Transcription factor Nkx2-3 maintains the self-renewal of hematopoietic stem cells by regulating mitophagy
Published in Healthcare & Nursing
The problem we focused
In recent years, metabolic regulation of stem cell fate has become an increasing concern [1]. Under normal circumstances, hematopoietic stem cells (HSCs) mainly depend on anaerobic glycolysis rather than oxidative phosphorylation (OXPHOS) for energy supply [2, 3]. Previous studies, including our own work, have reported that abnormal activation of mitochondria will produce excessive metabolic byproducts, which disrupt HSC quiescence and function [4, 5]. Surprisingly, mitochondrial mass is higher in HSCs than their progenies, whereas how to maintain them in a low metabolic state is not completely understood.
Mitochondrial homeostasis is maintained by a combination of biosynthesis, fusion/fission and autophagy, also known as mitophagy [5-7]. In this study, we show for the first time that Nkx2-3/ULK1 pathway-mediated mitophagy plays an important role in sustaining the self-renewal capacity of HSCs. We find that the transcription factor Nkx2-3 is specifically enriched in HSCs and its deficiency results in gradual decreases in HSC number and long-term reconstitution ability in both mice and humans. Mechanistically, Nkx2-3 promotes the metabolic fitness of HSCs by directly regulating the transcription of ULK1, which is required to clear activated mitochondria via mitophagy. Collectively, our findings unveil new insights into the mitophagy regulation of HSCs.
What did we show?
We first performed single-cell RNA sequencing (scRNA-seq) analysis of murine hematopoietic stem progenitor cells (HSPCs) and identified 12 different cell populations. It was found that Nkx2-3 was highly expressed in the most primitive HSC population (C1 cluster). Indeed, its expression pattern was similar to previously identified HSC regulators, such as Egr1, Hlf, Hes1, Mecom, Nr4a1, etc. Of note, NKX2-3 expression was also highly enriched in human HSCs (Figure 1).
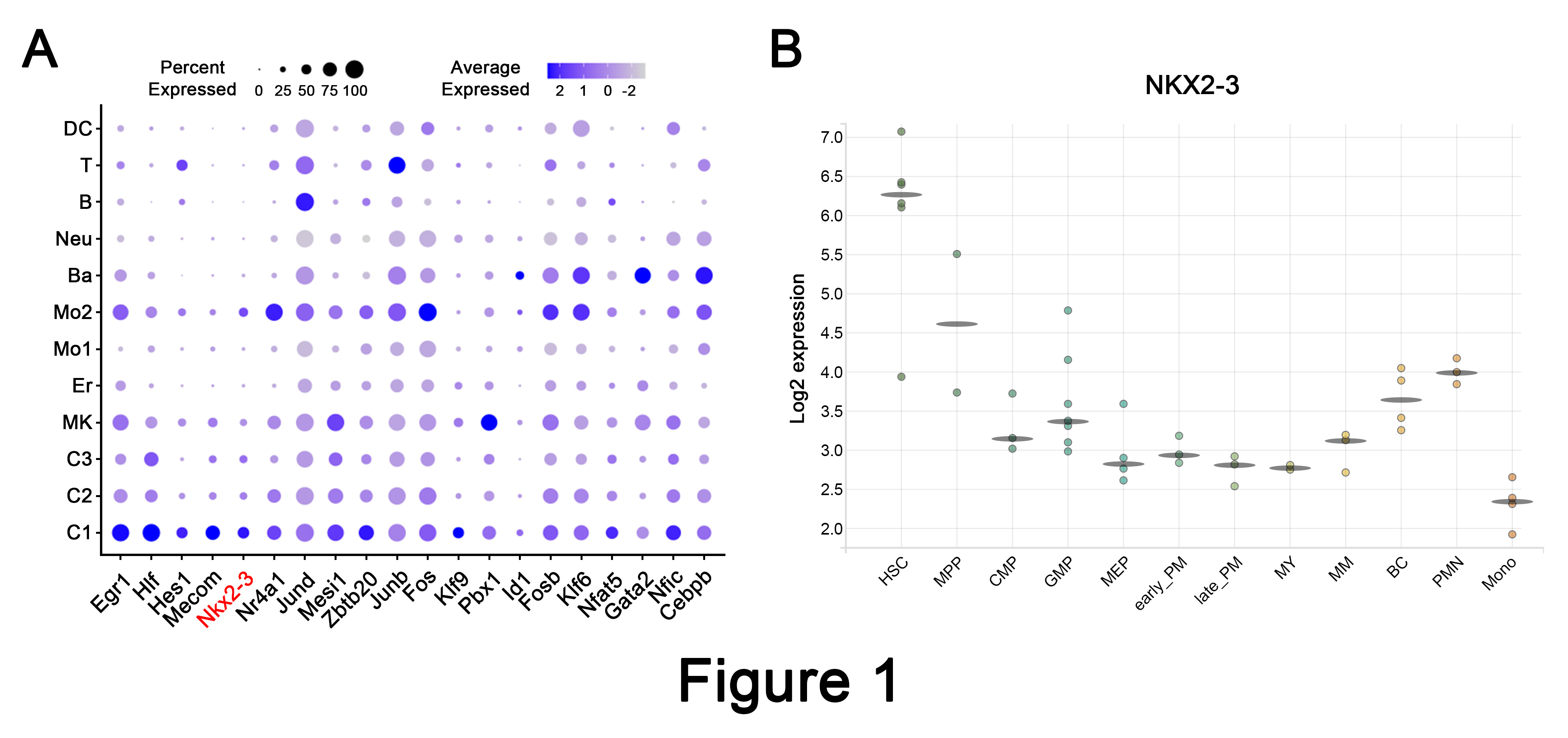
Figure 1. Nkx2-3 is preferentially expressed in primitive HSCs.
To determine the role of Nkx2-3 in HSCs, we used a conditional Nkx2-3 knockout mouse model (Nkx2-3fl/fl/Mx1-Cre). Intriguingly, we found that the number of HSCs was largely unchanged at 4 weeks but was significantly decreased at 20 weeks in Nkx2-3cKO mice after pIpC injection. To test whether Nkx2-3 deletion affects HSC potential to self-renew, we then performed a competitive bone marrow transplantation assay and found that the long-term hematopoietic reconstruction ability of HSCs was remarkably dampened after Nkx2-3 ablation. In contrast, overexpression of Nkx2-3 improved HSC function (Figure 2).
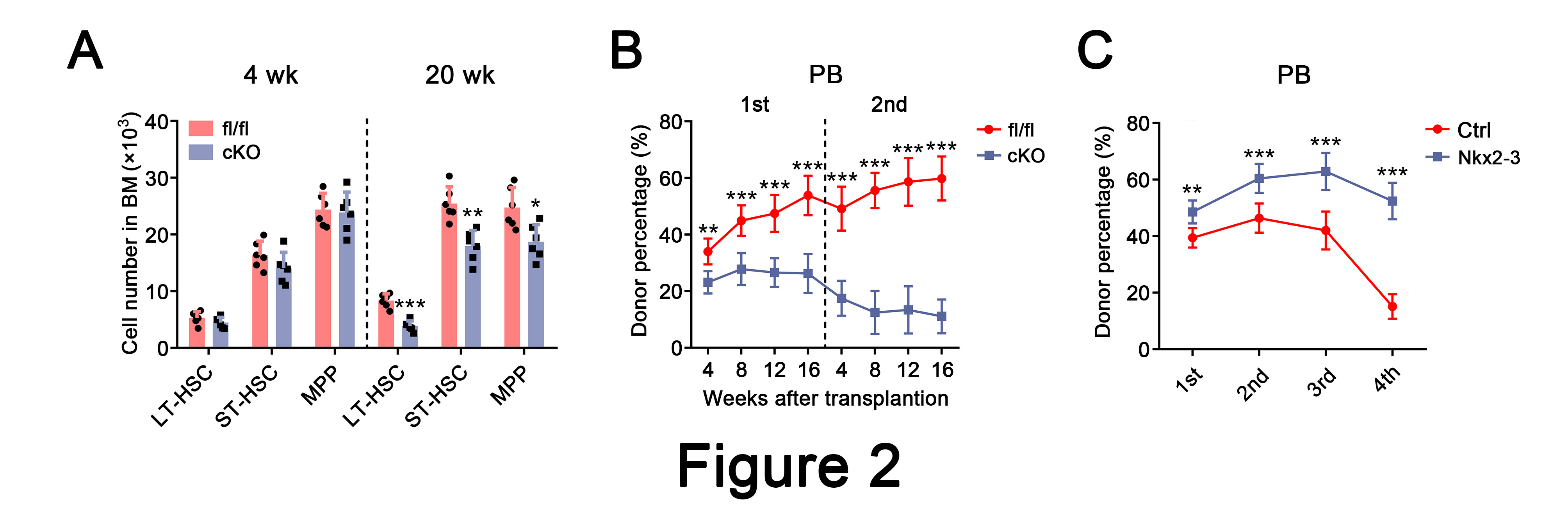
Figure 2. Nkx2-3 is required to regulate the self-renewal of HSCs.
Further investigations showed that Nkx2-3 deficiency resulted in the accumulation of activated mitochondria in HSCs due to impaired mitophagy. Importantly, we found a significant decrease in the expression of ULK1, a critical mitophagy regulator, in HSCs following Nkx2-3 deletion. Moreover, mechanistic studies revealed that Nkx2-3 could directly bind to the Ulk1 promoter region and thus regulated its expression in HSCs (Figure 3).
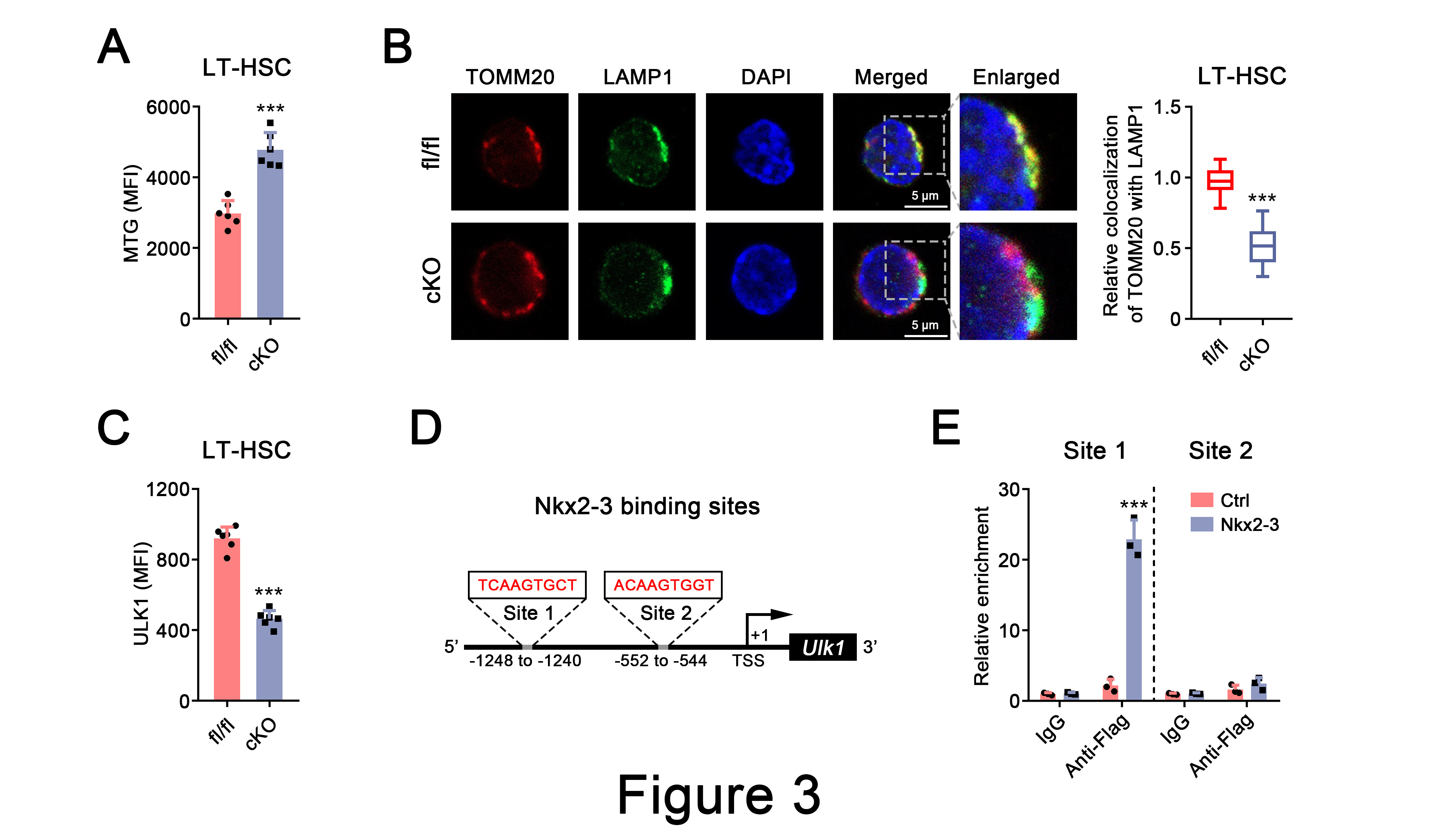
Figure 3. Nkx2-3 deficiency impairs ULK1-dependent mitophagy in HSCs.
Conclusions and implications of our study
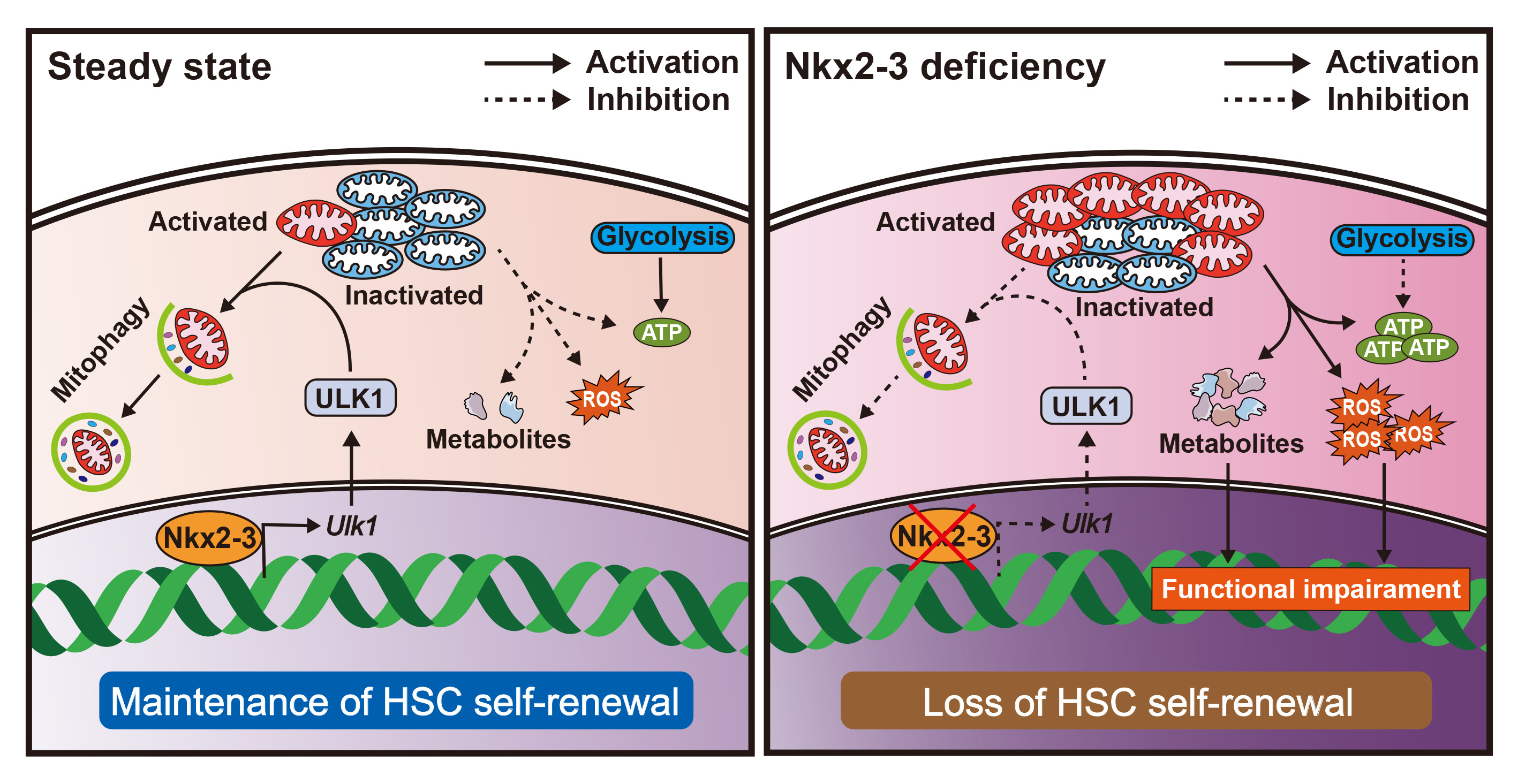
HSCs exhibits a unique capacity to self-renew and differentiate into all blood cells throughout the lifetime, whereas how to prevent HSC pool exhaustion during long-term hematopoietic output is not fully understood. The present study uncovers the significant role of Nkx2-3/ULK1/mitophagy axis in promoting the long-term maintenance of HSCs by regulating metabolic homeostasis (Figure 4). Therefore, our data provide a promising strategy to improve the function of HSCs when suffering from proliferative stress.
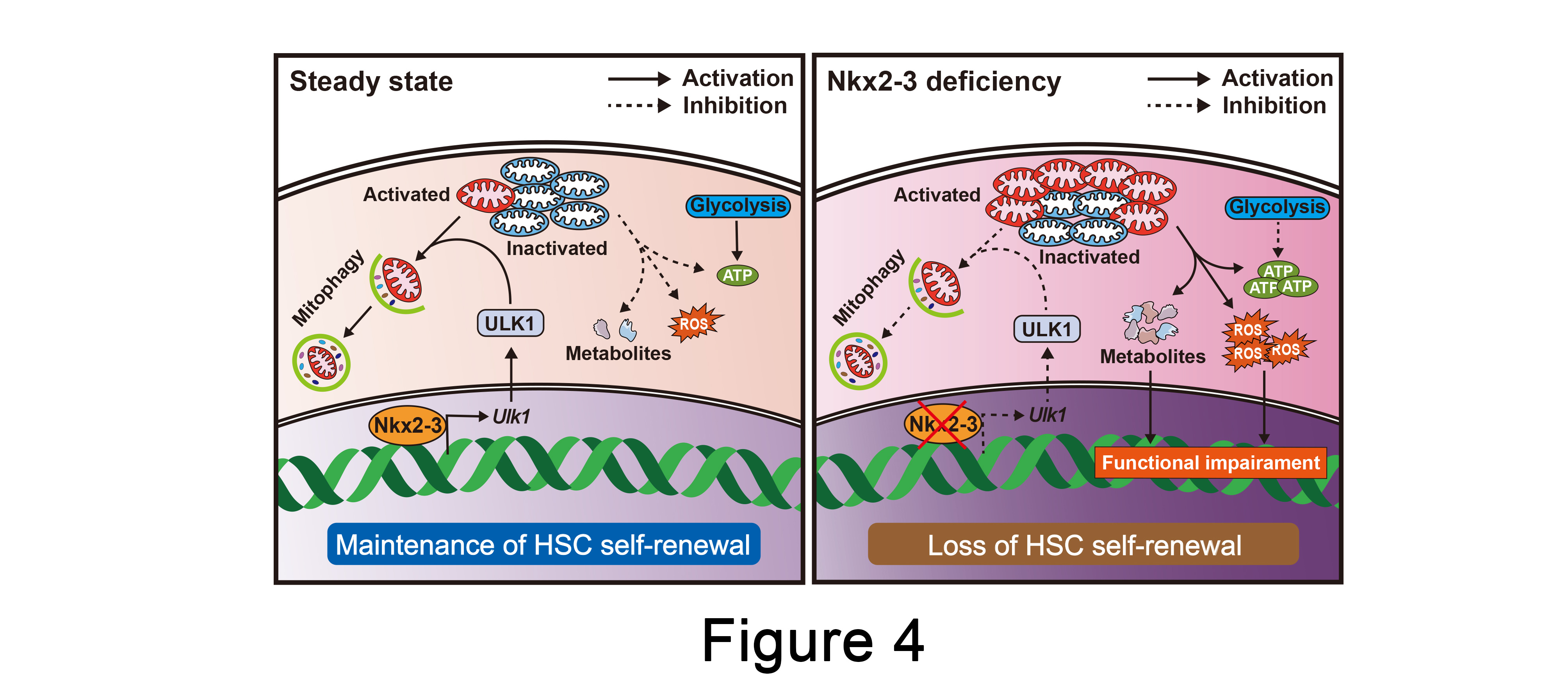
Figure 4. The Nkx2-3/ULK1/mitophagy axis maintains HSC self-renewal.
References
- Nakamura-Ishizu A, Ito K, Suda T. Hematopoietic Stem Cell Metabolism during Development and Aging. Dev Cell. 2020;54:239-55.
- Suda T, Takubo K, Semenza GL. Metabolic regulation of hematopoietic stem cells in the hypoxic niche. Cell Stem Cell. 2011;9:298-310.
- Hu M, Wang J. Mitochondrial metabolism and the maintenance of hematopoietic stem cell quiescence. Curr Opin Hematol. 2019;26:228-34.
- Hu M, Zeng H, Chen S, Xu Y, Wang S, Tang Y, et al. SRC-3 is involved in maintaining hematopoietic stem cell quiescence by regulation of mitochondrial metabolism in mice. Blood. 2018;132:911-23.
- Qian P, He XC, Paulson A, Li Z, Tao F, Perry JM, et al. The Dlk1-Gtl2 Locus Preserves LT-HSC Function by Inhibiting the PI3K-mTOR Pathway to Restrict Mitochondrial Metabolism. Cell Stem Cell. 2016;18:214-28.
- Luchsinger LL, de Almeida MJ, Corrigan DJ, Mumau M, Snoeck HW. Mitofusin 2 maintains haematopoietic stem cells with extensive lymphoid potential. Nature. 2016;529:528-31.
- Jin G, Xu C, Zhang X, Long J, Rezaeian AH, Liu C, et al. Atad3a suppresses Pink1-dependent mitophagy to maintain homeostasis of hematopoietic progenitor cells. Nat Immunol. 2018;19:29-40.
Follow the Topic
-
Leukemia
This journal publishes high quality, peer reviewed research that covers all aspects of the research and treatment of leukemia and allied diseases. Topics of interest include oncogenes, growth factors, stem cells, leukemia genomics, cell cycle, signal transduction and molecular targets for therapy.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in