NPRC deletion mitigated atherosclerosis by inhibiting oxidative stress, inflammation and apoptosis in ApoE knockout mice
Published in Healthcare & Nursing and General & Internal Medicine
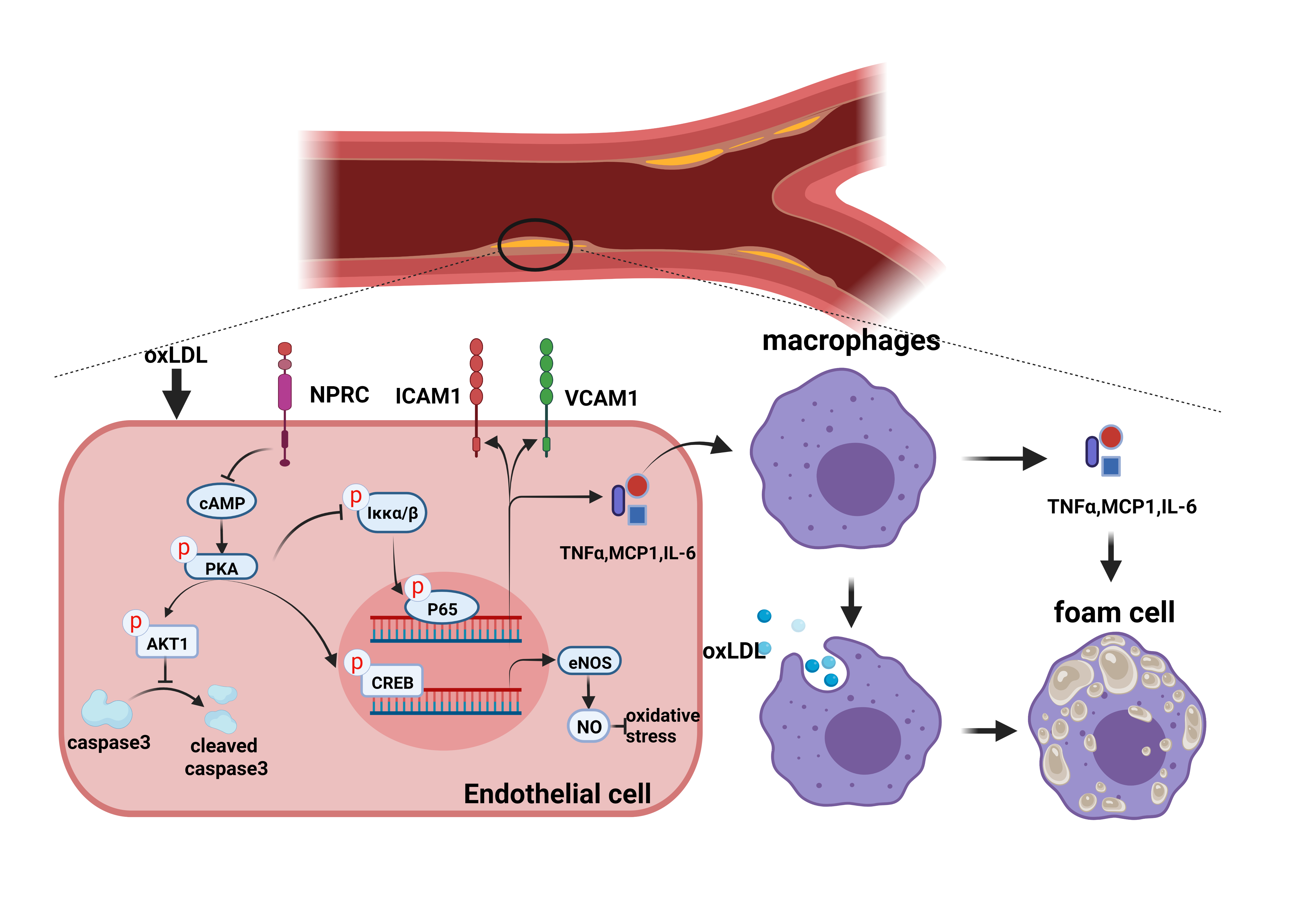
Fig1. Schematic diagram showing the mechanism of NPRC- promoted atherosclerosis.
Background
To discover susceptible genes and explore a genetic marker of coronary artery disease (CAD) in the Chinese Han population, we recently performed a genome-wide association study in 3363 CAD patients and 3148 controls, and found 6 novel loci in natriuretic peptide receptor C (NPRC), which showed a significant association with CAD even after adjustment for traditional risk factors of CAD, suggesting that NPR-C gene SNPs significantly contribute to CAD susceptibility in the Chinese Han population1. However, the molecular mechanism underlying the association between NPRC gene polymorphism and CAD is unclear.
Natriuretic peptides are a family of multiple functional proteins secreted by several organs, which bind with their own receptors to play physiological roles2. In the past, NPRC was deemed as a clearance receptor to degrade natriuretic peptides including ANP, BNP and CNP3. However, recent studies have revealed that NPRC also acts as an important signaling receptor expressed abundantly in endothelial cells to regulate a series of cellular biological processes3-6. Although our genome-wide association study discovered a significant contribution of NPR-C gene to CAD susceptibility in the Chinese Han population1, it remains elusive whether NPRC exerts a beneficial or harmful effect on AS lesions and what mechanism is involved in these effects.
How did we investigate this?
This study was designed to test the hypothesis that NPRC may promote AS lesion formation and instability by enhancing oxidative stress, inflammation and apoptosis via protein kinase A (PKA) signaling. ApoE-/-mice were fed chow or Western diet for 12 weeks and NPRC expression was significantly increased in the aortic tissues of Western diet-fed mice. Systemic NPRC knockout mice were crossed with ApoE-/- mice to generate ApoE-/-NPRC-/- mice, and NPRC deletion resulted in a significant decrease in the size and instability of aortic atherosclerotic lesions in ApoE-/-NPRC-/- versus ApoE-/- mice. In addition, endothelial cell-specific NPRC knockout attenuated atherosclerotic lesions in mice. In contrast, endothelial cell overexpression of NPRC aggravated the size and instability of atherosclerotic aortic lesions in mice. Experiments in vitro showed that NPRC knockdown in human aortic endothelial cells (HAECs) inhibited ROS production, pro-inflammatory cytokine expression and endothelial cell apoptosis, and increased eNOS expression. Furthermore, NPRC knockdown in HAECs suppressed macrophage migration, cytokine expression and phagocytosis via its effects on endothelial cells. On the contrary, NPRC overexpression in endothelial cells resulted in opposite effects. Mechanistically, the anti-inflammation and anti-atherosclerosis effects of NPRC deletion involved activation of cAMP/PKA pathway, leading to downstream upregulated AKT1 pathway and downregulated NF-κB pathway.
Our findings
There were several important findings in the present study. First, NPRC expression was increased in atherosclerotic lesions in ApoE-/- mice; Second, novel mouse models of ApoE-/- NPRC-/- and NPRCecKO were created and NPRC deletion reduced the size and increased stability of atherosclerotic lesions; Third, endothelial overexpression of NPRC in ApoE-/- and ApoE-/- NPRC-/- mice increased the size and instability of atherosclerotic lesions; Fourth, loss of NPRC lessened oxidative stress, pro-inflammatory cytokine expression and endothelial cell apoptosis, and enhanced eNOS expression, which were reversed by NPRC overexpression; Fifth, NPRC knockdown inhibited macrophage migration, pro-inflammatory cytokine expression and phagocytosis via its effects on endothelial cells and overexpression of NPRC resulted in an opposite effect; Finally, the mechanism underlying the beneficial effects of NPRC deletion involved activation of cAMP/PKA pathway, leading to downstream upregulated AKT1 pathway and downregulated NF-κB pathway. To the best of our knowledge, our study is the first in the literature to report the anti-atherosclerotic effects and underlying mechanisms of NPRC deletion in ApoE-/- mice.
Conclusions
NPRC expression was increased in atherosclerotic lesions and NPRC deletion reduced the size and increased stability of these lesions. Loss of NPRC attenuated oxidative stress, inflammation and endothelial cell apoptosis and increased eNOS expression via upregulated cAMP/PKA-AKT1 pathway and downregulated NF-κB pathway (Figure 1). Thus, targeting NPRC may provide a promising approach to the prevention and treatment of atherosclerosis.
For more details of our experimental methodology and results, please read our newly published paper in Signal Transduction and Targeted Therapy (DOI:10.1038/s41392-023-01560-y).
1 Hu, Q. et al. NPR-C gene polymorphism is associated with increased susceptibility to coronary artery disease in Chinese Han population: a multicenter study. Oncotarget 7, 33662-33674 (2016).
2 McGrath, M. F., de Bold, M. L. K. & de Bold, A. J. The endocrine function of the heart. Trends Endocrinol Metab 16, 469-477 (2005).
3 Shao, S. et al. Renal Natriuretic Peptide Receptor-C Deficiency Attenuates NaCl Cotransporter Activity in Angiotensin II-Induced Hypertension. Hypertension 77, 868-881 (2021).
4 Murthy, K. S. & Makhlouf, G. M. Identification of the G protein-activating domain of the natriuretic peptide clearance receptor (NPR-C). J Biol Chem 274, 17587-17592 (1999).
5 Pagano, M. & Anand-Srivastava, M. B. Cytoplasmic domain of natriuretic peptide receptor C constitutes Gi activator sequences that inhibit adenylyl cyclase activity. J Biol Chem 276, 22064-22070 (2001).
6 Rubattu, S. et al. Epigenetic control of natriuretic peptides: implications for health and disease. Cell Mol Life Sci 77, 5121-5130 (2020).
Follow the Topic
-
Signal Transduction and Targeted Therapy
This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in