Urine-derived bladder cancer organoids (urinoids) as a tool for cancer longitudinal response monitoring and therapy adaptation – behind the paper
Published in Cancer, Cell & Molecular Biology, and Genetics & Genomics

Background on bladder cancer, available research models and non-invasive model need.
Bladder cancer ranks among the top five and ten most common cancers worldwide in men and women, respectively. Urothelial carcinoma (UCC) is the predominant histopathological subtype of bladder cancer (BC) and on initial staging presents as non-muscle invasive bladder cancer (NMIBC; 73% of total) or muscle-invasive bladder cancer (MIBC). Unfortunately, bladder cancer is a complex disease, due to its highly heterogeneous nature and high mutational rate, for which ‘tailor-made’ therapies are not available. . Adequate in vitro research models that show the same characteristics and tumour-heterogeneity as the tumour in the patients during disease development and during patient treatment are missing. Therefore, there is an unmet need for research models that allow researchers and clinicians to (I) monitor bladder cancer in the lab during patient treatment, (II) test drug responses and (III) develop adaptive treatment strategies based on measured sensitivities.
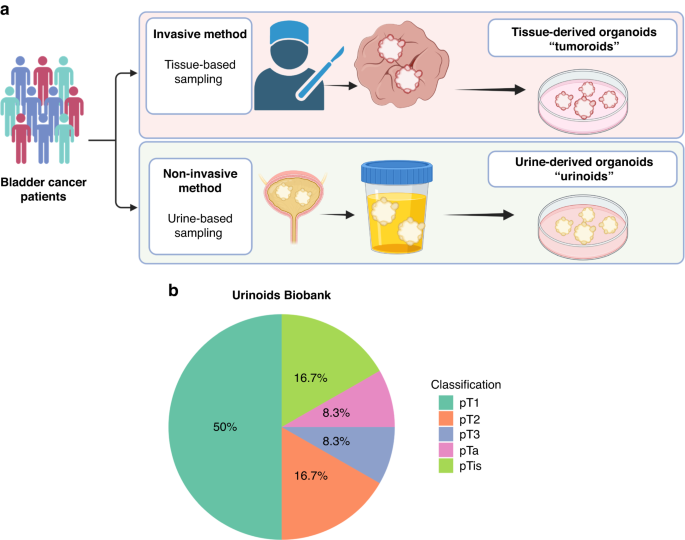
To model the heterogeneity of bladder cancer, recent advanced models were developed that recapitulate these tumour-characteristics using three-dimensional tumour structures called organoids. The limitation of these tissue-based organoids is that they are derived from surgical biopsies, harvested by an invasive surgical transurethral procedure (tumor resection). Therefore, it is often not possible or desirable to sample patients before, during and after their systemic treatment. Novel methods are currently being held back for clinical application due to the lack of availability of tumor material at the start and during patient treatment. Generally, in bladder cancer patients exfoliated viable urothelial cancer cells can be found in voided urine or a bladder-washout. This is especially the case for large and high-grade tumors. In fact, urinary cytology is one of the corner stones in diagnostic tests for bladder cancer.
This manuscripts hypothesis, conclusion and future prospective
We propose a novel non-invasive platform for tumor pathogenesis and drug-testing: urine-based bladder tumoroids (urinoids). In this manuscript, we demonstrate that the urinoids, accurately represent the original tumour of bladder cancer patients. Furthermore, we show that these urinoids can be used to follow patients during their systemic treatment. Following patients with urinoids allows us to study the patient’s tumour both with genetics and therapy-responses from the start until the end of their treatment schedule. We show that over 84 days, patient 4 could be closely monitored using the urinoids method. Over the course of 3 rounds of immunotherapy, this patient developed a de novo genomic rearrangement, found back in both the tissue-based organoids and in the urinoids. Furthermore, cellular machinery involved in cell divisions, part of the microtubule network, was also found to be significantly mutated after immunotherapy in patient 4. These mutations were found in both the tissue- and urine-derived organoids post-immunotherapy. By targeting this microtubule network with chemotherapy, we show that we can specifically target these tumour cells by exploiting these newly developed mutations. Taken together, we show that urinoids allow for non-invasive sampling of bladder cancer patients. These urinoids can be used for the monitoring of patients in the lab before, during and after their treatment. This non-invasive technique with urinoids opens a whole field of patient and time specific targeted interventions. As shown in this manuscript, these targeted interventions can be based on genetic alterations, changes in drug-response and acquired treatment resistance over time in a patient specific manner. This will allow researchers and clinicians to not only provide more targeted care, but also prevent unnecessary patient therapy toxicity due to ineffective treatments.
Ultimately, we hope that these newly developed urinoids will finally allow bladder cancer patients to be treated on a first-time-right approach, directly providing feedback when therapies become less effective and determine the following treatment course (Figure 1).
Follow the Topic
-
British Journal of Cancer
This journal is devoted to publishing cutting edge discovery, translational and clinical cancer research across the broad spectrum of oncology.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in