Precise regulation of the hyperacidic lysosomes in M2-like macrophages to achieve macrophage-based tumour immunotherapy
Published in Biomedical Research

Tumour-associated macrophages (TAMs), which predominantly display a tumour-promoting M2-like phenotype, play a crucial role in tumour progression, metastasis, immune evasion, and resistance to immunotherapy1. Currently, some anti-TAMs drugs are under preclinical and clinical evaluation, consisting of three main strategies: (i) inhibition of TAMs recruitment, (ii) TAMs depletion, and (iii) re-education of M2-like TAMs2. When the inhibition of TAMs recruitment and survival might not suffice to stimulate durable anti-tumour response, researchers are more likely to choose the re-education strategy which not only ameliorates the immunosuppressive functions but also potentiates antigen cross-presentation3. However, a significant challenge arises when attempting to repolarize M2-like phenotype in tumour tissues because it is hard to specifically regulate M2-like TAMs without affecting the lysosomal function of M0-, or M1-like macrophages in the healthy tissues. To overcome this hurdle, researchers have turned to investigate the subtle differences between macrophages of various phenotypes.
TAMs in M2-like phenotype present an upregulated lysosomal function, such as increased compartmental acidity and high level of lysosomal enzymes, which easily results in accelerated antigen degradation4. Conversely, pro-inflammatory M1-like macrophages with reduced lysosomal function have the potential for antigen cross-presentation that activates CD8+ T cells for effective tumour elimination, similar to the behaviour observed in mature dendritic cells5. Consequently, it becomes imperative to explore whether it’s feasible to utilize the heightened lysosomal acidity of M2-like macrophages to specifically manipulate TAMs to promote anti-tumour immune response while sparing macrophages in healthy tissues.
Based on our finding that the lysosomal pH difference between M2-like TAMs (pHL ~ 4.4) and M0-/M1-like macrophages (pHL ~ 5.2), we are introducing a pH-gated nanoparticle (PGN) to unambiguously distinguish the acidic lysosomal milieu of different immune cells, allowing the specific targeting and reprogramming of M2-like macrophages in tumour microenvironment. Initially, the pH-gated nanoadjuvants tend to passively accumulate into tumour sites through the enhanced permeability and retention (EPR) effect, and be internalized by tumour-associated macrophages (Figure 1b). Following this, PGNs with a pH transition (pHt) of 4.9 could only respond to highly acidic lysosomal compartment of M2-like TAMs rather than counterpart of other macrophages, and disassemble into unimers. The Gly-Phe-Leu-Gly (GFLG) linkage is then exposed to cathepsin B and degraded, releasing free IMDQ and stimulating TLR7/8 in lysosomes (Figure 1c). Finally, this strategy provides a powerful toolbox for precise targeting and stimulating of distinct signaling pathway within specific endocytic organelles to advance cancer immunotherapy.
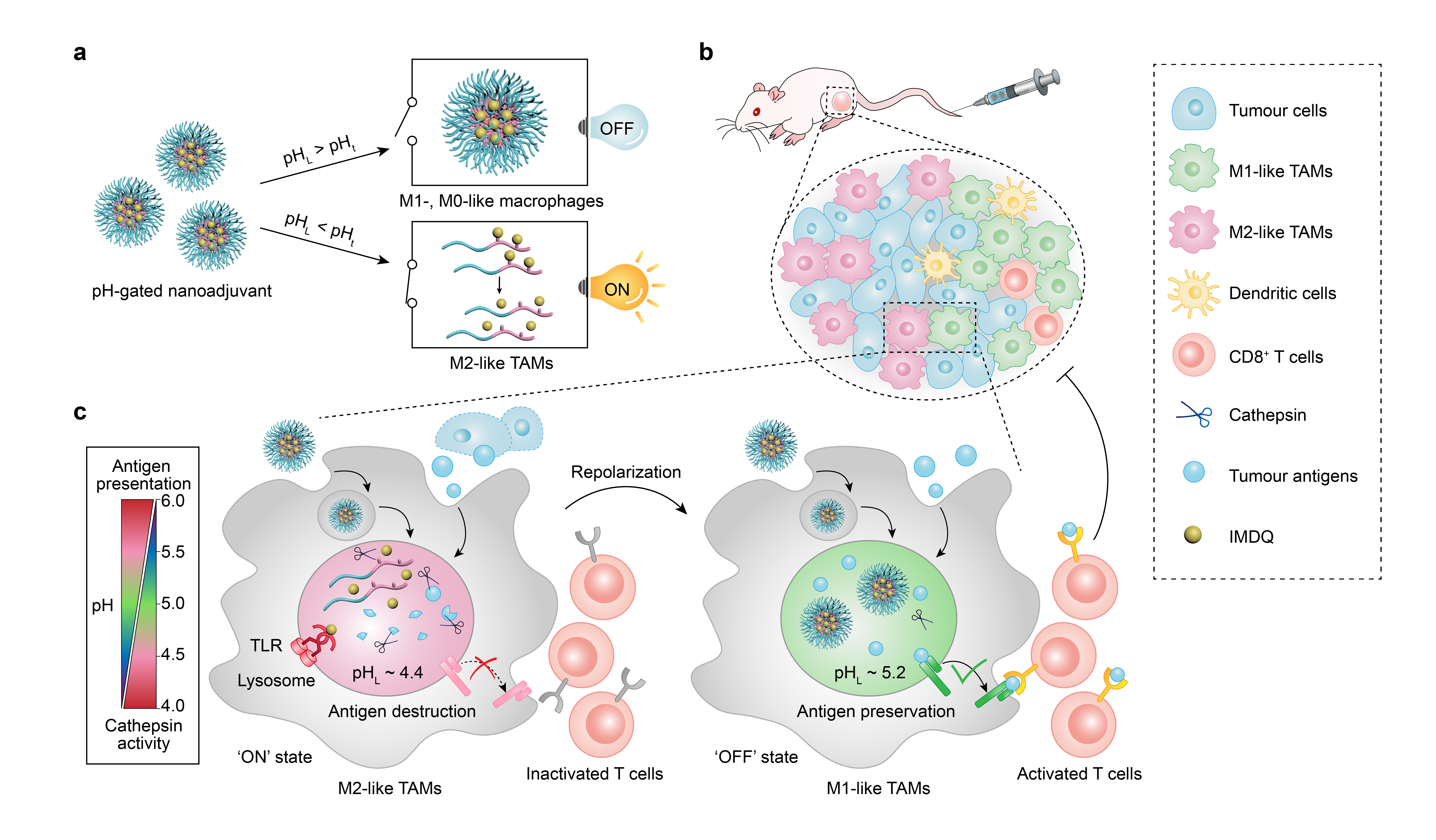
Figure 1. Design and mechanism of PGN4.9 nanoadjuvant for reprogramming M2-like TAMs into M1 phenotype via pH-gated regulation of lysosomal function.
In this study, we first determined the lysosomal pH of various living cell types in tumour microenvironment using the ratiometric pH quantification protocol. Our quantitative analysis confirms that the lysosomes of M2-like BMDMs (pHL ~ 4.4) were hyper-acidified as compared with other cell types (pHL > 5.2). This finding excited our curiosity to investigate whether our developed ultra-pH-sensitive nanotechnology6,7 could differentiate the subtle lysosomal pH deviation. Therefore, we construct a PGN library which consists of 11 nanoparticles with pHt values from 4.5 to 5.5 and 0.1 pH increment, covering lysosomal pH range of different cell types. Flow cytometry and confocal experiments revealed that PGN4.9 can unambiguously differentiate the lysosomal pH of M2-like macrophages (selectivity index ~ 5) from those of M0- and M1-like macrophages, tumour cells and normal cells in vitro. Moreover, we demonstrate that PGN4.9 selectively profiles intratumoural M2-like macrophages in vivo, while imaging signal versus the percentage of M2-like macrophages exhibited an excellent linear correlation (R = 0.7968, P < 0.001).
However, the story of M2-like macrophage-targeted strategy is far from over. We further designed the PGN4.9 nanoadjuvant which renders AND-gated behaviour that converts two orthogonal inputs (i.e., pH and cathepsin activity) into IMDQ release as a single output. The pHt tunability facilitates the successful screening of PGN4.9 nanoadjuvant for the specific targeting of lysosomal pH of M2-like TAMs, followed by enzymatic cleavage-mediated drug release to achieve logic-gated immunotherapies. PGN4.9 nanoadjuvant efficiently tunes down lysosome acidification and proteolysis of M2-like TAMs, thereby dramatically potentiates antigen cross-presentation, and activates CD8+ T cell function to elicit adaptive tumour immunity in various tumour models. In contrast, PGN4.9 nanoadjuvant keeps inert in other cell types and circumvents acute systemic toxicity for cancer immunotherapy.
In summary, the nanotechnology represents a powerful platform in macrophage-based tumour immunotherapy. By selectively fine-tuning lysosomal proteolysis of M2-like TAMs, we have unlocked a novel approach that promotes antigen cross-presentation and elicits adaptive tumour immunity. Our findings may inspire a new insight into how to specifically regulate lysosomal function of TAMs for efficient cancer immunotherapy.
References
- Noy, R. & Pollard, J.W. Tumor-associated macrophages: from mechanisms to therapy. Immunity 41, 49-61 (2014).
- Pittet, M.J., Michielin, O. & Migliorini, D. Clinical relevance of tumour-associated macrophages. Nat Rev Clin Oncol 19, 402-421 (2022).
- Rodell, C.B., et al. TLR7/8-agonist-loaded nanoparticles promote the polarization of tumour-associated macrophages to enhance cancer immunotherapy. Nat Biomed Eng 2, 578-588 (2018).
- Cui, C., et al. A lysosome-targeted DNA nanodevice selectively targets macrophages to attenuate tumours. Nat Nanotechnol 16, 1394-1402 (2021).
- Muntjewerff, E.M., Meesters, L.D. & van den Bogaart, G. Antigen Cross-Presentation by Macrophages. Front Immunol 11, 1276 (2020).
- Wang, Y., et al. A nanoparticle-based strategy for the imaging of a broad range of tumours by nonlinear amplification of microenvironment signals. Nat Mater 13, 204-212 (2014).
- Chen, B., et al. A pyroptosis nanotuner for cancer therapy. Nat Nanotechnol 17, 788-798 (2022).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in