Vascular aging-targeted intervention based on NAD+ elevation as a novel measure to treat hypertension
Published in Healthcare & Nursing and General & Internal Medicine
Hypertension represents a major public health issue that is responsible for 8.5 million deaths due to stroke, ischemic heart disease and renal disease worldwide.1 Endothelial dysfunction and arterial stiffness are considered hallmarks of hypertension and play an essential role in the pathogenesis of atherosclerotic cardiovascular diseases.2-4 Therefore, further understanding of the specific molecular mechanisms of hypertension and its associated vascular damage is of great clinical significance in seeking an innovative therapeutic strategy for the management of hypertension.
Nicotinamide adenine dinucleotide (NAD+) is a central cofactor involved in cellular biological activities that declines during aging.5-8 Restoring NAD+ levels with dietary supplement and regulating NAD+ metabolic enzymes with small molecular compounds have emerged as a potential therapeutic measure to elevate NAD+concentrations, thereby providing opportunities for ameliorating aging.13-16 Hypertension is acknowledged as a kind of aging-related disease and NAD+ supplement may be a promising clinical strategy as an aging-targeted intervention for hypertension. However, changes in NAD+ levels and their relationship with blood pressure (BP) elevation and vascular damage have not yet been explored in patients with hypertension. Therefore, in the present study, we found the phenomenon of NAD+ exhaustion in endothelial cells in hypertension, explored the mechanism of NAD+ exhaustion mediated by CD38 activation and put forward that NAD+ boosting therapy may be a promising therapeutic measure to treat patients with hypertension.
Our study found a 44% decrease in NAD+ levels in peripheral blood mononuclear cells of hypertensive patients, which is associated with increased blood pressure and impaired vascular function. Further examination of aortic tissue in hypertensive patients confirmed decreased NAD+ levels. At the same time, we constructed a hypertensive mice model and found that the levels of NAD+ levels in the aortic tissue and endothelial cells of hypertensive mice decreased, which was consistent with the clinical results. Moreover, NAD+ supplement contributed to reducing blood pressure, improving vascular function, and alleviating vascular remodeling in hypertensive mice. To further validate the beneficial effects of NAD+, a clinical trial was carried out in hypertensive patients receiving NAD+ precursors β-nicotinamide mononucleotide (NMN) for 6 weeks. Results showed a 43% increase in NAD+ levels in hypertensive patients, accompanied by a decrease in arterial blood pressure, improvement in endothelial function, and decline in arterial stiffness.
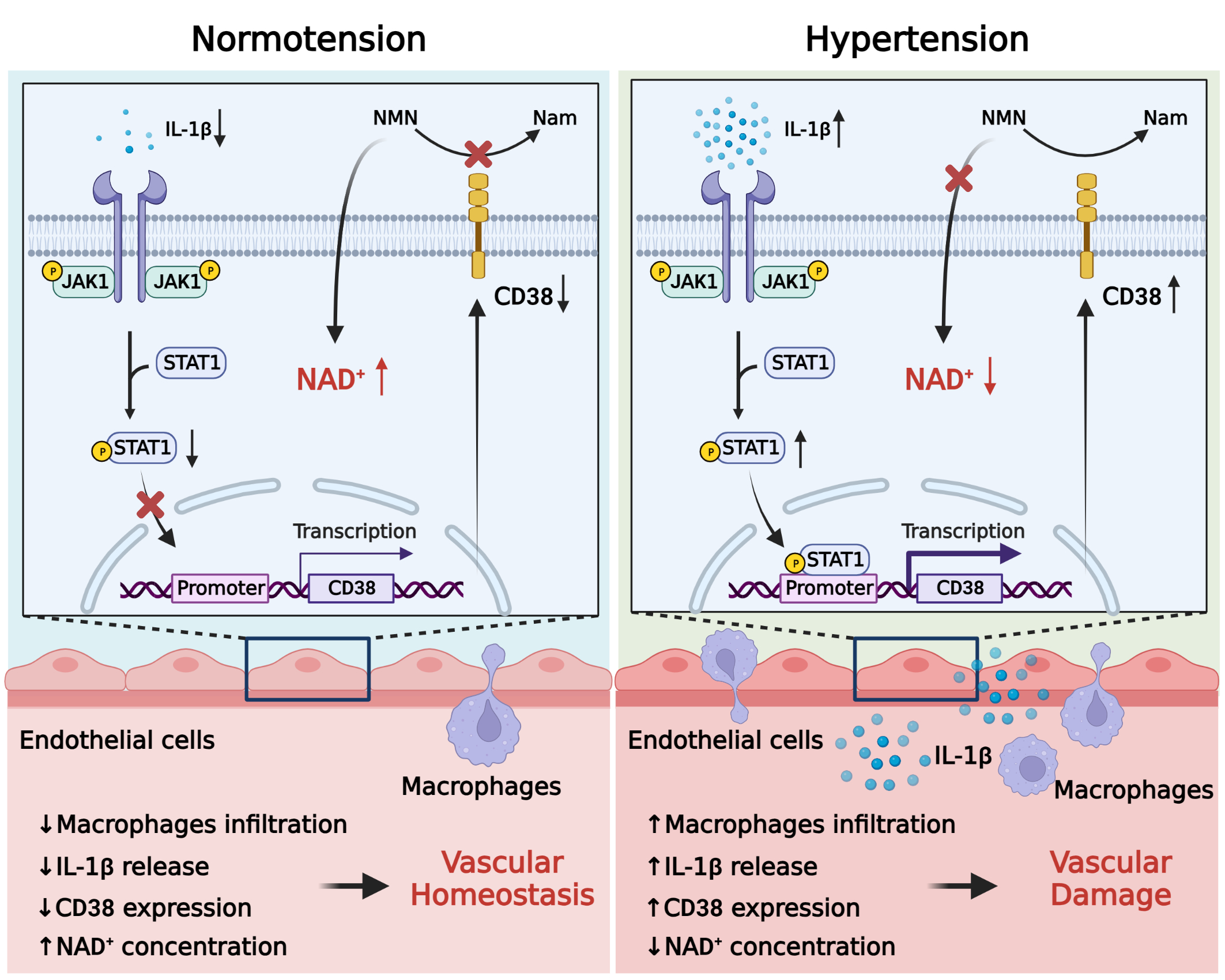
To determine the reasons for the decrease in NAD+ levels in hypertensive endothelial cells, we screened critical enzymes that were involved in the biosynthesis or metabolism of NAD+. CD38, as a key rate-limiting enzyme for NAD+ degradation, was significantly elevated in hypertension. To confirm the relationship between CD38 upregulation-mediated NAD+ depletion and elevated blood pressure and vascular injury, we first found endothelial cells that overexpression of CD38 leads to a decrease in NAD+ levels in vitro. Then, an in vivohypertensive CD38-knockout mouse model was established, and the results showed that the blood pressure of CD38-knockout mice decreased. Furthermore, the mouse bone marrow transplantation model and the adeno-associated virus endothelial CD38-specific knockdown mouse model were used to confirm that high expression of CD38 in endothelial cells accelerated NAD+ depletion, leading to increased blood pressure and vascular homeostasis imbalance.
Furthermore, we investigated how CD38 catalyzes NAD+ degradation. there are 3 forms of CD38 (type II transmembrane, type III transmembrane and soluble forms), of which the type II membrane orientation with an ecto-NAD+ glycohydrolase site outside the cells is the major one. We have found that the ecto-enzymic activity of CD38 can consume extracellular NMN, thereby affecting NMN entry into cells to synthesize NAD+. Both the CD38 inhibitor 78c, which targets all activity, and the CD38 inhibitor isatuximab, which targets ectoenzymatic activity, reversed the decrease in NAD+ levels, indicating that the increased ecto-enzymic activity of CD38 was a key molecule leading to a decrease in NAD+ levels in hypertension.
It is well-known that hypertension is a chronic inflammatory disease, and a large amount of evidence suggested that the infiltration of macrophages in vessels and the release of inflammatory cytokines promoted the development of hypertension. Therefore, we investigated whether the interaction between macrophages and endothelial cells promotes the expression of CD38. The results confirmed that macrophage infiltration increased in hypertensive vessels as well as IL-1β secretion. The IL-1β activated the JAK1/STAT1 signaling pathway in endothelial cells, leading to an increase in CD38 expression. Conversely, inhibition of JAK1/STAT1 signaling can partly reverse the effect. The results indicated that macrophage secretion of IL-1β activated the JAK1/STAT1 signaling pathway in endothelial cells, which contributed to CD38 overexpression.
In summary, the present study demonstrates that NAD+ augmentation lowered BP and improved vascular dysfunction in the context of hypertension, and provides robust evidence to support vascular aging-targeted intervention as an alternative and novel means to control BP and improve vascular function in hypertension. Of particular importance, we make the effort to explore the potential possibility of NAD+treatment from the perspective of aging to clinical application for the management of hypertension. Overall, the present study may shed new light on novel strategies in which NAD+ boosting therapy, including NMN supplement and CD38 inhibition, may turn out to be a promising therapeutic measure to treat patients with hypertension.
Reference
1 Mills, K. T., Stefanescu, A. & He, J. The global epidemiology of hypertension. Nat. Rev. Nephrol. 16, 223-237 (2020).
2 Boutouyrie, P., Chowienczyk, P., Humphrey, J. D. & Mitchell, G. F. Arterial Stiffness and Cardiovascular Risk in Hypertension. Circ. Res. 128, 864-886 (2021).
3 Cohen, J. B. et al. Arterial Stiffness and Diabetes Risk in Framingham Heart Study and UK Biobank. Circ. Res. 131, 545-554 (2022).
4 Zhou, Z. et al. Hypertension, Arterial Stiffness, and Clinical Outcomes: A Cohort Study of Chinese Community-Based Population. Hypertension 78, 333-341 (2021).
5 Covarrubias, A. J., Perrone, R., Grozio, A. & Verdin, E. NAD(+) metabolism and its roles in cellular processes during ageing. Nat. Rev. Mol. Cell Biol. 22, 119-141 (2021).
6 Chini, C. C. S. et al. CD38 ecto-enzyme in immune cells is induced during aging and regulates NAD(+) and NMN levels. Nat. Metab. 2, 1284-1304 (2020).
7 Covarrubias, A. J. et al. Senescent cells promote tissue NAD(+) decline during ageing via the activation of CD38(+) macrophages. Nat. Metab. 2, 1265-1283 (2020).
8 Navas, L. E. & Carnero, A. NAD(+) metabolism, stemness, the immune response, and cancer. Signal Transduct. Target Ther. 6, 2 (2021).
Follow the Topic
-
Signal Transduction and Targeted Therapy
This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in