The role of Cu1–O3 species in single-atom Cu/ZrO2 catalyst for CO2 hydrogenation
Published in Chemistry

The massive burning of fossil fuels in recent decades has led to a dramatic increase in CO2 greenhouse gas emissions, which has led to serious environmental problems, including global warming, sea level rise and ocean acidification1. Therefore, it is urgent to find an effective way to alleviate this severe situation. One of the most promising ways to solve above problems is to take CO2 as the carbon source and convert it into high-value-added chemicals through catalytic reaction. It can not only significantly reduce the excessive emission of CO2 greenhouse gases but also effectively alleviate the energy crisis2, 3, 4. As a basic chemical raw material, methanol can be used to synthesize chemical products with higher value, such as dimethyl ether5 and low-carbon olefin6. It can be seen that the conversion of CO2 to methanol has very important industrial value. Therefore, in the past few decades, researchers have done a lot of work to find catalysts with high catalytic performance, including metal-metal oxide catalysts7, metal-oxide solid solutions8 and metal alloys9.
Among CO2 hydrogenation catalysts, copper-based catalysts have been widely studied due to their excellent performance and low price10. However, it is difficult to identify its real active site precisely due to the complex structure of Cu-based catalysts. The variable valence of copper species and the reductive reaction atmosphere means copper species are likely to exist in the form of mixed valence states during the reaction process. There are conflicting reports in the literature suggesting that Cu0, Cu+ or Cuδ+ may be the active center of the reaction11, 12, 13. In addition, different particle sizes of copper species also significantly affect its catalytic performance14, 15. These different factors have a great influence on the coordination structure of the copper itself, which makes it difficult to study the actual active copper site in CO2 hydrogenation. Despite substantial progress in understanding the active site of copper catalysts, there is still much controversy regarding the structure-property correlation between catalysts and reactions.
For the above reasons, it is essential to find a copper-based catalyst with a clear and stable structure to further study the potential relationship between catalysis and activity. As reported, the single-atom catalyst is an ideal model for the structure-activity relationship because of its definite active structure16. However, the literature on single-atom catalysts for heterogeneous thermal catalytic CO2 hydrogenation to methanol is very scarce.
Here, we designed and prepared a stable single-atom Cu-based catalyst (Cu1/amorphous-ZrO2) for hydrogenation of CO2 to methanol at low temperature (180℃). Compared with the traditional Cu-Zr catalysts, the single-atom Cu-Zr catalyst showed a higher TOF value for methanol and 100% methanol selectivity. Excellent activity usually comes from the unique structure of the catalyst, and the results of fitting X-ray absorption spectra and advanced theoretical calculations (walking surface potential) show that the single-atom catalyst has a Cu1-O3 quasi-planar structure during the reaction, and the Cu species are still in the cationic state (~1.4+). Moreover, the structure of the active site is very stable, and in terms of activity, no obvious deactivation was found after 100 hours of reaction. With the increase of Cu loading, the catalytic performance of the samples and the coordination structure of copper species are changed at the same time. It was finally determined that the Cu1-O3 site of copper was the only active site for methanol synthesis in the low temperature reaction condition, and the nanoclusters and nanoparticles were active sites for CO formation, while larger copper particles have no activating ability for CO2. The schematic diagram is as follows: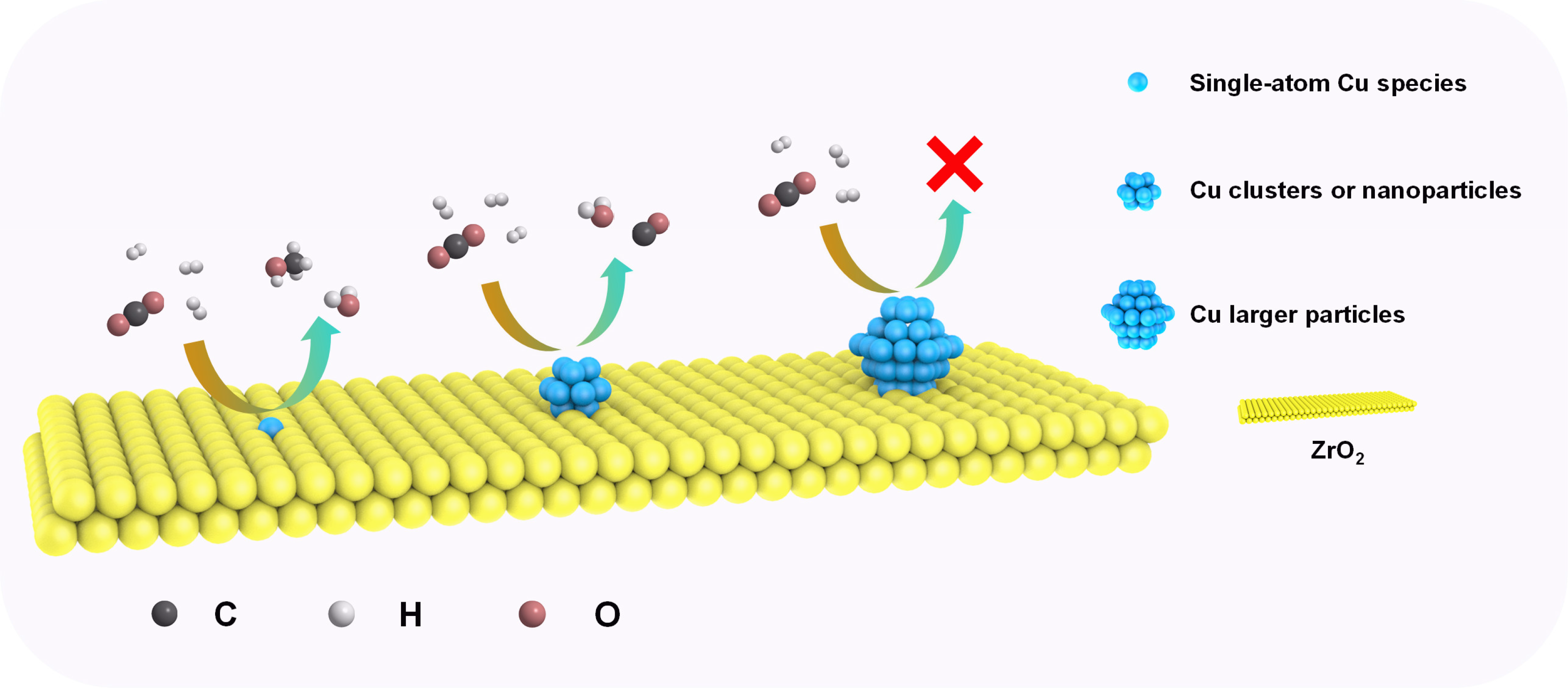
Studies have shown that the gases used in the pretreatment or reaction process greatly affect the structure of catalysts, including active species migration. In our catalytic system, the internal copper species gradually migrate to the surface of the catalyst during the reaction. The migration phenomenon is more obvious in the samples with high Cu loading, which may be the reason for the long activation period of the reaction.
The hydrogenation of CO2 to methanol usually includes two important mechanisms, namely HCOO* mechanism and COOH* mechanism. In our Cu/ZrO2 system, in situ high-pressure infrared spectroscopy and theoretical calculations indicate that the reaction prefers to follow the formate hydrogenation route, and the hydrogenation of HCOO* to CH3O* is the rate-limiting step of the reaction.
The characteristic geometry and unique activity revealed in the copper single-atom catalyst provides a deep understanding of copper-catalyzed CO2 hydrogenation and will guide future applications of single-atom catalysts in thermal catalytic CO2 transformations.
- Jenkinson DS, et al. Model estimates of CO2 emissions from soil in response to global warming. Nature 351, 304-306 (1991).
- Hu J, et al. Sulfur vacancy-rich MoS2 as a catalyst for the hydrogenation of CO2 to methanol. Nat Catal 4, 242-250 (2021).
- Wang Y, et al. Rationally Designing Bifunctional Catalysts as an Efficient Strategy To Boost CO2 Hydrogenation Producing Value-Added Aromatics. ACS Catal 9, 895-901 (2018).
- Liu C, et al. Gallium nitride catalyzed the direct hydrogenation of carbon dioxide to dimethyl ether as primary product. Nat Commun 12, 2305 (2021).
- Feng W-H, et al. Insights into Bimetallic Oxide Synergy during Carbon Dioxide Hydrogenation to Methanol and Dimethyl Ether over GaZrOx Oxide Catalysts. ACS Catal 11, 4704-4711 (2021).
- Gao P, et al. Direct Production of Lower Olefins from CO2 Conversion via Bifunctional Catalysis. ACS Catal 8, 571-578 (2017).
- Behrens M, et al. The Active Site of Methanol Synthesis over Cu/ZnO/Al2O3 Industrial Catalysts. Science 336, 893-897 (2012).
- Martin O, et al. Indium Oxide as a Superior Catalyst for Methanol Synthesis by CO2 Hydrogenation. Angew Chem Int Ed 55, 6261-6265 (2016).
- Sharafutdinov I, et al. Intermetallic compounds of Ni and Ga as catalysts for the synthesis of methanol. J Catal 320, 77-88 (2014).
- Graciani J, et al. Highly active copper-ceria and copper-ceria-titania catalysts for methanol synthesis from CO2. Science 345, 546-550 (2014).
- Samson K, et al. Influence of ZrO2 Structure and Copper Electronic State on Activity of Cu/ZrO2 Catalysts in Methanol Synthesis from CO2. ACS Catal 4, 3730-3741 (2014).
- Wu C, et al. Inverse ZrO2/Cu as a highly efficient methanol synthesis catalyst from CO2 hydrogenation. Nat Commun 11, 5767 (2020).
- Yang H, et al. A Highly Stable Copper-Based Catalyst for Clarifying the Catalytic Roles of Cu0 and Cu+ Species in Methanol Dehydrogenation. Angew Chem Int Ed 57, 1836-1840 (2018).
- Zhu Y, et al. Copper-zirconia interfaces in UiO-66 enable selective catalytic hydrogenation of CO2 to methanol. Nat Commun 11, 5849 (2020).
- Zhou H, et al. Engineering the Cu/Mo2CTx (MXene) interface to drive CO2 hydrogenation to methanol. Nat Catal 4, 860-871 (2021).
- Qiao B, et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat Chem 3, 634-641 (2011).
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in