RTS,S, the first malaria vaccine - what mediates protection and how long does immunity last?
Published in Microbiology, General & Internal Medicine, and Immunology

Currently, RTS,S is the only malaria vaccine that has completed phase III clinical trials in African children, and in October 2021 the WHO recommended widespread use of RTS,S among children in sub-Saharan Africa and other regions with moderate to high P. falciparum malaria transmission.
Modest vaccine efficacy and short durability for RTS,S
RTS,S demonstrated only modest efficacy with relatively short longevity. In young children aged 5–17 months, RTS,S vaccine efficacy against clinical malaria was ~50% over 18 months of follow-up. Efficacy waned quickly such that there was little or no significant efficacy after 18 months. When a booster dose was given at 18 months, vaccine efficacy over 3-4 years was 36%. Notably, other vaccine candidates tested in clinical trials have either failed to confer significant protection in target populations of malaria-exposed individuals or have not yet demonstrated higher efficacy or greater durability than RTS,S in young children that is reproducible in multiple trials. So while RTS,S is imperfect, there is no vaccine on the horizon that is markedly superior.
What protective immune responses are induced by RTS,S?
The RTS,S vaccine is based on the circumsporozoite protein (CSP), a major surface-expressed antigen on the sporozoite stage of P. falciparum, and antibodies are the main mediator of vaccine efficacy. However, the mechanisms of action of these antibodies are not fully understood, especially among children. This lack of knowledge impedes progress to improve on RTS,S and constrains the development of more efficacious second-generation vaccines. Recent studies have demonstrated that antibody interactions with Fc-receptors on immune cells can play important roles in mediating immunity against sporozoites, including opsonic phagocytosis by neutrophils and monocytes and cellular cytotoxicity by NK cells.
We investigated the induction and longevity of functional antibodies in a phase IIb trial of RTS,S among Mozambican children aged 1-5 years old. We found that RTS,S vaccination induced specific IgG with FcγRIIa and FcγRIII binding activity and promoted phagocytosis by neutrophils, THP-1 monocytes, and primary human monocytes, neutrophil respiratory burst activity, and NK cell activation, suggesting these mechanisms could be contributing to vaccine efficacy.
Biostatistical modelling suggested IgG1 and IgG3 contribute in promoting FcγR binding and phagocytosis, and IgG targeting the NANP-repeat and C-terminal regions of CSP were similarly important for functional activities
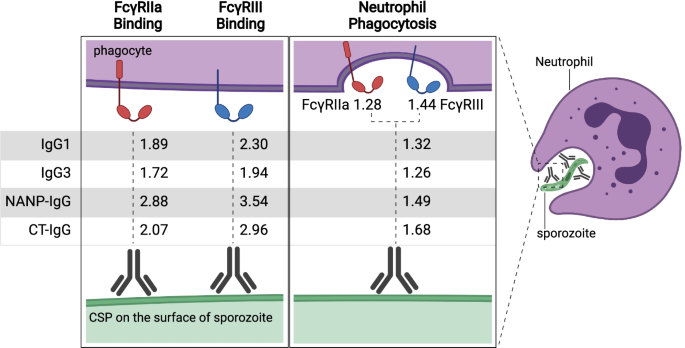
Figure Legend: Antibodies against CSP opsonise sporozoites and interact with FcγRs to promote phagocytosis, which is predominantly mediated by neutrophils. Rate ratios are shown for the relationship between IgG subclasses and IgG to NANP-repeat and C-terminal domains of CSP, and antibody functional activities (FcγRIIa or III-binding and neutrophil phagocytosis). Rate ratios are also shown for the relationship between FcγRIIa and FcγRIII binding and neutrophil phagocytosis. Values are rate ratios and represent the percent change in participants’ antibody functional activities for each unit increase in IgG reactivity or FcγR binding. p≤0.001 for all rate ratios except neutrophil phagocytosis with IgG3 (p=0.023) and IgG to NANP (p=0.023).
Induction of functional antibodies is highly variable and negatively influenced by malaria exposure
Responses were highly heterogenous among children, and the magnitude of neutrophil phagocytosis by antibodies was relatively modest, which may reflect modest vaccine efficacy. Interestingly, induction of functional antibodies was lower among children who had experienced higher malaria exposure. The basis for this negative influence is not known, but could reflect impacts of malaria on the innate immune system or CD4+ T-cells, which are important for vaccine responses. This contrasts to what is seen with COVID-19 vaccines where prior infection with SARS-CoV-2 is associated with better vaccine responses.
Protective immune responses are relatively short-lived
There is very limited data on the longevity of functional antibodies after vaccination with RTS,S. We found that functional antibodies largely declined within a year post-vaccination, and decay was highest in the first 6 months, consistent with the decline in vaccine efficacy over that time. Decay rates varied for different antibody parameters. Interestingly, decay was slower for neutrophil phagocytosis. This may be because the sensitivity of neutrophil phagocytosis to FcγR engagement by IgG means that there is a lower IgG threshold concentration required for activity, which may partly be explained by the expression of three activating FcγRs (FcγRIIIa, IIIb, and IIa) on the neutrophil surface.
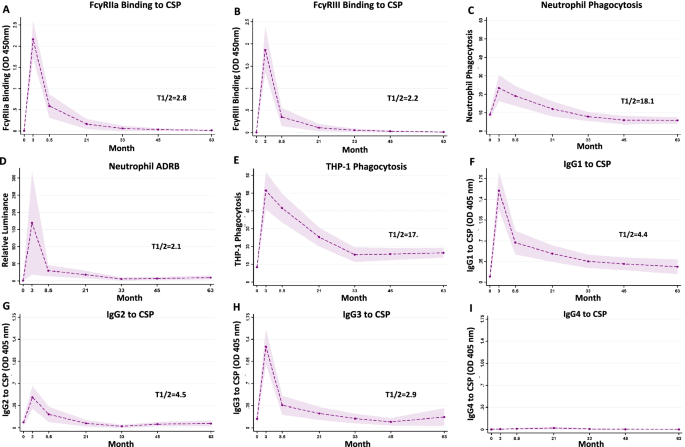
Figure Legend: Serum samples were collected at baseline (month 0, M0), 30 days after the third vaccination (month 3, M3) and later time points (M8.5, M21, M33, M45 and M63). Samples were tested for A FcγRIIa-binding and B FcγRIII-binding to CSP, C opsonic phagocytis by neutrophils and D antibody-dependent respiratory burst (ADRB) in neutrophils and E opsonic phagocytosis by THP-1 cells. Antibodies were also tested for IgG subclasses to CSP, including F IgG1, G IgG2, H IgG3 and I IgG4. Data from immunologic assays were analysed in generalised linear mixed model (GLMM). The dashed lines represent the predicted means, the shaded area represents the 95% CIs and T1/2 (in months) indicates the estimated half-lives from the generalised linear mixed model. Half-life represents the time for magnitude to reduce by 50% from the M3 time-point.
Conclusions
Achieving high vaccine efficacy and greater longevity of efficacy are key global goals outlined by WHO and funding partners. We established key functional antibody activities induced by RTS,S, and the temporal kinetics of these responses, in young African children, who are the primary target group of the vaccine. Our results provide insights to understand the modest and time-limited efficacy of RTS,S in children. In future studies, analysis of these functional antibodies needs to be extended into the RTS,S phase III trial, including evaluation of responses in different populations, and an analysis of correlations of protection. Findings from this study address knowledge gaps around RTS,S immunity and may contribute to improving RTS,S efficacy and longevity in the future or the development of next-generation vaccines.
Link to the paper:
https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-022-02466-2
Feng G, Kurtovic L, Agius PA, Aitken EH, Sacarlal J, Wines BD, Hogarth PM, Rogerson SJ, Fowkes FJI, Dobaño C, Beeson JG. Induction, decay, and determinants of functional antibodies following vaccination with the RTS,S malaria vaccine in young children. BMC Med. 2022 Aug 25;20(1):289
Link to a related paper - Induction and decay of functional complement-fixing antibodies by the RTS,S malaria vaccine in children, and a negative impact of malaria exposure
https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-019-1277-x
Follow the Topic
-
BMC Medicine
This journal publishes outstanding and influential research in all areas of clinical practice, translational medicine, medical and health advances, public health, global health, policy, and general topics of interest to the biomedical and sociomedical professional communities.
Ask the Editor - Immunology, Pathogenesis, Inflammation and Innate Immunity
Got a question for the editor about the complement system in health and disease? Ask it here!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Advancing research and management of menopause
BMC Medicine is calling for submissions to our new Collection, Advancing research and management of menopause, focusing on new perspectives and care practices that support individuals during this significant life transition. This Collection seeks to advance the understanding and management of menopause by gathering clinical research on its physiological, psychological, and social dimensions. We welcome submissions that explore topics such as perimenopause, menopausal symptoms, early menopause, and menopausal hormone therapy (MHT) but also innovative interventions that help individuals navigate menopause beyond pharmacological treatment.
The menopause journey, encompassing the climacterium and the natural conclusion of reproductive years, is a pivotal phase in the lifecycle that impacts health and well-being. This Collection aims to consolidate research that addresses the multifaceted aspects of menopause, including its physiological, psychological, and social dimensions, thereby offering a comprehensive view of the experiences faced during this transition.
Advancements in the understanding and management of menopause have opened new avenues for improving health. Research indicates that the heterogeneity of menopause (surgical, spontaneous, induced, early) as well as the heterogeneity of MHT (formulations, duration, age of initiation relative to menopause onset, cyclicity, progestrogen/progestin) can differentially affect outcomes, yet are not as well understood or studied. Studies on menopausal symptoms have enhanced our understanding of their prevalence and impact on daily life. Research on menopausal hormone therapy has evolved, shedding light on its benefits and risks, while growing evidence highlights a number of other effective options for managing common menopausal symptoms. Additionally, the focus on long-term effects of menopause, e.g. osteoporosis and prevention strategies, underscores the necessity for ongoing research and tailored interventions during and after this transition.
Continued exploration in this field promises to yield significant insights, potentially leading to the development of novel therapeutic options, personalized management strategies, and a broader understanding of the heterogeneity of menopause and individuals’ experiences. As research progresses, we may see improved guidelines for addressing early menopause and premature ovarian insufficiency, ultimately empowering individuals to navigate this life stage with confidence and health.
Topics of interest include but are not limited to:
- Management strategies for menopausal symptoms
- MHT: risks and benefits
- Post-menopausal osteoporosis prevention
- Ovarian aging and its implications
- Understanding early menopause and premature ovarian insufficiency
This collection supports and amplifies research related to SDG 3: Good Health & Well-Being and SDG 5: Gender Equality.
Publishing Model: Open Access
Deadline: Jul 17, 2026
Cardiac immunity and inflammation
BMC Medicine invites submissions to a new Collection focused on the interplay between cardiac immunity and inflammation. We welcome original research, translational studies, and comprehensive reviews that elucidate the underlying mechanisms, clinical consequences, and therapeutic opportunities arising from immune-cardiac interactions. By highlighting the importance of this link, we aim to foster collaboration and advance our understanding of how to improve patient outcomes in heart disease.
The intersection of immunity and cardiovascular biology represents a transformative frontier in modern medicine. Recent studies have highlighted the role of immune responses in various heart conditions, including myocarditis, cardiac xenotransplantation, and heart failure. Inflammation is increasingly recognized as a contributing factor to the pathogenesis and residual risk factor of cardiovascular diseases. This Collection aims to explore the mechanisms by which immune responses influence cardiac function, the role of clonal hematopoiesis and heart disease, and the implications of such interactions for cardiac outcomes.
Understanding cardiac immunity and inflammation is crucial for developing more targeted therapies and accurate CVD risk prediction. Past advances provided insights into the inflammatory processes involving cardiovascular conditions, leading to innovative approaches for diagnosis and treatment. For instance, the identification of biomarkers such as C-reactive protein enhances our ability to assess inflammation in patients with cardiovascular disease. Furthermore, the intersection of cardio-oncology has opened new avenues for research into how cancer therapies influence cardiac immune responses, emphasizing the need for a multidisciplinary approach to patient management.
Continued research in this field holds promise for unveiling drug discovery targets and strategies to mitigate inflammation-related cardiac damage. As we deepen our understanding of the immune mechanisms at play, we may see the development of more clinically relevant models of human disease and precision medicine approaches tailored to individual patients. The integration of this knowledge with advancements in technology, such as biomarker discovery and imaging techniques, could revolutionize the way we approach cardiac care.
Topics of interest include (but are not limited to):
- Role of immunity in cardiovascular biomedicine
- Systemic and cardiac-specific inflammatory pathways
- Residual inflammatory risk, e.g. “SMuRF-less but inflamed”
- Cross-organ communication and immune crosstalk
- Structural and functional cardiac remodeling driven by inflammation
- Novel biomarkers and prediction models
- Clonal hematopoiesis
- Cardio-oncology & therapy-induced immune modulation
- The role of immunity in cardiac xenotransplantation including partial heart transplantation
- Exposome/environmental exposures
- Epigenetic regulation of cardiac immunity and inflammation
- Immune memory and trained immunity in cardiovascular contexts
- Multi-omics integration (epigenomics, transcriptomics, proteomics) in cardiac disease
All manuscripts submitted to this journal, including those submitted to collections and special issues, are assessed in line with our editorial policies and the journal’s peer review process. Reviewers and editors are required to declare competing interests and can be excluded from the peer review process if a competing interest exists.
This Collection supports and amplifies research related to SDG 3 (Good Health and Well Being).
Publishing Model: Open Access
Deadline: Feb 01, 2027







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in