TIF1β activates FOSL1 and drives BCR::ABL1 myeloid leukemia.
Published in Cancer, Cell & Molecular Biology, and General & Internal Medicine
Introduction
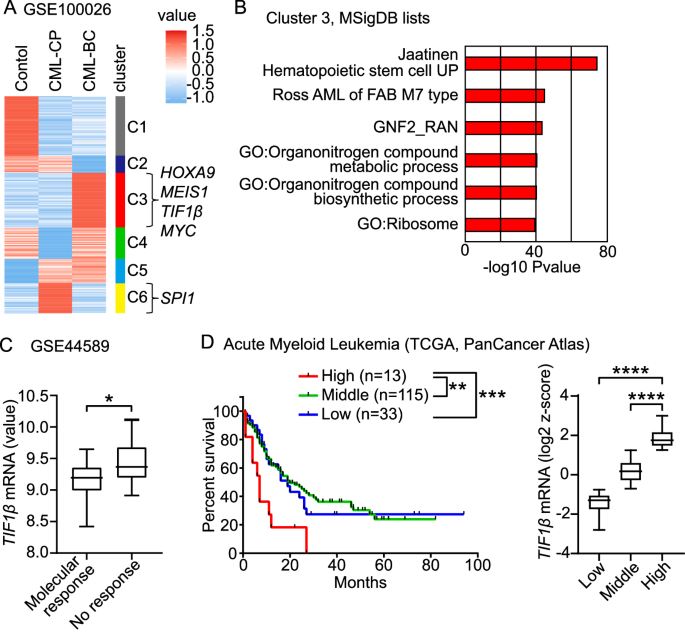
TIF1β/KAP1/TRIM28, a chromatin modulator, both represses and activates the transcription of genes in normal and malignant cells (1). Analyses of datasets on leukemia patients revealed that the expression level of TIF1β was increased in patients with chronic myeloid leukemia (CML) at the blast crisis and acute myeloid leukemia. The BCR::ABL1 fusion protein develops both CML and B-cell acute lymphoblastic leukemia (B-ALL) (2). In the early 2000s, the advent of BCR::ABL1 tyrosine kinase inhibitors (TKI) markedly improved the outcomes of patients with CML in the chronic phase (CML-CP) (3–5). However, the TKI therapy cannot be expected to effectively eradicate advanced CML-blast crisis (CML-BC) (6). While additional genetic mutations and chromosomal anomalies are known to promote the blast crisis in patients with CML, the mechanisms by which the BCR::ABL1 protein regulates chromatin and the transcription of genes to drive the blast crisis of CML remain unclear. In this study, we generated a BCR::ABL1 conditional knock-in (KI) mouse model, which developed aggressive myeloid leukemia, and investigated whether the deletion of the Tif1β gene inhibited the progression of myeloid leukemia (7).
Major Findings
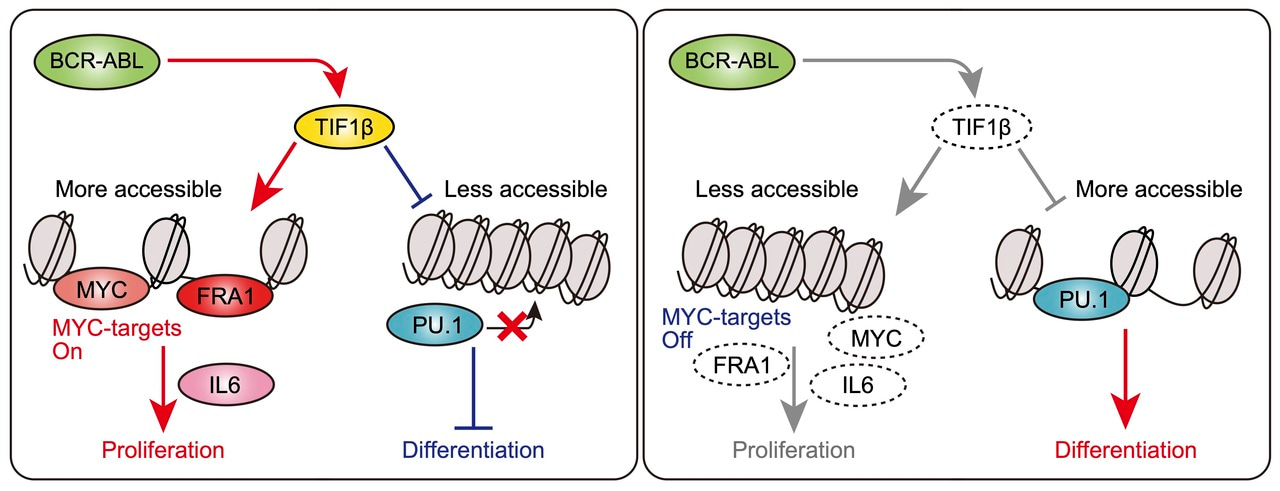
- TIF1β opened chromatin and activated the transcription of oncogenes in BCR::ABL1-expressing stem cells. We confirmed that the deletion of the Tif1β gene inhibited the progression of myeloid leukemia and showed longer survival than that in BCR::ABL1KI mice, suggesting that Tif1β drove the progression of BCR::ABL1-induced leukemia. The deletion of Tif1β decreased the expression levels of both MYC-target genes and TIF1β-target genes and chromatin accessibility peaks enriched with the Fosl1/Fra1-binding motif in BCR::ABL1 KI stem cells, but also increased chromatin accessibility peaks enriched with PU.1. In human BCR::ABL1-expressing leukemic cells, TIF1β directly bound to the promoters of proliferation genes, such as FOSL1, in which TIF1β and FOSL1 bound to adjacent regions of chromatin. Therefore, Tif1β modulated chromatin in BCR::ABL1-expressing hematopoietic stem cells (HSCs) to increase chromatin accessibility of the AP-1 TF family but decrease those of myeloid TFs, such as PU.1. We then investigated the roles of the Fosl1 gene in primary BCR::ABL1 KI cells and found that the Fosl1 gene was required for the enhanced growth of BCR::ABL1-expressing leukemia under in vitro and in vivo conditions. Tif1β and Fosl1 interacted to activate the leukemic transcriptional program in and cellular function of BCR::ABL1 KI stem cells and drove the progression of myeloid leukemia.
- The loss of Tif1β sensitized the BCR::ABL1 leukemic cells to the TKI treatment. Based on that Tif1β opened chromatin and activated the transcription of oncogenes in BCR::ABL1-expressing HSCs to drive the progression of BCR::ABL1-induced myeloid leukemia, we investigated whether the Tif1β gene was required for the leukemia-initiating capacity after a treatment with a BCR::ABL1 TKI in our murine model. After the TKI treatment, BCR::ABL1 mice had a higher WBC count with the expansion of leukemic blast cells than TKI-treated BCR::ABL1 and DMSO-treated BCR::ABL1-Tif1β KO mice, whereas TKI-treated BCR::ABL1-Tif1β KO mice did not. Therefore, the deletion of Tif1β sensitized BCR::ABL1-expressing cells to the TKI treatment.
Conclusion
We demonstrated that Tif1β/Kap1/Trim28 opened chromatin and activated the transcription of genes, including Fosl1/Fra1, in HSCs to drive the progression of BCR::ABL1-induced myeloid leukemia. In addition, the deletion of Tif1β sensitized BCR::ABL1-induced leukemia to TKI treatments. We elucidated the molecular mechanism by which the Tif1β protein remodels BCR::ABL1-expressing leukemic stem cells via the formation of the Tif1β-Fosl1 axis and provide it as a new therapeutic rationale for advanced myeloid leukemia.
References
- Czerwińska P, Mazurek S, Wiznerowicz M. The complexity of TRIM28 contribution to cancer. J Biomed Sci. 2017;24(1):1–14.
- Foà R, Chiaretti S. Philadelphia Chromosome–Positive Acute Lymphoblastic Leukemia. New England Journal of Medicine. 2022;386(25):2399–411.
- Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S, et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Med. 1996;2(5):561–6.
- Druker BJ, Guilhot F, O’Brien SG, Gathmann I, Kantarjian H, Gattermann N, et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355(23):2408–17.
- Saußele S, Richter J, Hochhaus A, Mahon FX. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30(8):1638–47.
- Braun TP, Eide CA, Druker BJ. Response and Resistance to BCR-ABL1-Targeted Therapies. Cancer Cell. 2020;37(4):530–42.
- Morii M, Kubota S, Iimori M, Yokomizo-Nakano T, Hamashima A, Bai J, et al. TIF1β activates leukemic transcriptional program in HSCs and promotes BCR::ABL1-induced myeloid leukemia. Leukemia. 2024;38(6):1275–86.
Follow the Topic
-
Leukemia
This journal publishes high quality, peer reviewed research that covers all aspects of the research and treatment of leukemia and allied diseases. Topics of interest include oncogenes, growth factors, stem cells, leukemia genomics, cell cycle, signal transduction and molecular targets for therapy.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in