Signalling failure delays training of antibody in early life.
Published in Microbiology
This may come as a surprise, but there are other viruses that infect our lungs than COVID! One of them is called Respiratory Syncytial Virus (RSV). This innocuously named virus is THE leading cause of hospitalisation in children during winter months, but somehow doesn’t get the same level of attention as its sexier relatives influenza and SARS. One of my colleagues Prof Peter Openshaw has previously suggested it be called ‘deadly killer virus’ to get it the attention it deserves.
One of the interesting aspects about RSV is that it is possible to get re-infected with the same virus. This is unusual because the assumption is mostly that once the immune system has seen a virus once it is then better trained to deal with it in the future, preventing further infections. However, some viruses such as RSV (and also coronaviruses, like the one that causes COVID) can reinfect. We don’t fully understand why this is the case, but one contributing factor is a protein in the blood called antibody. Antibodies are highly specific molecules made by the immune system that can bind and kill viruses.
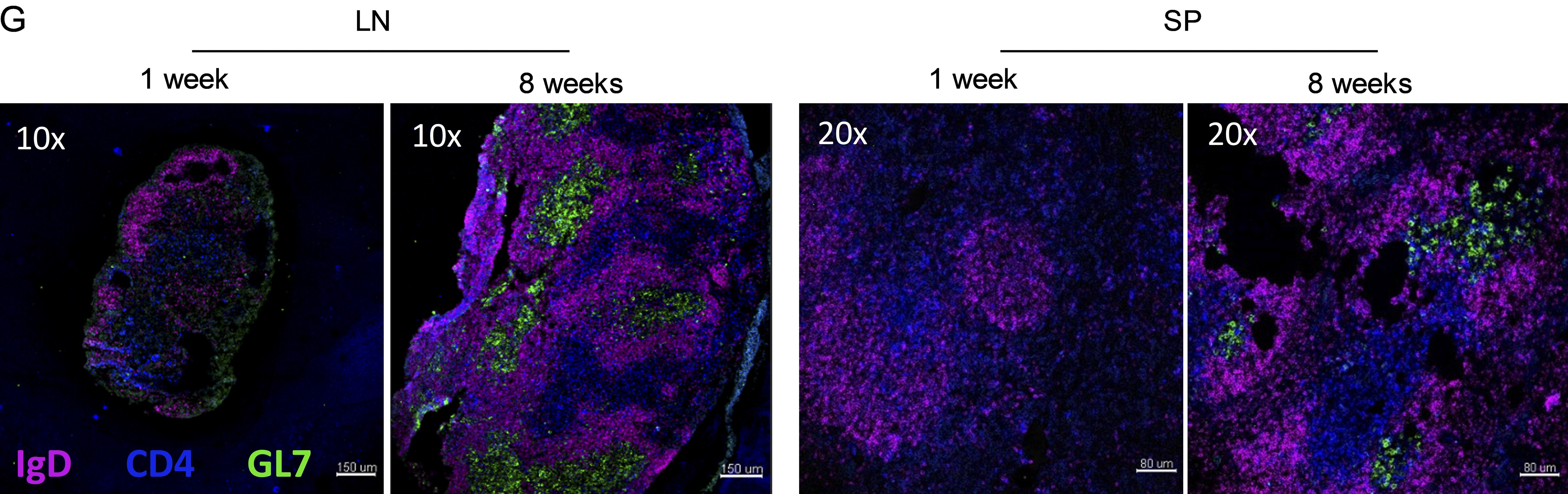
Antibody molecules are produced by a white blood cell called the B cell, but in order to produce the best possible antibody, B cells need help from another type of cell called the T cell. The conversation between T and B cells happens in the lymph nodes – which is why you get swollen glands after infection or immunisation. In our recent work, ‘Enhanced IL-2 in early life limits the development of TFH and protective antiviral immunity‘ recently published in the Journal of Experimental Medicine we explored this interaction. Specifically, we asked the question are there differences between the T-B cell crosstalk in early life – the time of greatest susceptibility to RSV.
We found that baby mice infected with RSV produce less antibody than adult mice infected with the same dose. This lack of antibody left the mice susceptible to re-infection with the virus. Side by side with the reduction of antibody, there were fewer of the helpful T cells needed to train the B cells. If we specifically removed those helpful T cells (called T follicular helper cells or Tfh) from adult mice before RSV infection, we saw a very similar effect – re-infection.
We dug deeper as to why these handy Tfh cells are not so active in early life. We identified a role for a molecule that cells use to talk to each other called interleukin two (the sequel to the commercially more successful, but less interesting interleukin one). There is more of this molecule sloshing around in early life and it may play a role in training the early immune system what is good and what is bad.
Overall, out findings may help us to develop better vaccines that work from the moment babies are born and stop them catching viruses such as RSV.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in