Our publication in Nature Communications, which describes a new approach for engineering perfused tissues, is the result of many years of engineering work before we could finally show how it performed in biological applications. Here is the story of this long road to perfused tissues. When I engaged with organoids in my research, like many others, I quickly realized basic limitations of this model: low fidelity of cellular phenotypes, immaturity of the tissue and randomness of its structure. The issues were particularly prominent in brain organoids as they are usually grown to a large size during prolonged periods, with the goal of obtaining complex structural and cellular traits found in the in-vivo brains. However, lacking a vasculature system, organoids can only rely on diffusion to deliver oxygen and nutrients inside the organoid, which is effectively limited by 200-250µm distance from the surface. The necrotic processes developing within the organoid are thought to be one of the major factors impeding normal developmental processes in the tissue and result in the immature cellular identities and the random architecture commonly seen in such organoids.
As one would expect, there have been numerous efforts to provide physiologically relevant nutrient, oxygen and waste exchange to engineered tissues. However, none of the developed approaches thus far yielded reproducible and well-defined micro-perfusion. For this reason, tissues that require extensive blood flow (brain, liver etc.) have been limited to about 500µm in at least one of the tissue dimensions.
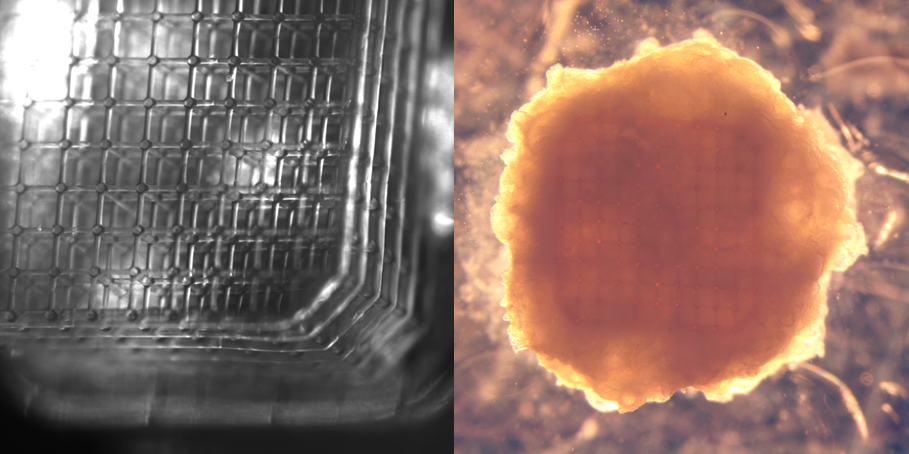
Thinking about how to solve “the vascularization problem” in a simple engineering way, I reasoned that tiny capillaries made of a permeable material would be a simplest approach: oxygen and nutrients from the fresh medium, pumped through the capillaries, and waste products from the tissue could freely exchange across the capillary walls. Diving into the literature, I discovered that gelatin, a natural hydrogel, could be cross-linked by the light in the presence of Rose Bengal, a commonly used stain in ophthalmology. At that time, 2-photon microscopy was an integral part of my research projects and I was very impressed with high, sub-micron resolution of the images generated by 2-photon imaging. Making a logical step from 2-photon imaging to 2-photon lithography was easy and after some trials end error, I obtained the first gelatin capillaries. Their handling was difficult and they crumbled after washing, but this reassured us that the strategy was viable.

Figure 1. 3D mesh of gelatin capillaries. Capillaries collapsed after washing out non-cross-linked gelatin, supporting them.
As a next step, we developed a denser photopolymer which still allowed for diffusion but, unlike gelatin, did not swell in culture medium. This was a critical advance, as this enabled the direct fabrication of the capillary structure on a hard plastic disk with perfusion holes. The capillaries attached to the disk could then be plugged into a custom designed perfusion chip and connected to a medium circulation system.
As a biological proof of concept, we designed “perfusion baskets” for tissues, arranging vertical capillaries in a regularly spaced array with bordering “fences”, which resembled a grid structure. To generate our tissues, we seeded these grids with multiple human pluripotent stem cell spheroids and cultured them with medium, constantly perfusing through the capillaries. These experiments also included grids that were bathed in medium but not connected to a perfusion, as well as organoids cultured conventionally as controls. After 8 days of growth and neuronal differentiation the spheroids fused and formed tissues reaching approx. 3.5x3.5x2.5mm in size. We then stopped the experiments and analyzed the tissues.
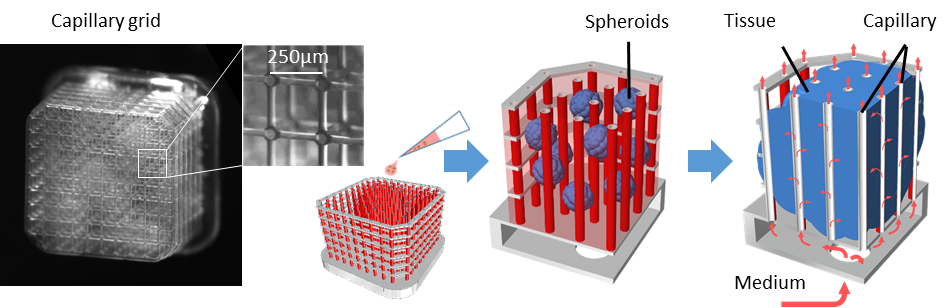
Figure 2 Capillary grid design and tissue generation strategy. From left to right: photograph of the empty “perfusion basket” (capillary grid); a CAD model of the grid with capillaries shown in red; the grid seeded with spheroids (blue) suspended in Matrigel (pink); after several days of perfusion a cohesive tissue is formed, flow direction shown with red arrows
The difference between perfused and non-perfused samples was striking. Immunohistochemistry and scRNAseq data revealed two major differences. First, while conventionally grown organoids and non-perfused constructs predictably suffered from hypoxia and apoptosis, the perfused constructs were perfectly viable and remained proliferative. Second, differentiation in our perfused constructs was significantly accelerated. In just two days of neuronal induction a large proportion of cells in perfused constructs acquired early neuronal identity, while conventional organoids and non-perfused constructs remained entirely pluripotent.
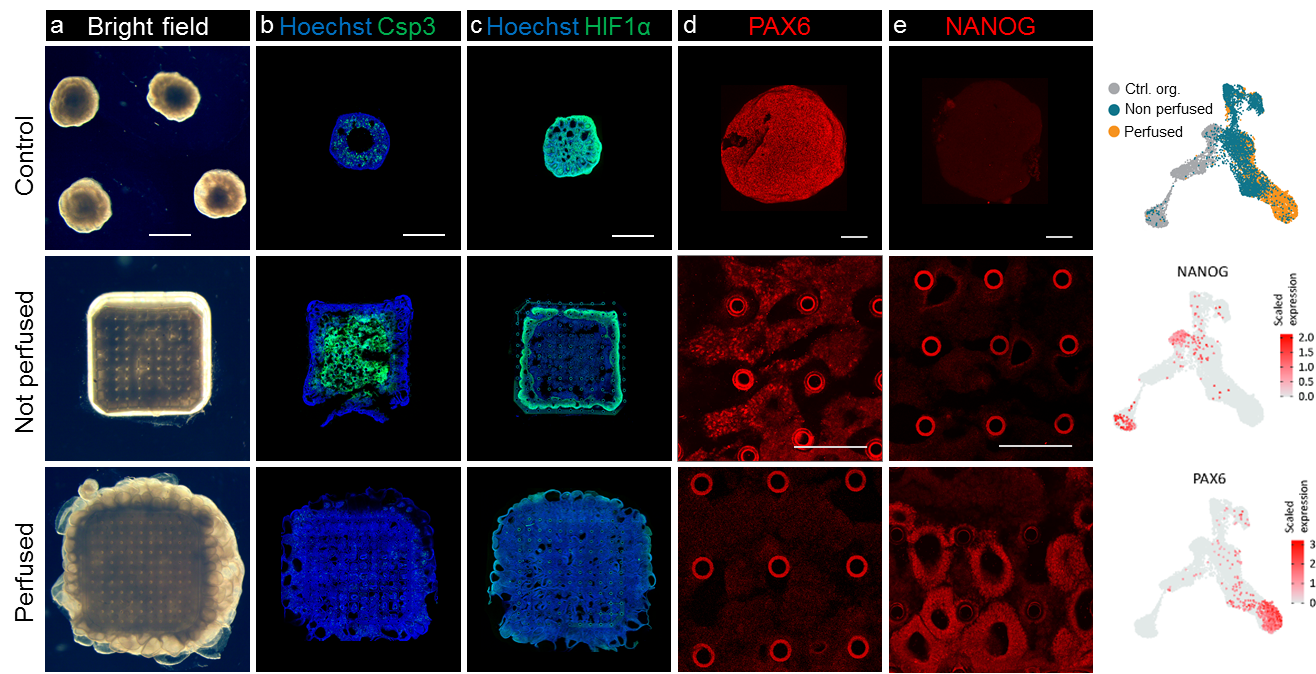
Figure 3 Perfusion prevents apoptosis and accelerates differentiation in neural constructs. a Bright field images of organoids, non-perfused and perfused constructs. b Immunofluorescent images of apoptotic marker cleaved Caspase 3 (green). c Hypoxia marker HIF1α (green). d Early neural marker PAX6. e Stem cell marker NANOG. f UMAP plots of the combined dataset highlighting locations of PAX6 and NANOG expressing cells in the UMAP space. a-c Scale bar 1mm. d, e Scale bar 250µm
Inspired by the outcome of the short-term experiments, we decided to try longer cultivation periods. We thought a liver tissue would be an optimal model to showcase the versatility and practical value of our vascularization platform. We differentiated liver tissues for 40 days and revealed much greater expression levels of key liver metabolic proteins, including CYP450 enzymes. In addition to expressing key liver markers, our 50 day differentiated tissues were metabolically active, producing albumin and urea, and our drug metabolism tests revealed that activity of CYP450 enzymes was similar to their activity in humans. More importantly, the human clearance of theophylline, a slowly metabolized drug, was very well predicted by the perfused liver constructs, which, I believe, opens up new perspectives in developing new highly predictive ADME assays.
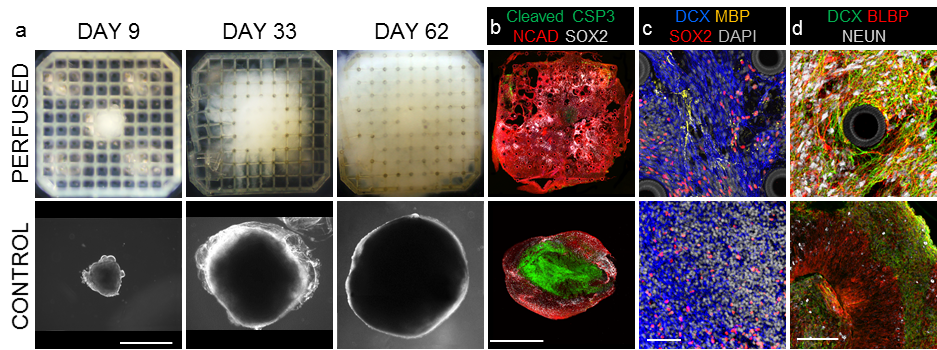
Figure 4 Perfusion of cortical organoids. a Bright field images of perfused organoids growing in grids (top row) and control organoids in floating culture. b Immunofluorescent images demonstrating viability and enhanced phenotypical features of perfused organoids. Cleaved Caspase 3 (green) indicates apoptotic cores in control organoids, while healthy tissue shows neuronal identity (N-Cadherin, red, SOX2, white). c Perfused organoids exclusively express myelin basic protein (MBP, yellow), marker of mature oligodendrocytes, migratory cortical neuron marker Doublecortin (DCX, blue). c Staining for radial glia, BLBP (red) and DCX (green), demonstrate a neuropil-like structure in perfused organoids formed by dense and intertwined networks of radial glia and neuronal processes, including a populations of mature neurons (NeuN, white), while in control organoids the cellular arborization is almost absent. a, b Scale bar 1mm. c, d Scale bar 70µm
Finally, we showed that single cortical organoids could be cultured in our platform over a long period. We seeded individual neural spheroids one per grid and compared them to the similar spheroids in suspension culture after two months of neural differentiation. Perfused organoids grew larger than control organoids with no signs of massive apoptosis. The perfused organoids abundantly expressed adult neuronal marker (NEUN) and were more developed showing extensive neuronal and radial glia arborization and signs of myelinization, which were lacking in control organoids.
In this work we implemented a complete 3D culture platform which provides perfusion for engineered tissues, thereby overcoming one of the major challenges in tissue engineering. While the current implementation of the platform does not recapitulate such properties of vascular networks as selective transport or vascular remodeling, this approach could be combined with endothelial vascularization, providing tissue support during the period, while the endothelial vasculature is establishing a perfusable capillary network. We believe this platform provides a technological foundation for the development of novel in vitro models of increased complexity and scale.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in