Sense of smell under stress: Circuit mechanisms of olfactory perceptual deficits induced by early life adversity
Published in Neuroscience
‘Stress is how we respond to events based upon our perception’ – Henry Marsh
The perception of adverse events guides our behavioral responses towards stressed conditions. While the human psychophysical readouts from clinics limit our understanding of the circuit mechanisms, we have to rely on pre-clinical animal models for probing the neural basis of pathophysiological conditions. A multipronged experimental approach is an absolute necessity. Our sense organs collect and process the chemical and physical stimuli to facilitate the percept formation. Stress during initial years of one’s life, often referred to as the Early Life Stress (ELS) is capable of negatively affecting our sensory systems in more ways than what we know in the field today. So, what happens if our perception gets affected? Does it render us incapable of enjoying our life to the best? Sensory systems are largely intertwined with our stress-responsive centers, making them plausibly vulnerable to stressful conditions. In fact, a study showed reduction in the volume of Olfactory Bulb (OB) in humans who suffered from childhood disturbances [1]. OB is the first relay station, where the sensations from smell molecules (called odours) are transferred to projection neurons (mitral/tufted, M/T cells), which can be monitored as odour-evoked glomerular maps, as a first step in forming olfactory percepts [2–4]. The inhibitory network of OB filters and refines this information before sending to higher centres [5, 6]. What goes wrong inside the OB neuronal network due to ELS is still enigmatic. The bi-directional connectivity in the olfactory-limbic axis and the top-down modulation of OB circuits make it highly prone to adverse effects of any stressors. In fact, the inter-neurons of OB can be modulated under different affective states, which can cause changes in their activity profiles leading to compromised odour information processing [6].
We began by looking at the effect of ELS on olfactory perception by weaning the mice at an age as early as post-natal day 14. We focused on quantifying different behavioural readouts of olfactory detection, discrimination learning and memory by employing an automated Go/No-Go odor discrimination paradigm using custom-built olfactometers. In this paradigm, a mouse is supposed to lick on a lick tube in response to an odour that is paired with water-reward while upon learning, it should avoid licking for an unrewarded odour. Over time, mice can accurately distinguish the odours. This discrimination learning was poor in ELS mice when compared to normal-weaned control mice. We confirmed that the perceptual learning deficit did not arise due to their inability to detect the odours as their olfactory detection quantified by buried food test, turned out to be normal. We verified that their usual non-olfactory based learning and memory on a novel object recognition and discrimination task was unaffected, suggestive of a specific olfactory perceptual deficit caused by ELS. As a first step towards unravelling the neural circuit mechanism, quantification of an Immediate early gene protein, c-Fos, which reliably marks the active neurons during a behavioural response, was carried out on completion of odour discrimination paradigm. A reduction in the number of active cells was found in the external plexiform layer in ELS mice, one of the layers of OB that harbours most of the somatostatin-releasing GABAergic inhibitory interneurons (SOM-INs). We focused our attention to this class of interneurons as somatostatin has been shown to have anxiolytic effects in higher brain areas [8, 9], and therefore we could anticipate adverse effects on their functioning due to ELS. As per our expectations, we observed reduced neuronal branching pattern of SOM-INs in ELS mice.
SOM-INs of OB have known anatomical connectivity through the olfactory-limbic axis [10–13]. As the number and branching pattern of SOM-INs were reduced in ELS mice, we next asked if this is reflected in the output signals from OB. By using slice electrophysiology, we indeed figured that in the ELS mice, the inhibitory feedback on projection neurons, Mitral/Tufted (M/T) cells, were compromised. As the output of OB projection neurons can be modulated by the entire inhibitory network [5], we further established that the reduced inhibitory feedback we observed in ELS mice is due to the malfunctioning of SOM-INs. To prove this, we decided to optogenetically activate the SOM-INs and see if they release the inhibitory neurotransmitter GABA. Activating them by shining blue-light on channel-rhodopsin expressing interneurons, through a custom-built automated multi-LED system, resulted in the release of somatostatin and GABA from OB slices, as quantified using the ELISA technique. Further, to check if this experimental strategy can cause any shift in the inhibitory feedback onto the M/T cells, we carried out a critical experiment. Upon recording the activity of M/T cells while optogenetically activating the SOM-IN using electrophysiology, a light-induced increase in the inhibition was observed in ELS mice. These findings established the synaptic connectivity between the projection neurons and SOM-INs and confirmed the malfunctioning of SOM-INs under ELS conditions.
Once we know the neural circuit affected, the other experiments one could plan are in vivo imaging from the behaving animals and optogenetic modulation of circuits to study their effect on animals’ behavioural responses. To study how the activity of SOM-INs change during different phases of learning, we recorded calcium dynamics from SOM-INs expressing GCaMP6f. We observed a learning-dependent reduction of calcium activity in normal-weaned animals whereas, no such changes were observed in ELS mice. Finally, to confirm if the reduced synaptic inhibition on the M/T cells, resulted from the malfunctioning of SOM-INs, was driving the behavioral impairments, we carried out in vivo optogenetic activation (using ChR2 expression in SOM-INs) and inhibition (using Arch expression in SM-INs) experiments while the mice were involved in odor discrimination task. Activating the SOM-INs caused a rescue of the learning deficits observed in ELS mice. Inhibiting the SOM-INs in control mice which was a tool to reduce the inhibition on the M/T cells, decreased their learning ability and mimicked the slower learning pace of ELS mice. Further, optogenetically inhibiting SOM-INs of ELS mice disabled their odor discrimination abilities resulting in a severe learning-deficient phenotype. Such a multipronged experimental approach allowed us to establish the role of SOM-INs involving circuitry of the OB in mediating perceptual learning deficits in ELS mouse model.
Our study establishes the experimental strategy for answering more questions in the future, by tapping into the modulatory peptidergic versus GABAergic role of SOM-INs in the olfactory functions given that OB harbours different types of SOM-receptors across its different layers [11]. Also, since we know, that olfactory problems can arise due to depression, stress and childhood maltreatment in humans, we plan to measure olfactory fitness of the ELS-affected population using lab-invented precise and robust methods [14–16]. This would help in identifying and characterizing sensory and cognitive deficits at early stages and in designing better treatment strategies.
References
- Croy I, Negoias S, Symmank A, Schellong J, Joraschky P, Hummel T. Reduced olfactory bulb volume in adults with a history of childhood maltreatment. Chem Senses. 2013;38:679–684.
- Abraham NM, Spors H, Carleton A, Margrie TW, Kuner T, Schaefer AT. Maintaining accuracy at the expense of speed: stimulus similarity defines odor discrimination time in mice. Neuron. 2004;44:865–876.
- Abraham NM, Vincis R, Lagier S, Rodriguez I, Carleton A. Long term functional plasticity of sensory inputs mediated by olfactory learning. Elife. 2014;3:e02109.
- Bhattacharjee AS, Konakamchi S, Turaev D, Vincis R, Nunes D, Dingankar AA, et al. Similarity and strength of glomerular odor representations define a neural metric of sniff-invariant discrimination time. Cell Rep. 2019;28:2966–2978.
- Abraham NM, Egger V, Shimshek DR, Renden R, Fukunaga I, Sprengel R, et al. Synaptic inhibition in the olfactory bulb accelerates odor discrimination in mice. Neuron. 2010;65:399–411.
- Gschwend O, Abraham NM, Lagier S, Begnaud F, Rodriguez I, Carleton A. Neuronal pattern separation in the olfactory bulb improves odor discrimination learning. Nat Neurosci. 2015;18:1474–1482.
- Brunert D, Rothermel M. Extrinsic neuromodulation in the rodent olfactory bulb. Cell Tissue Res. 2021;383:507–524.
- Engin E, Stellbrink J, Treit D, Dickson CT. Anxiolytic and antidepressant effects of intracerebroventricularly administered somatostatin: behavioral and neurophysiological evidence. Neuroscience. 2008;157:666–676.
- Lin LC, Sibille E. Somatostatin, neuronal vulnerability and behavioral emotionality. Mol Psychiatry. 2015;20:377–387.
- Tomoda T, Sumitomo A, Newton D, Sibille E. Molecular origin of somatostatin-positive neuron vulnerability. Mol Psychiatry. 2022;27:2304–2314.
- Nocera S, Simon A, Fiquet O, Chen Y, Gascuel J, Datiche F, et al. Somatostatin serves a modulatory role in the mouse olfactory bulb: neuroanatomical and behavioral evidence. Front Behav Neurosci. 2019;13:61.
- Lepousez G, Mouret A, Loudes C, Epelbaum J, Viollet C. Somatostatin contributes to in vivo gamma oscillation modulation and odor discrimination in the olfactory bulb. Journal of Neuroscience. 2010;30:870–875.
- Lepousez G, Csaba Z, Bernard V, Loudes C, Videau C, Lacombe J, et al. Somatostatin interneurons delineate the inner part of the external plexiform layer in the mouse main olfactory bulb. Journal of Comparative Neurology. 2010;518:1976–1994.
- Abraham NM. Olfactory-action meter for precise quantification of olfactory dysfunctions and neurocognitive deficits. 2022.
- Bhattacharjee AS, Joshi S V, Naik S, Sangle S, Abraham NM. Quantitative assessment of olfactory dysfunction accurately detects asymptomatic COVID-19 carriers. EClinicalMedicine. 2020;28:100575.
- Bhowmik R, Pardasani M, Mahajan S, Magar R, Joshi S V, Nair GA, et al. Persistent olfactory learning deficits during and post-COVID-19 infection. Current Research in Neurobiology. 2023;4:100081.
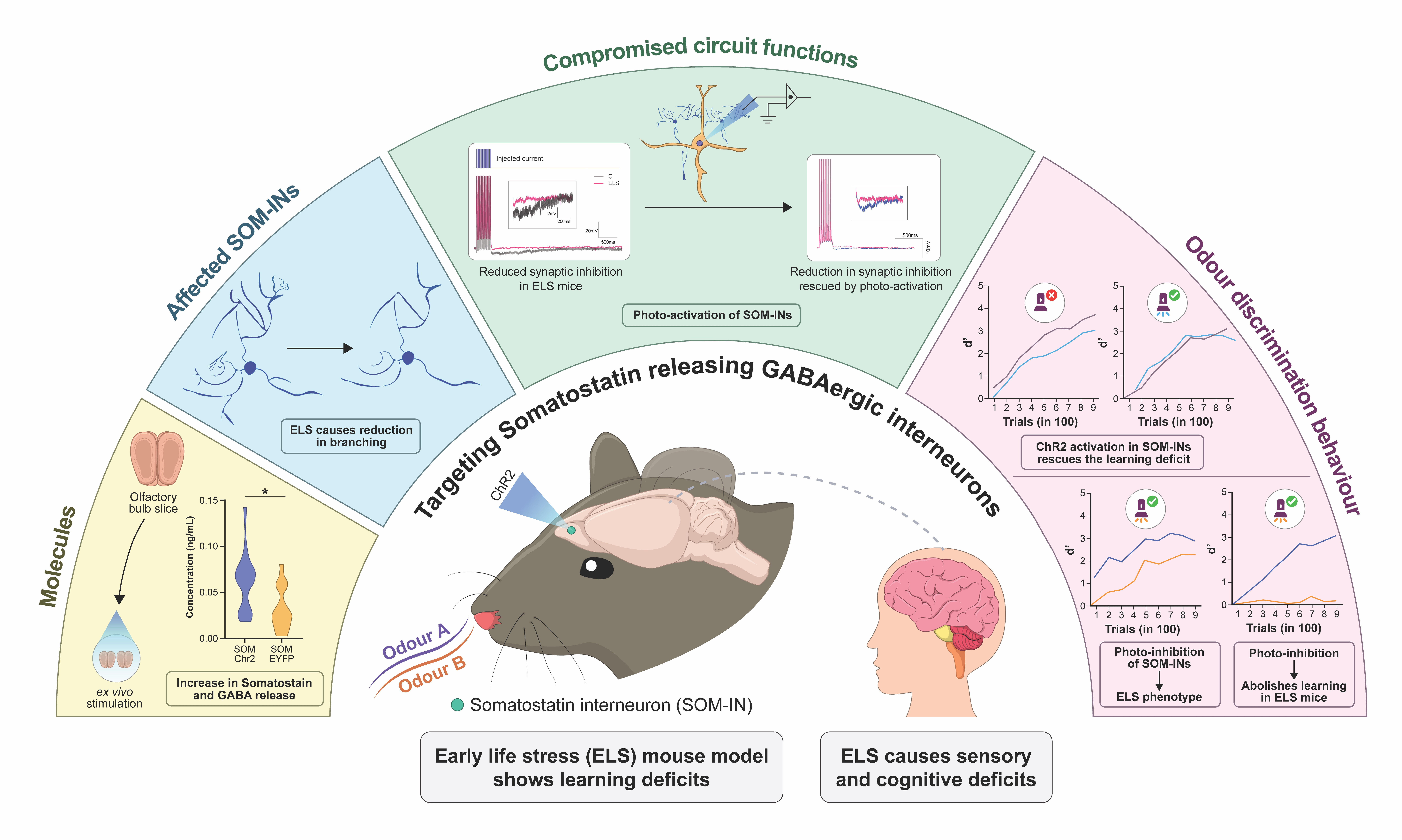 Figure: Usage of multipronged experimental approach in studying the neural circuit mechanism of early life stress- mediated olfactory perceptual learning dysfunctions in mice
Figure: Usage of multipronged experimental approach in studying the neural circuit mechanism of early life stress- mediated olfactory perceptual learning dysfunctions in mice
Follow the Topic
-
Molecular Psychiatry

This journal publishes work aimed at elucidating biological mechanisms underlying psychiatric disorders and their treatment, with emphasis on studies at the interface of pre-clinical and clinical research.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcement

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in