Trapping seed crystallites to peer inside a black box
Published in Chemistry
The paper in Nature Communications is here: http://go.nature.com/2IFSLsP
A black box. When I have one in
front of me, I am always curious to know what is inside. This probably comes
from my facetious parents. Indeed, when we were given a gift, my siblings and I
could never rely on the packaging.
Self-seeding is like one of these mysterious boxes of my childhood. If a solution containing a crystallizable polymer is heated above the dissolution temperature of the polymer, and cooled to a given crystallization temperature, then polymer crystals broadly dispersed in size are obtained. Intriguingly, if this solution is reheated at a self-seeding temperature, Ts, at which all the crystals seem to have disappeared, and cooled to the same crystallization temperature, crystals of identical size and morphology form.
This discovery is not recent. It was first reported in 1966 by Blundell, Keller, and Kovacs who studied the crystallization of polyethylene in xylene.1 Several questions are, however, still lingering: what happens during the annealing at the self-seeding temperature? How do the seeds dissolve?
There are techniques that could allow one to look inside the box and study the dissolution of these crystals at the self-seeding temperature, but these techniques are often limited by sample concentration. As soon as a large fraction of the polymer crystals dissolves, the technique loses its sensitivity, and the box recloses.
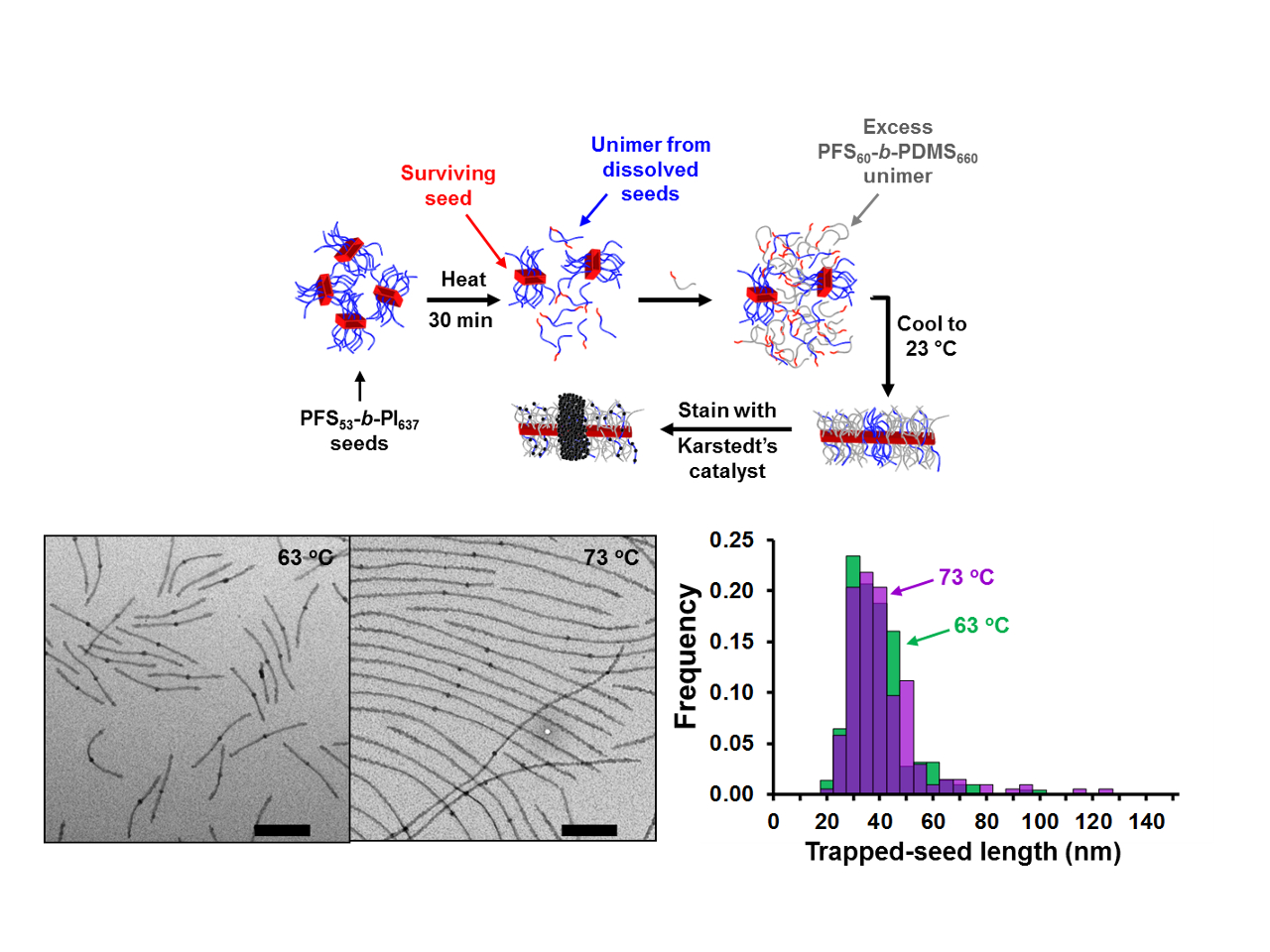
We thus used a different approach. We trapped the crystallites that survived dissolution at Ts, with a second crystalline polymer, which allowed us to distinguish these surviving crystallites from the crystals that regrew from them at the crystallization temperature.
We have studied the crystallization-driven self-assembly of coil-crystalline block copolymers for many years. In our hands, the crystalline block has mainly been polyferrocenyldimethylsilane, that we affectionately call PFS (although PFDMS is a more accurate acronym). For the present work, we used PFS-b-PI (PI = polyisoprene) and PFS-b-PDMS (PDMS = polydimethylsiloxane). When heated in decane at 100 °C and cooled to room temperature, PFS-b-PI and PFS-b-PDMS form 1 dimensional (1D) structures, with a crystalline PFS core. These 1D micelles possess very interesting properties: sonication breaks them into short fragments (crystallites) that are stable for months in solution, and these crystallites can easily be extended by adding free chains (unimer) of PFS block copolymer, to form 1D micelles of uniform and predictable length.
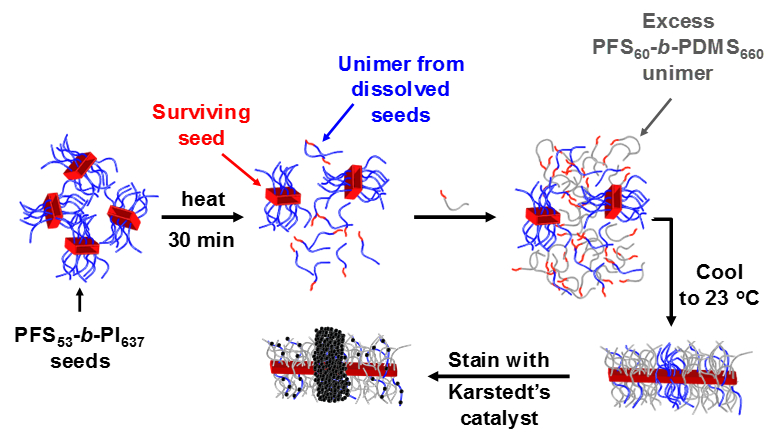
For our experiments, dilute solutions of PFS-b-PI crystallites were annealed at different self-seeding temperatures. Before cooling, we injected a large excess of PFS-b-PDMS unimer to trap the surviving crystallites. During recrystallization, the large excess of PFS-b-PDMS unimer (and PFS-b-PI unimer coming from the dissolved crystallites) add to the ends of the surviving crystallites to form M(PFS-PI)-r-M(PFS-PDMS)-b-M(PFS-PI)-b-M(PFS-PDMS)-r-M(PFS-PI) triblock co-micelles, with the trapped seeds located at the center. After selective staining of the PI block, the surviving crystallites could easily be observed by transmission electron microscopy (TEM), shining light into the self-seeding black box, even when very few crystallites survived.

The results are extremely interesting
and surprising, but I prefer to let you discover them in our paper, here : http://go.nature.com/2IFSLsP
Take it as a gift!
- Blundell, D. J., Keller, A. & Kovacs, A. J. A new self‐nucleation phenomenon and its application to the growing of polymer crystals from solution. J. Polym. Sci. B 4, 481–486 (1966).





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
VERY GOOD JOB !
Thank you. I appreciate your comment.