Solid-liquid-gas reaction accelerated by gas molecule tunnelling-like effect
Published in Materials
Solid-liquid-gas reactions are critical steps in various chemical processes, such as corrosion, photocatalysis, fuel cell, etc. Nevertheless, a comprehensive description of reactions at the solid-liquid-gas interface on the nanoscale is currently unavailable owing to the challenge of tracking the evolution of individual particles in a combined liquid and gas medium at nanoscale, especially with respect to why and how reaction efficiencies can be improved by better controlling the input gas flow. Visualization of solid-liquid-gas reactions at a single particle level could be expected to address this gap by identifying the specific details of interface evolution during the reaction.
The cutting-edge in-situ liquid cell transmission electron microscopy (LCTEM), which allows the direct imaging of dynamic reactions in liquids, has contributed a lot to visualize the critical pathways of solid-liquid reactions at the nanoscale. The radiolysis of water molecules under the electron beam can generate oxidative or reductive species including some gaseous products such as H2 and O2. Nanobubbles would be formed when the gaseous products (H2 and O2) exceed their saturated concentration. Since gold nanocrystals can be oxidative etched by oxygen in the presence of Br- ions, we came up with an idea to set up and directly image a solid-liquid-gas reaction on the surface of a gold nanorod with oxygen nanobubbles in aqueous hydrobromic through LCTEM (Fig. 1a). The electron energy loss spectroscopy of water with nanobubble shows a pre-peak ∼ 527 eV indicating the presence of molecular O2 (Fig. 1b). Since H2 nanobubbles act as a reservoir of reductant and don’t contribute to the etching of Au, Therefore, the enhanced etching of gold nanorods by the O2 nanobubbles (at least nanobubbles contain most O2) can be distinguished (Fig. 1c).
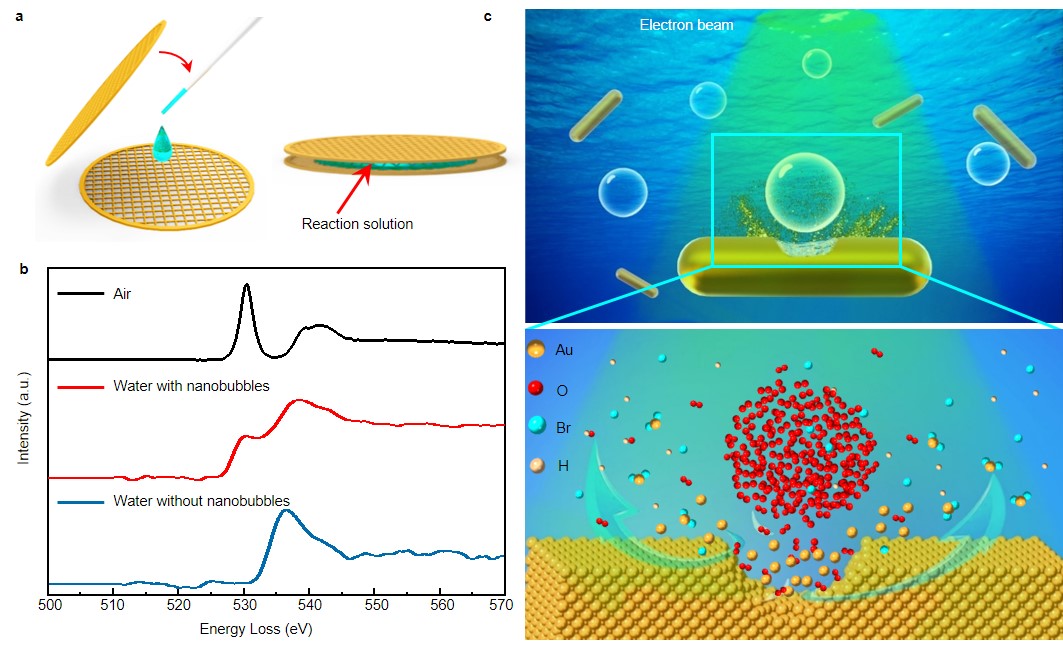
Fig. 1. Schematic illumination of a solid-liquid-gas reaction established in a liquid cell. a, Schematic liquid cell encapsulating a reaction solution. b, Electron energy loss spectroscopy (EELS) shows O K-edge of air (black, reference spectrum from Gatan EELS Atlas), water without nanobubble (blue) and the water with nanobubbles generated by electrolysis of water at a later stage (red). The O K-edge of water with nanobubbles shows evidence of molecular O2 at ∼527 eV as compared with the reference spectrum from O2 gas (black). c, Schematic illustration of the etching of gold nanorods in HBr solution with O2 nanobubbles in the near distance.
The strong oxidative species generated by the radiolysis of water such as OH• and H2O2 can oxidize gold atoms into ions. The nanorod typically adopts an ellipsoidal shape with a smooth surface and is gradually dissolved since these species are considered to be uniformly distributed in water in the irradiated area (Fig. 2a, b). However, when nanorods are surrounded by nanobubbles, the scenario becomes very different. When the nanobubbles with O2 are close to the gold nanorods, the nanorods are etched not only by the oxidizing species in the solution but also by the O2 from the nanobubbles. Subsequently, the nanorods exhibit local accelerated etching rather than uniform etching. The nanorod develops an irregular shape with indents near the nanobubbles (Fig. 2c, d). Interestingly, the nanorods and nanobubbles are not in direct contact while the indent is developed but are separated by an ultra-thin water layer (Fig. 2e, f). And the calculated ratio between O2 molecules and water molecules near the nanobubble is about three orders of magnitude higher than the saturation values of dissolved oxygen. Therefore, the environment near the nanobubble should be treated as a gas-liquid-solid system and the reaction mechanism in it is dramatically different from the solid-liquid etching (Fig. 2 a).
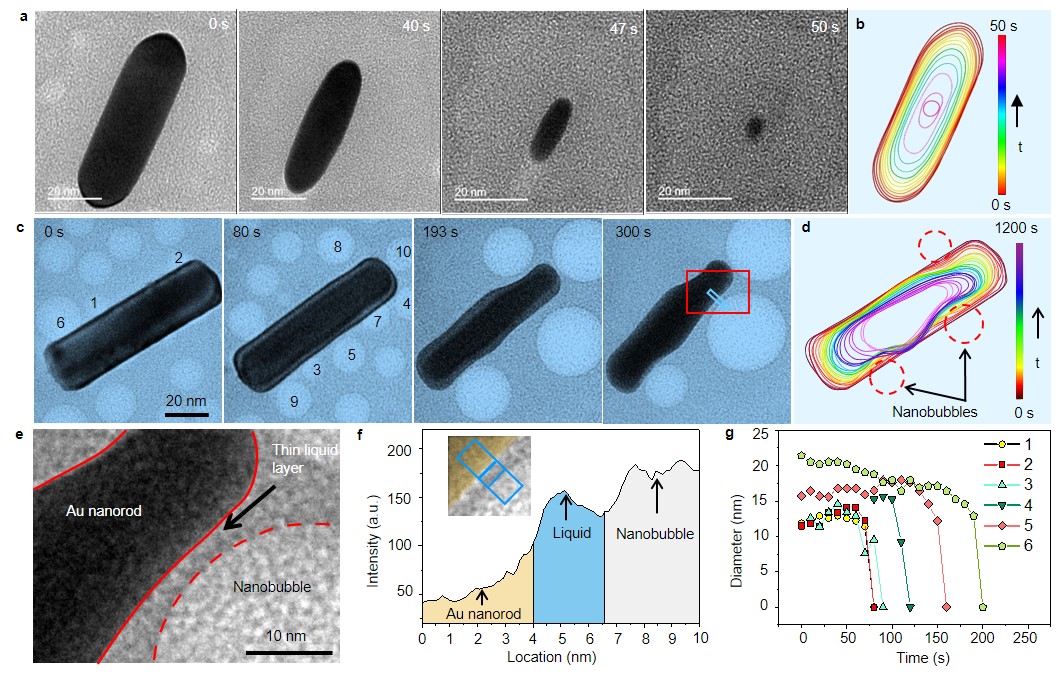
Fig. 2. a, Sequence of in situ TEM images showing the morphology changes of Au nanorod during etching without nanobubbles. b, The corresponding contour map highlights the uniform shape evolution of the nanorod. c, Time sequential TEM micrographs show the real-time shape evolution of the nanorod during the etching. Images are extracted from Video 1. d, The corresponding contour map highlights the indents developed near the nanobubbles. e, The enlarged figure (marked with red rectangle in a) clearly shows the thin liquid layer between the nanorod and nanobubble. f, Intensity profile of the selected area shown in c. g, Diameter changes of nanobubbles with time. The numbers represent the different nanobubbles marked in a.
It illustrates two novel findings. First, an accelerated reaction could be expected once the ultra-thin liquid layer between the solid and gas is formed instead of delivering gases to the solid surface directly. Second, different from the conventional sense that the closer the bubble is to the solid, the faster the solid-liquid-gas reaction performs, it shows no obvious accelerating effect until the distance between nanobubble and nanorod is reduced to a critical range. Further quantitative results show that the local reaction could be dramatically accelerated by up to 20 times when the distance between the gas bubbles and the solid (nanorods) is less than 1 nm (Fig. 3). Combining with MD simulations, we propose a thorough solid-liquid-gas reaction pathway, which includes gas transport through diffusion (>1 nm), adsorption (<1 nm) and surface reaction, and highlights the critical role of the thin liquid layer (less than 1 nm) between the solid and gas in significantly enhancing the reaction rate at triple phase regions.
This finding makes it possible to improve the controllability of the etching direction in wet etching. In addition, we propose several methods suitable for different scenarios to enhance the three-phase reaction, which is of great significance for the future regulation of micro-nano fabrication and heterogeneous catalysis involving solid liquid gas three-phase.
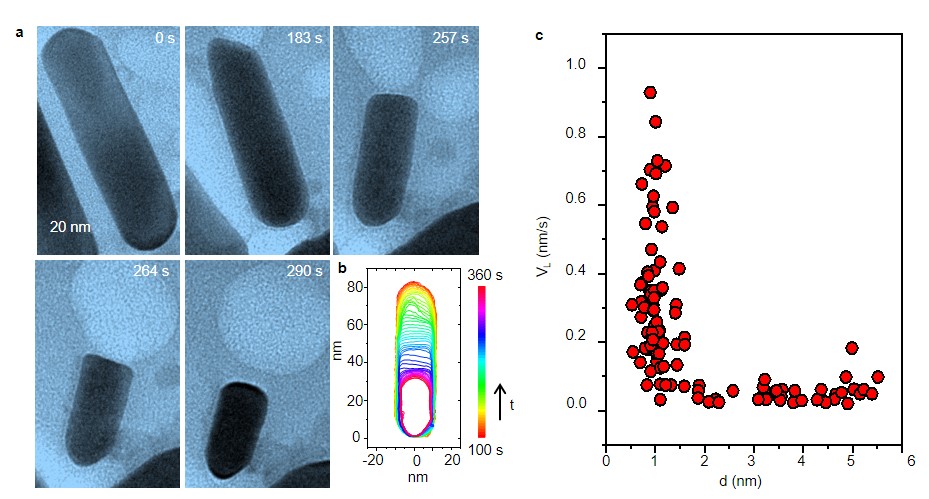
Fig. 3. Real-time observation of a gold nanorod etching with an O2 gas nanobubble at the end of the nanorod. a, Sequential TEM images of the Au nanorod during etching. b, The time-labeled contours 5 show the shape evolution of the gold nanorod in a. c, A statistic plot showing the longitudinal etching rate as a function of the distance between the nanobubble and nanorod surface.
For more details, please check out our paper “ Solid-liquid-gas reaction accelerated by gas molecule tunnelling-like effect” in Nature Materials.
Link: https://www.nature.com/articles/s41563-022-01261-x
Follow the Topic
-
Nature Materials
A monthly multi-disciplinary journal that brings together cutting-edge research across the entire spectrum of materials science and engineering, including applied and fundamental aspects of the synthesis/processing, structure/composition, properties and performance of materials.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in