Omicron, the perfect shape and solid interior for more stable surviving and massive immune evasion?
Published in Healthcare & Nursing
Severe acute respiratory syndrome coronavirus 2 (SARS-COV-2), the pathogen of the coronavirus disease 2019 (COVID-19) pandemic, emerged in late 2019 and rapidly spread around the world. By the end of February 2023, there have been more than 750 million confirmed cases of COVID-19, including over 6.8 million deaths worldwide (https://covid19.who.int/). Similar to other coronaviruses, SARS-COV-2 is an enveloped virus, that replicates in the host cytoplasm and assembles at intracellular membranes. The main structural components of SARS-COV-2 are the spike protein S, the transmembrane proteins M and E, and the nucleocapsid protein N. The N protein forms a viral ribonucleoprotein complex (RNP) with the viral RNA (Jack et al., 2021).
In November 2021, a new SARS-COV-2 variant named Omicron (B.1.1.529) was reported in South Africa, and within several days on November 26th, WHO designated it as a variant of concern (VOC) (Kannan et al., 2021). Omicron features a high mutation rate, particularly in the spike protein. These mutations lead to a higher affinity of RBD to ACE2 and massive immune evasion of previously developed monoclonal antibodies and vaccines (Cao et al., 2022). A comprehensive analysis of the whole virion structure in situ is necessary to fully understand how the Omicron virions evade the immune system and how the spike protein functions.
In this study, we report the authentic architecture of both Omicron BA.1 and BA.2 lineages using cryo-electron tomography (cryo-ET) and sub-tomogram averaging. We isolated authentic Omicron subvariants from nasopharyngeal swabs of COVID-19 patients and propagated them in Vero E6 cells. For biosafety issues, BA.1 and BA.2 virions were deactivated with paraformaldehyde. Direct visualization of the intact virions shows typical features of a coronavirus: a spherical particle containing a dense viroplasm, bounded by a lipid bilayer with spike protrusions (Figure 1). Both Omicron lineages BA.1 and BA.2 display uniform spherical shapes, with enveloped long/short axis ratios close to 1.
Compared to the WT, the lumen of both BA.1 and BA.2 lineages are densely packed with bucket-like densities, most likely representing ribonucleoproteins (RNPs). Interestingly, the mean number of RNPs increased from 30 in WT to 43 and 48 in BA.1 and BA.2, respectively. To rigorously examine the abundance of nucleocapsids, an equal amount of focus forming units (FFUs) or RNA genome copies among WT, BA.1, and BA.2 were collected and subjected to Western Blot analysis. The immunoblotting results indicated that the level of nucleocapsid protein was approximately 2-fold higher in BA.1 and BA.2 than in WT. SARS-CoV-2 RNA genome-pull down assays were further performed to determine the level of RNA genome-binding nucleocapsid during the life cycle of the viruses. We found nearly 2-fold nucleocapsid protein in BA.1/BA.2 RNA-pull down enrichments than WT. The 2-fold increase in RNA-protein binding affinity may be jointly contributed by the mutations in viral RNA and nucleocapsid protein.
Our cryo-ET study of the BA.1 and BA.2 subvariants provides the overall landscape and fine structural insight into Omicron. Sub-tomogram averaging analysis resolved the needle-like post-fusion spikes and the pre-fusion spikes in both open and close states. We report a significant increase in the number of RNPs in both BA.1 and BA.2 lineages. However, we did not observe any packing units of the RNPs. We propose that these RNP particles are highly heterogeneous and densely but randomly distributed inside viral lumen. It has been reported that, when mixed with RNA, SARS-CoV-2 N protein can form gel-like condensates, structurally similar to RNPs. This phase separation behavior can provide a membrane-less compartment to concentrate and protect viral RNA. Previous studies have suggested a role of N protein in the immune evasion of SARS-CoV-2. After virus infection, the N protein can prevent the recognition and cleavage of viral dsRNA and suppress siRNA-induced RNA degradation. Moreover, the N protein can significantly elevate the replication of virus RNA and down-regulate genes that establish the host antiviral state. Our observation of up-regulated N protein in omicron variants may hint at their massive immune evasion and suggest N protein as a promising diagnostic and therapeutic target against the SARS-CoV-2 epidemic.
For more detail on the experiments and results, please read our paper
https://www.nature.com/articles/s41392-023-01385-9
Reference
- Jack, A., Ferro, L.S., Trnka, M.J., Wehri, E., Nadgir, A., Nguyenla, X., Fox, D., Costa, K., Stanley, S., Schaletzky, J., and Yildiz, A. (2021). SARS-CoV-2 nucleocapsid protein forms condensates with viral genomic RNA. PLoS Biol 19, e3001425. 10.1371/journal.pbio.3001425.
- Kannan, S., Shaik Syed Ali, P., and Sheeza, A. (2021). Omicron (B.1.1.529) - variant of concern - molecular profile and epidemiology: a mini review. Eur Rev Med Pharmacol Sci 25, 8019-8022. 10.26355/eurrev_202112_27653.
- Cao, Y., Wang, J., Jian, F., Xiao, T., Song, W., Yisimayi, A., Huang, W., Li, Q., Wang, P., An, R., et al. (2022). Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Nature 602, 657-663. 10.1038/s41586-021-04385-3.
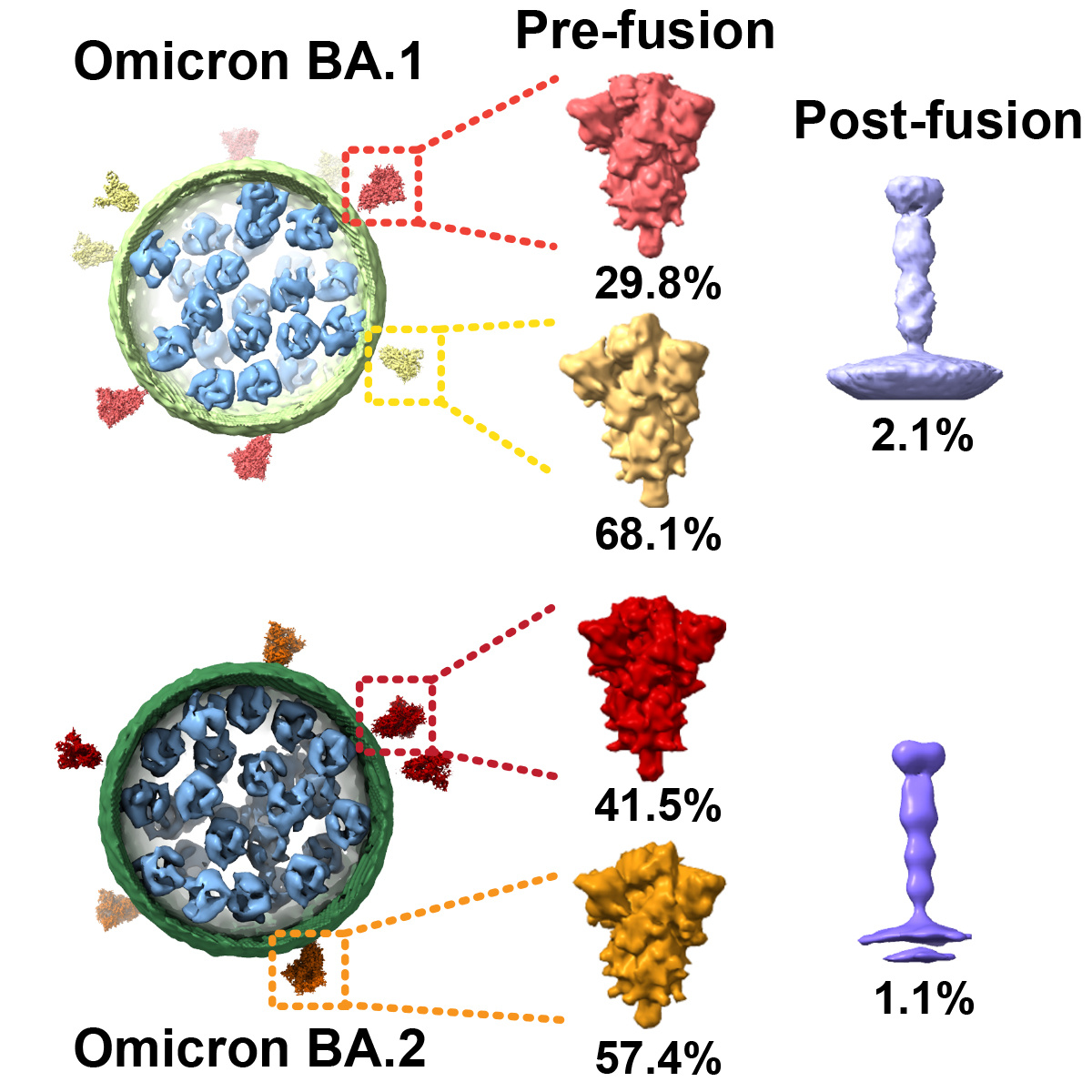
Figure 1. Architecture of Omicron variants BA.1 and BA.2, revealed by cryo-ET.
Follow the Topic
-
Signal Transduction and Targeted Therapy
This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in