The underestimated role of bone marrow T cells as important predictor for treatment response and survival in acute myeloid leukemia
Published in Cancer
Background
The concept of cancer immunosurveillance remained controversial through much of the 20th century. In last two decades, however, this field has progressed rapidly and led to breakthrough therapeutic developments mainly harnessing T cells. Amongst these, immune checkpoint inhibition and chimeric antigen receptor T cells have been the most transformative approaches.1,2 The use of these immunotherapies spans a broad range of solid tumors and extends to lymphoid cancers.
In acute myeloid leukemia (AML), the role of the immune system during leukemogenesis has been scarcely investigated. The ability of T cells to recognize AML antigens has been formally proven by the success of allogeneic hematopoietic stem cell transplantation (HSCT) that is a curative approach in this disease3,4 although – due to its high treatment-related mortality - reserved to younger patients without comorbidities. Other successful drugs in AML treatment such as hypomethylating agents may also have immunomodulatory effects.5,6 Yet, the reported results are partly contradictory and far from being elucidated mechanistically. To pave the way for immunotherapies beyond HSCT in AML patients, a better understanding of T cell biology in this disease is needed.
What did we investigate?
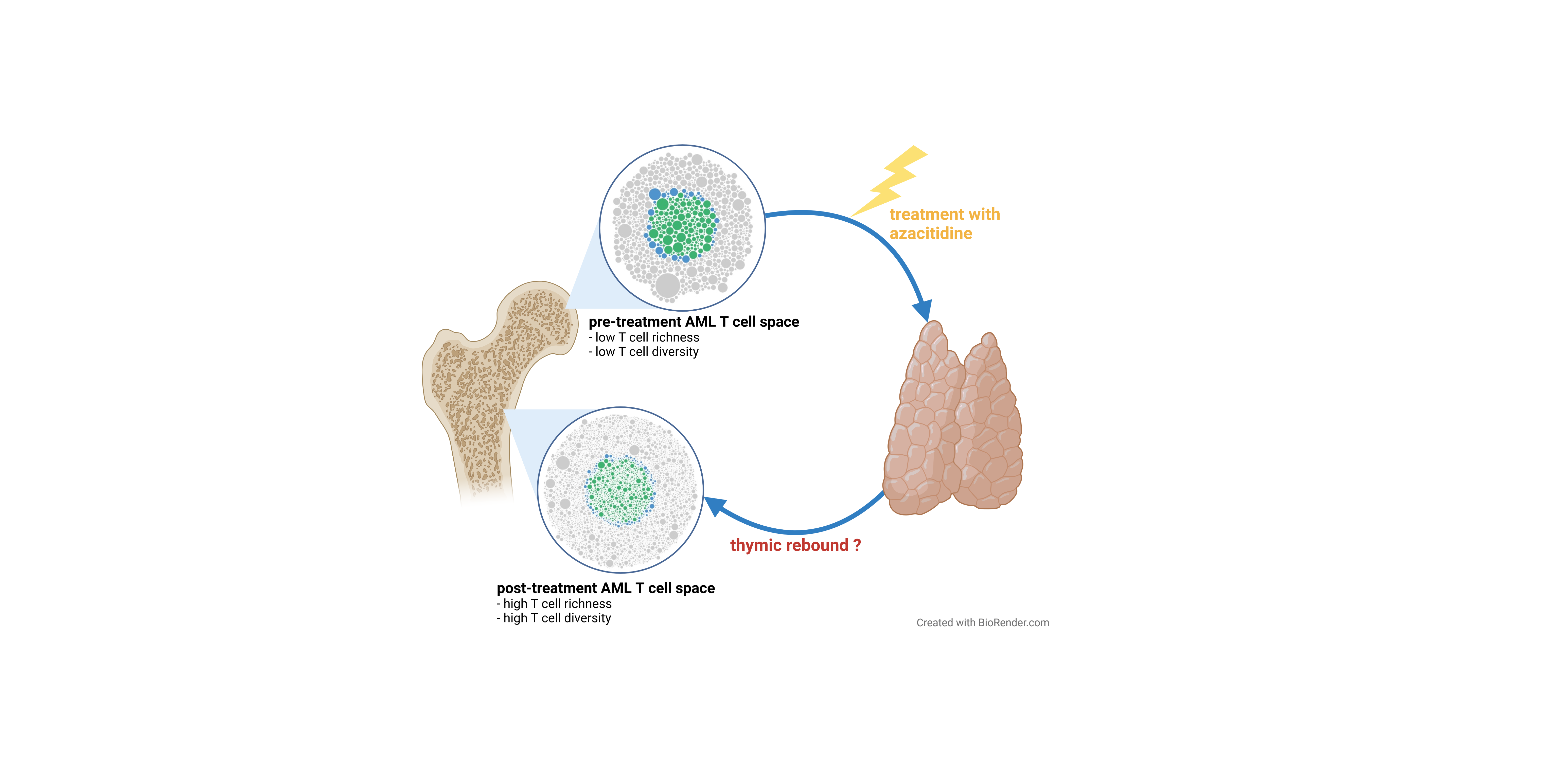
In our study, we recorded T cell repertoire fingerprints from bone marrow aspirates of AML patients by next-generation sequencing. Our patients were treated on a clinical trial combining the hypomethylating agent azacitidine with intensive chemotherapy in a response-based sequential approach. By investigating matched bone marrow samples taken before treatment initiation and very early on treatment (day 15), we searched for characteristics of the T cell fingerprint predictive for response to treatment with hypomethylating agents that may shed light on the effect of these drugs on the bone marrow T cell architecture.
What did we find?
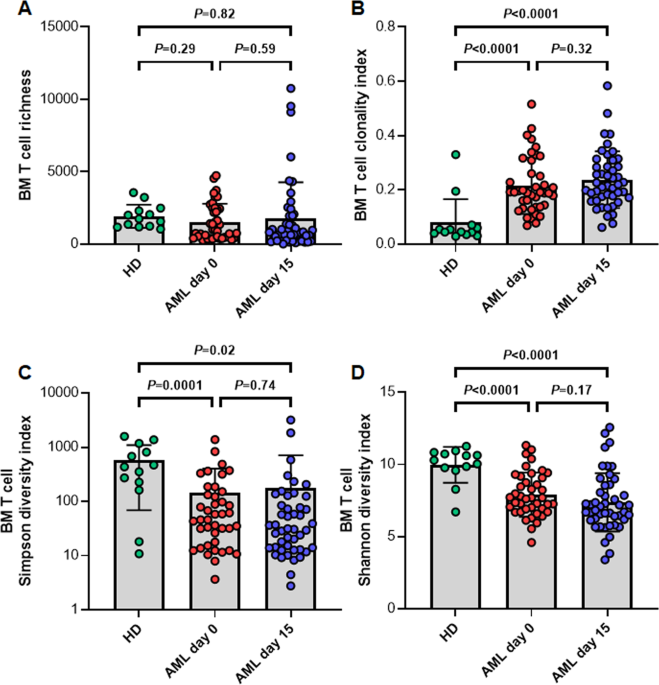
We found that the richness and diversity of the T cell space in the bone marrow of AML patients was a predictor of treatment success. Patients that showed particularly diverse T cell repertoires as found in healthy individuals had longer event-free and overall survival compared to AML patients with less diverse repertoires. Additionally, we showed that about one quarter of AML patients experienced a boost in T cell richness in response to azacitidine. These patients also showed improved treatment benefit. The azacitidine-induced broadening of the bone marrow T cell space was accompanied by a skewed usage of TRB V and J genes. This skewing of T cell receptor gene usage across patients was suggestive of directional shifts in the repertoire reflecting expansion of T cells with similar epitope recognition. In support of this hypothesis, we also found a high number of T cell clusters in these patients. Clusters are groups of T cells that share T cell receptor properties in the crucial parts of the paratope with predicted recognition of identical epitopes. These T cell clusters expanding in patients with favorable treatment response likely have functional relevance since they showed a low generation probability and thus were unlikely to emerge by chance. Very interestingly, the T cell repertoire diversity and richness metrics were independent of the blast counts of these patients and they remained a strong favorable prognostic factor upon multivariate testing.
Taken together, these results imply that high up-front T cell diversity may facilitate antigen-driven anti-leukemic immune responses in AML patients. Moreover, azacitidine is able to boost T cell richness even in some patients that show rather clonal T cell repertoires prior to treatment. The boost of T cell richness in the bone marrow niche is an interesting pattern compatible with some sort of thymic reactivation in these patients in response to treatment. Overall, this data underlines the so far underestimated prognostic role of the bone marrow T cell niche in AML.
Where do we go from here?
We demonstrated that AML patients with a “healthy-like” bone marrow T cell fingerprint either before treatment or as a result of treatment with the hypomethylating agent azacitidine experienced improved survival. The prolonged survival in these patients was independent from the applied treatment regimen (only azacitidine versus azacitidine plus chemotherapy). Consequently, immunologically responsive patients of borderline age and/or fitness should be spared unnecessary chemotherapy, preventing severe toxicities while still offering excellent prognosis. To clinically implement such strategies, the immunological analyzes used in our study should be prospectively validated in future clinical trials.
Interestingly, the very similar broad immune repertoire metrics associated with response to immunomodulatory treatment have also been found by our group in other cancers. One such example is a cohort of patients with prostate cancer treated on several trial protocols with immune checkpoint inhibiting antibodies.7 This cohort included both patients with microsatellite stable and unstable tumors. Their T cell fingerprints determined from peripheral blood showed increased treatment benefit in patients with high repertoire diversity and richness compared to their peers. The validation of this favorable T cell pattern across different disease and immunotherapy settings is a priority of our team that we will work on intensively in the next years.
References
- Hong M, Clubb JD, Chen YY. Engineering CAR-T Cells for Next-Generation Cancer Therapy. Cancer cell 2020; 38: 473–488.
- Morad G, Helmink BA, Sharma P, Wargo JA. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell 2021; 184: 5309–5337.
- Horowitz MM, Gale RP, Sondel PM, Goldman JM, Kersey J, Kolb HJ, et al. Graft-versus-leukemia reactions after bone marrow transplantation. Blood 1990; 75: 555–562.
- Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jacobsen N, Arcese W, et al. Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood 1995; 86: 2041–2050.
- Goodyear OC, Dennis M, Jilani NY, Loke J, Siddique S, Ryan G, et al. Azacitidine augments expansion of regulatory T cells after allogeneic stem cell transplantation in patients with acute myeloid leukemia (AML). Blood 2012: 3361–3369.
- Yang H, Bueso-Ramos C, DiNardo C, Estecio MR, Davanlou M, Geng Q-R, et al. Expression of PD-L1, PD-L2, PD-1 and CTLA4 in myelodysplastic syndromes is enhanced by treatment with hypomethylating agents. Leukemia 2014: 1280–1288.
- Simnica D, Smits M, Willscher E, Fanchi LF, Kloots ISH, van Oort I, et al. Responsiveness to Immune Checkpoint Inhibitors Is Associated With a Peripheral Blood T-Cell Signature in Metastatic Castration-Resistant Prostate Cancer. JCO precision oncology 2020; 4: 1374–1385.
Follow the Topic
-
Blood Cancer Journal
This journal seeks to publish articles of the highest quality related to hematologic malignancies and related disorders.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in