Unexpected beneficial effects of forest edges on pollinator networks
Published in Ecology & Evolution

The worldwide decline in insect pollinators is a topical and concerning issue of global importance. The accepted narrative is that ecosystem decay due to habitat fragmentation and edge effects can exacerbate insect pollinator decline due to habitat loss. However, in forested ecosystems, open light-filled forest edges support more flowers and more pollinators than the dark interior within closed-canopy forests, such that forest edges might actually be beneficial for pollinators. The mismatch in observations is almost certainly because most studies on the effects of habitat fragmentation on plant-pollinator communities have focused on open-habitat systems such as grasslands, croplands or savannah woodlands where edge effects are likely negative or neutral. In our research, we found that forest edges indeed have unexpected beneficial effects on pollinator network, buffering plant-pollinator interaction networks from cascading secondary extinctions in the face of declining forest area.
The research was carried out in the Thousand Islands Lake study system in eastern China, which has been the subject of a large number of habitat fragmentation studies in recent years. From 2017, I commenced sampling plant-pollinator interactions along edge and interior gradients of regenerating secondary forests on 41 islands and 16 mainland sites in a fragmented land-bridge island system (Fig. 1). Over 3 years, our team recorded nearly 20,000 individual pollinator interactions with plants in almost 1000 hours of flower observations (Fig. 2), building networks of associations between 68 species of flowering plants and 313 species of pollinators (Fig. 3).

Fig. 1. Study sites in the Thousand-Island Lake. Photo credit: Jincao Pan.

Fig. 2. Field sampling at the Thousand Island Lake. (a) On the way to the sampling sites. (b) Arriving at one of the sampling sites. (c) Returning to the field station. Photo credits: (a-b) Peng Ren and (c) Juan Liu.

Fig. 3. The common flower-visiting insects of the Thousand Island Lake, China. Photo credit: Peng Ren.
After the sampling was completed in 2019, I was very fortunate to obtain an overseas research placement with Prof. Raphael Didham at The University of Western Australia. This gave me an opportunity to apply for funding under the PhD joint training program of the China Scholarship Council (CSC), and the CSC awarded me a grant under the support of my advisor Prof. Ping Ding and Zhejiang University. Then I visited Prof. Raphael’s laboratory for one year (see Fig. 4), which was my first time studying abroad for such long period of time, encountering many exciting and novel challenges. During the study in Prof. Raphael's laboratory, we had at least one discussion each week to solve the specific problems I encountered in my analysis and writing. When unlocking R's new skills, I felt that only through tackling a very large and difficult problem could I gain a deeper understanding of R. I also learned lots of new skills in writing using English during the process of revising the drafts with Prof. Raphael. I realized that it is not easy to clearly describe a scientific fact or result. Deep understanding is the premise of being able to narrate clearly, then revise repeatedly (and further revise again and again!) to achieve a really clear explanation of the findings.

Fig. 4. Visiting Prof. Raphael’s laboratory at The University of Western Australia. Photo credits: (a) Shu Li and (b) Hui Zhang.
From the outset it was immediately evident that there was something very special in the results, with much higher abundance and richness of pollinators at forest edges in TIL, rather than the negative edge effects found in many other pollinator studies. Applying a structural equation modelling approach, we showed the positive impact of forest edges extended not only to biodiversity metrics, but also to plant-pollinator network structure as well. Surprisingly, despite many excellent studies applying structural equation modelling in ecology, we found there were still gaps in the available methods and the rigour of application across studies. In particular, methods to deal with incomplete sampling and stochastic sampling biases have been poorly developed in SEM studies, making it hard to judge whether the significance of causal paths is indeed an objective and credible result, or an artefact of stochastic sampling biases. After brainstorming potential standardization approaches and sensitivity tests, my discussions with Prof. Raphael gave the inspiration for explicit integration of null model resampling approaches into the SEM, which turned out to be a simple and effective approach to the problem. One important (and in hindsight quite intuitive) finding was that some causal paths with very large effect sizes in the SEM (e.g., paths 32 and 43 in Fig. 5) are not actually that ecologically important, in terms of what they tell us about fragmentation effects, because they are just what we would expect from a null ‘random’ draw from the underlying reference state or regional pool. As a result, we can clearly know which paths in the SEM are indeed causally influenced by fragmentation, over and above stochastic sampling effects and null expected relationships among variables that occur even in the absence of fragmentation.
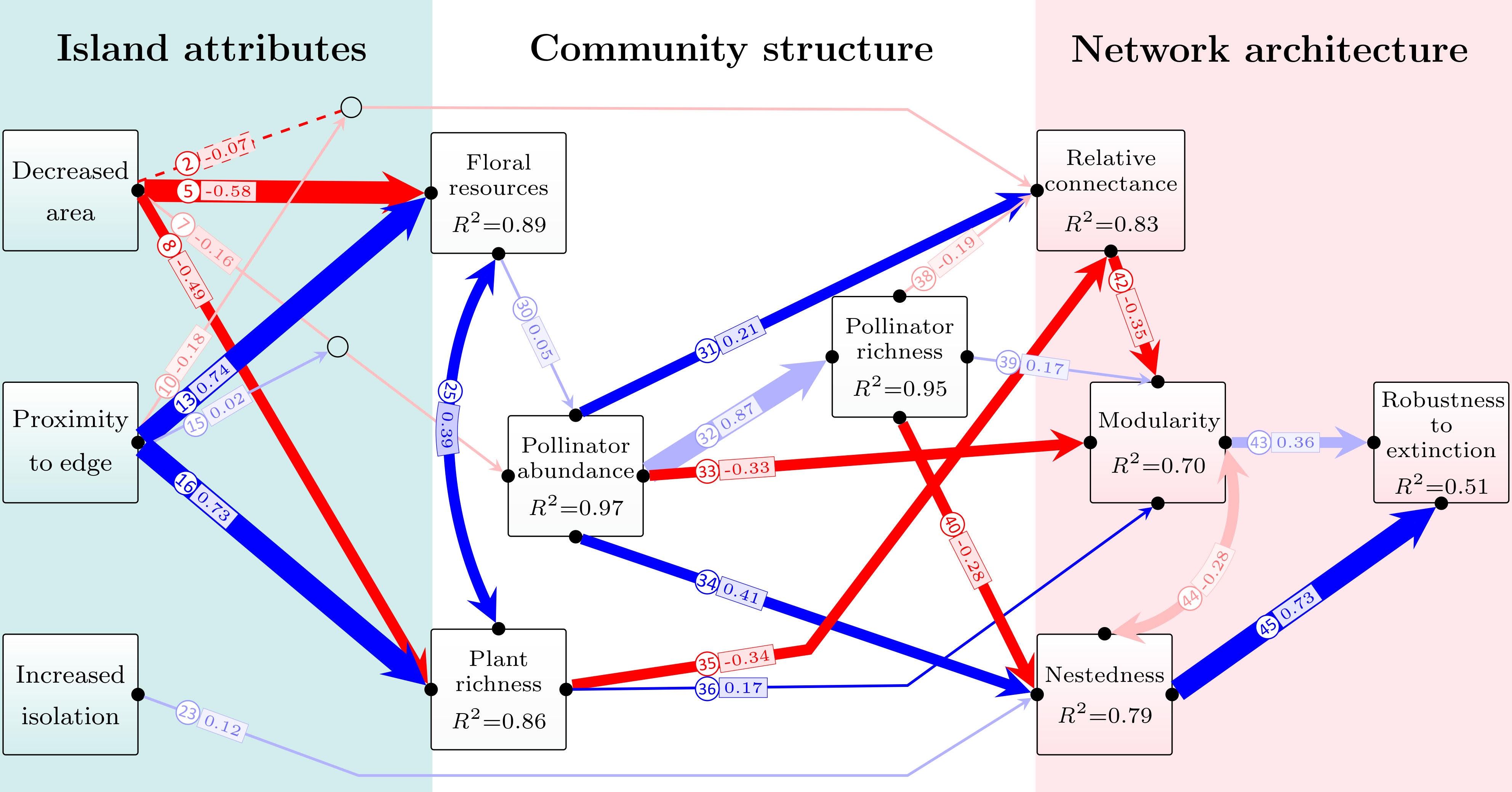
Using these approaches, we found that plant richness and floral resources decline with the decrease of forest area at both edge and interior sites, but edges maintain 10-fold higher pollinator abundance and richness regardless of area loss. Surprisingly, these edge networks contained many highly specialized species, even compared to interior networks. Most importantly, the edges of secondary forests provide an anthropogenic analogue of natural gap-phase conditions, and we showed that edges increased network robustness to extinction and reduced the risk of plant-pollinator system collapse, thus having a positive, rather than negative, edge effect on pollinator communities in forest systems.
From a conservation perspective, in situations where there is a strong move toward reforestation (for restoration or carbon offsetting, etc), we should pay more attention to pollinator loss because dense closed secondary forests do not have the right abiotic conditions and floral resources for many pollinators. In this context, anthropogenic edges can actually buffer biodiversity loss, during the time it takes for gap-phase dynamics to re-establish in re-grown forests. Undoubtedly, our findings are very important for the large reforestation schemes in China and other parts of the world that only use one or a few tree species, and might plant large areas with dense even-aged forests. These areas could benefit greatly from considering heterogeneity of edge and gap-clearing conditions as part of the reforestation scheme.
It is important to recognize we are not advocating that forest fragmentation is ‘good’ for biodiversity, in a general sense, but in the absence of effective restoration strategies that promote heterogeneity of forest gap structure and diversity of floral resources for pollinators, anthropogenic edges can have an unexpected beneficial role to play in network resilience, especially in the early decades of forest restoration.
Read the paper at https://www.nature.com/articles/s41559-022-01973-y
Follow the Topic
-
Nature Ecology & Evolution
This journal is interested in the full spectrum of ecological and evolutionary biology, encompassing approaches at the molecular, organismal, population, community and ecosystem levels, as well as relevant parts of the social sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Biodiversity and ecosystem functioning of global peatlands
Publishing Model: Hybrid
Deadline: Jul 27, 2026
Understanding species redistributions under global climate change
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in