
Hornworts, liverworts, and mosses are three early diverging clades of land plants, together composing the bryophytes. Bryophytes are often referred to as the early land plants marking the transition from freshwater to a terrestrial ecosystem. The timing of the conquest of land and the relationships among the main lineages of plants have been a subject of debate for long. Because bryophytes emerged from the early split in the diversification of land plants, they are key to study early land plant evolution. In this context, hornworts have drawn the most attention due to their traits linking to algae, unique associations with cyanobacteria and may be reminiscent of some early and now extinct lineages of early land plants.
Hornworts were the only bryophyte lineage for which a high-quality reference genome was absent for comparative studies, which is essential to extend our understanding of land plant evolution. The team of Prof. Zhi-Duan Chen from State Key Laboratory of Systematic and Evolutionary Botany, Institute of Botany, Chinese Academy of Sciences (IBCAS) launched the hornwort-genome sequencing project in 2008. The study represents a contribution by more than 10 years of exhausted efforts and collaboration of 30 scientists from China, Belgium and United States. Whole-genome sequencing of hornwort Anthoceros angustus (Anthocerotaceae) (Figure 1) was carried out and a combination of de novo, homology-based, and RNA sequence-based predictions was used to obtain gene models for the A. angustus genome1. This genome completes the sampling of all major land plant lineages, provides insight into the unique genomic features of hornworts and their molecular adaptations to live on land.

Figure 1. Anthoceros angustus. Photograph by Li Zhang
Genome assembly and annotation
Chen and the collaborators sequenced the genome of Anthoceros angustus using a combination of Illumina and Oxford Nanopore high-throughput sequencing systems. This sequencing strategy facilitated high quality assembly, an optimized assembly of 119 Mb with a contig N50 length of 796.64 kb and a scaffold N50 length of 1.09 Mb was obtained, indicating the genome size may be the smallest among land plants. In total, we predicted 14,629 protein-coding genes and identified 30 known mature miRNAs, 180 novel mature miRNAs, 347 tRNAs, 94 rRNAs, and 83 snRNAs in the A. angustus genome.
Comparative genomic analysis
Comparative genomic analysis shows that the genome of A. angustus has lost many gene families and comparatively few gains. The genome has much lower percentage of multi-copy gene families than that of single-copy gene families, implying low genetic redundancy. Although the Anthoceros genome is small with low redundancy, expansions are observed in gene families related to RNA editing, UV protection and desiccation tolerance. Phylogenetic inferences from 85 single-copy nuclear genes sampled for A. angustus and 18 other green plants resolve hornworts (A. angustus), mosses (P. patens) and liverworts (M. polymorpha) as a monophyletic group, with hornworts sister to mosses and liverworts. Integration of data in phylogenomic inferences confirms that hornworts are resolved as sister to the setaphytes within a monophyletic bryophyte2-6 (Figure 2).
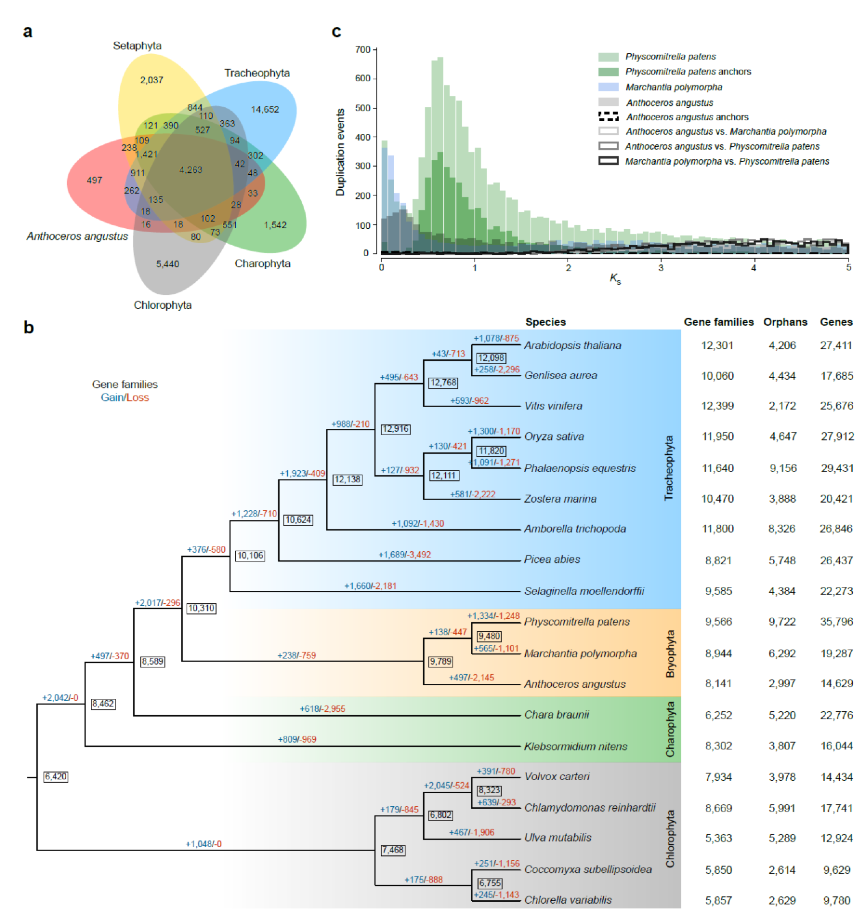
Figure 2. Comparative genomic analysis of Anthoceros angustus and other 18 plant species. a, Comparison of the number of gene families identified by OrthoMCL. b, Gene family gain (+) / loss (-) among 19 green plants. c, Comparison of whole paranome, anchor pair and one-to-one ortholog KS distributions across the three bryophyte species (Physcomitrella patens, Marchantia polymorpha and A. angustus).
Transcription factors
Unlike other land plants, the genome of Anthoceros, has a small number of TF genes and lacks evidence of having undergone a WGD, which supports the hypothesis that the WGD is an important mechanism for expanding of TF families7. In A. angustus, most genes involved in the development of plant body plans have a single copy, and a few A. angustus TF gene families even lost a subset of duplicates. A. angustus exhibits low redundancy for genes shaping the plant body plan (Fig. 2b). Such limited toolkit may be characteristic of the ancestor to bryophytes and hence perhaps of the earliest land plants with a dominant thalloid gametophyte, and provide the foundation to explaining the architectural simplicity of these plants. Although the genome of A. angustus seems poor in genes composing the network underlying the development of its body plan, the TF gene families linked to responses to terrestrial environmental contribute to lineage-specific gene expansions8.
Horizontal gene transfer
Bryophytes form symbioses with diverse bacteria and fungi9. The genome of A. angustus also bears the signatures of horizontally transferred genes from bacteria and fungi, in particular of genes operating in stress response, oxidation-reduction and metabolic pathways. Meanwhile, the gene inventory in A. angustus extended mainly through tandem duplication and HGT. In particular, the expansion of specific gene families and the acquisition of foreign genes have provided additional metabolic abilities in hornworts that likely facilitated the survival in a terrestrial environment.
As land pioneers, the three bryophyte groups form a well-supported monophyletic lineage, with hornworts sister to liverworts and mosses. The genome of hornwort A. angustus shows no evidence of WGDs and low genetic redundancy for networks underlying plant body plan. Meanwhile, the gene inventory in A. angustus extended mainly through tandem duplication and HGT. In particular, the expansion of specific gene families and the acquisition of foreign genes have provided additional metabolic abilities in hornworts that likely facilitated the survival in a terrestrial environment. Importantly, the hornwort genome fills the gap of early land plant study, provides a useful model for studying early land plant evolution and the mechanism of plant terrestrial adaptation.
This post is jointly written by Zhong-Jian Liu and Di-Yang Zhang at Key Laboratory of National Forestry and Grassland Administration for Orchid Conservation and Utilization at College of Landscape Architecture, Fujian Agriculture and Forestry University.
Reference
1. Zhang, J., Fu, X. X., Li, R. Q., Zhao, X, Liu, Y., Li, M. H., Zwaenepoel, A., Ma, H., Goffinet, B., Guan, Y. L., Xue, J. Y., Liao, Y. Y., Wang, Q. F., Wang, Q. H., Wang, J. Y., Zhang, G. Q., Wang, Z. W., Jia, Y., Wang, M. Z., Dong, S. S., Yang, J. F., Jiao, Y. N., Guo, Y. L., Kong, H. Z., Lu, A. M., Yang, H. M., Zhang, S. Z., Van de Peer, Y., Liu, Z. J., Chen, Z. D. The hornwort genome and early land plant evolution. Nature Plants https://doi.org/10.1038/s41477-019-0588-4 (2020)
2. Yue, J., Hu, X., Sun, H., Yang, Y. & Huang, J. Widespread impact of horizontal gene transfer on plant colonization of land. Nat. Commun. 3, 1152 (2012).
3. Puttick, M. N. et al. The interrelationships of land plants and the nature of the ancestral embryophyte. Curr. Biol. 28, 733–745 (2018).
4. Qiu, Y. L. et al. The deepest divergences in land plants inferred from phylogenomic evidence. Proc. Natl Acad. Sci. USA 103, 15511–15516 (2006).
5. Wickett, N. J. et al. Phylotranscriptomic analysis of the origin and early diversification of land plants. Proc. Natl Acad. Sci. USA 111, E4859–E4868 (2014).
6. Cox, C. J., Li, B., Foster, P. G., Embley, T. M. & Civan, P. Conflicting phylogenies for early land plants are caused by composition biases among synonymous substitutions. Systematic Biol. 63, 272–279 (2014).
7. Liu, Y., Cox, C. J., Wang, W. & Goffinet, B. Mitochondrial phylogenomics of early land plants: mitigating the effects of saturation, compositional heterogeneity, and codon-usage bias. Systematic Biol. 63, 862–878 (2014).
8. Lang, D. et al. Genome-wide phylogenetic comparative analysis of plant transcriptional regulation: a timeline of loss, gain, expansion, and correlation with complexity. Genome Biol. Evol. 2, 488–503 (2010).
9. Cenci, A. & Rouard, M. Evolutionary analyses of GRAS transcription factors in angiosperms. Front. Plant Sci. 8, 273 (2017).
Follow the Topic
-
Nature Plants
An online-only, monthly journal publishing the best research on plants — from their evolution, development, metabolism and environmental interactions to their societal significance.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in