Elicio Therapeutics. Simple AMP-modification of antigen or adjuvant payloads provides these vaccine components with a “ticket” to ride the albumin shuttle bus through chemical installation of an albumin-binding fatty acid. The resulting AMP-vaccines are transported into lymph nodes by albumin after injection and effectively delivered to the specialized immune cells tasked with coordinating the build-up of protective antibody and T cell immunity. Optimal engagement in the lymph nodes yields immune responses with higher magnitude and quality, including balanced antibody and T cell generation, and attractive potential for durable memory.
As variants continued to drive pandemic waves of infection over the last year, we saw attractive opportunities for the AMP-strategy to program immune responses with unique qualities important for addressing current challenges. The viral Spike protein has proven an effective target antigen for vaccination. However, in contrast to immunization with conventional adjuvants, we theorized that combination of Spike protein with a potent lymph node-targeting adjuvant, AMP-CpG, could better program immunity to enhance the strength and breadth of neutralizing antibody responses, while promoting cross-protective and persistent anti-viral T cells. Building from the prior platform experience, we were able to confirm that prime-boost vaccination with AMP-CpG in mice gave striking expansion of highly functional Spike-specific T cell responses in several key sites responsible for local and systemic immune protection. Conventional CpG vaccines, on the other hand, were nearly indistinguishable from inactive controls. As predicted, these responses were not only highly durable, but also showed rapid expansion upon re-exposure to viral antigen. Similar responses were observed following immunization in non-human primates, where AMP-CpG vaccination rapidly elicited strong antibody responses with the ability to neutralize multiple viral variants at levels ten-fold higher than those found in humans recovering from COVID-19 infection. The induced T cells were equally agile, demonstrating equivalent ability to recognize original virus and recent variants. Together, this combination of antibodies and T cells, each exhibiting a promising ability to search out and intercept variants could contribute to a powerful and more adaptable immune arsenal in the fight against COVID-19.
Application to COVID-19 vaccine development is only one example of the broad potential for AMP to program optimal immunity in the lymph nodes. Many vaccine components and therapeutics are potentially highly productive workers eager to reach the immune system’s command center but so far have been stranded at the bus stop without a ticket into the lymph nodes. With the AMP-ticket in hand, they can rapidly reach their destination and dramatically transform the lymph nodes to coordinate immune protection against infectious pathogens, or cancers. Beyond COVID-19, this approach is already being tested to treat patients with mKRAS-driven cancers (NCT04853017) with potential to apply the AMP toolkit to address numerous unmet medical needs spanning infectious disease vaccines and cancer immunotherapy.
Follow the Topic
-
npj Vaccines
A multidisciplinary journal that is dedicated to publishing the finest and high-quality research and development on human and veterinary vaccines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Therapeutic HPV vaccines
Publishing Model: Open Access
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in