A bioinspired sequential energy transfer system constructed via supramolecular copolymerization
Published in Chemistry
To slow down global warming, the world community has put forward the road to achieve carbon neutralization, which lay great emphasis on the use of renewable energy. Particularly, since our planet receives almost infinite energy from the sun every year, light energy has attracted considerable attention. For photosynthetic organisms, they can harvest light energy efficiently via light harvesting systems (LHSs). Hence, it would be meaningful if natural LHSs could be simulated via artificial systems. Frankly speaking, this is the primitive motivation to carried out this study. In the following part, I want to share my experience and thinking about the current research.
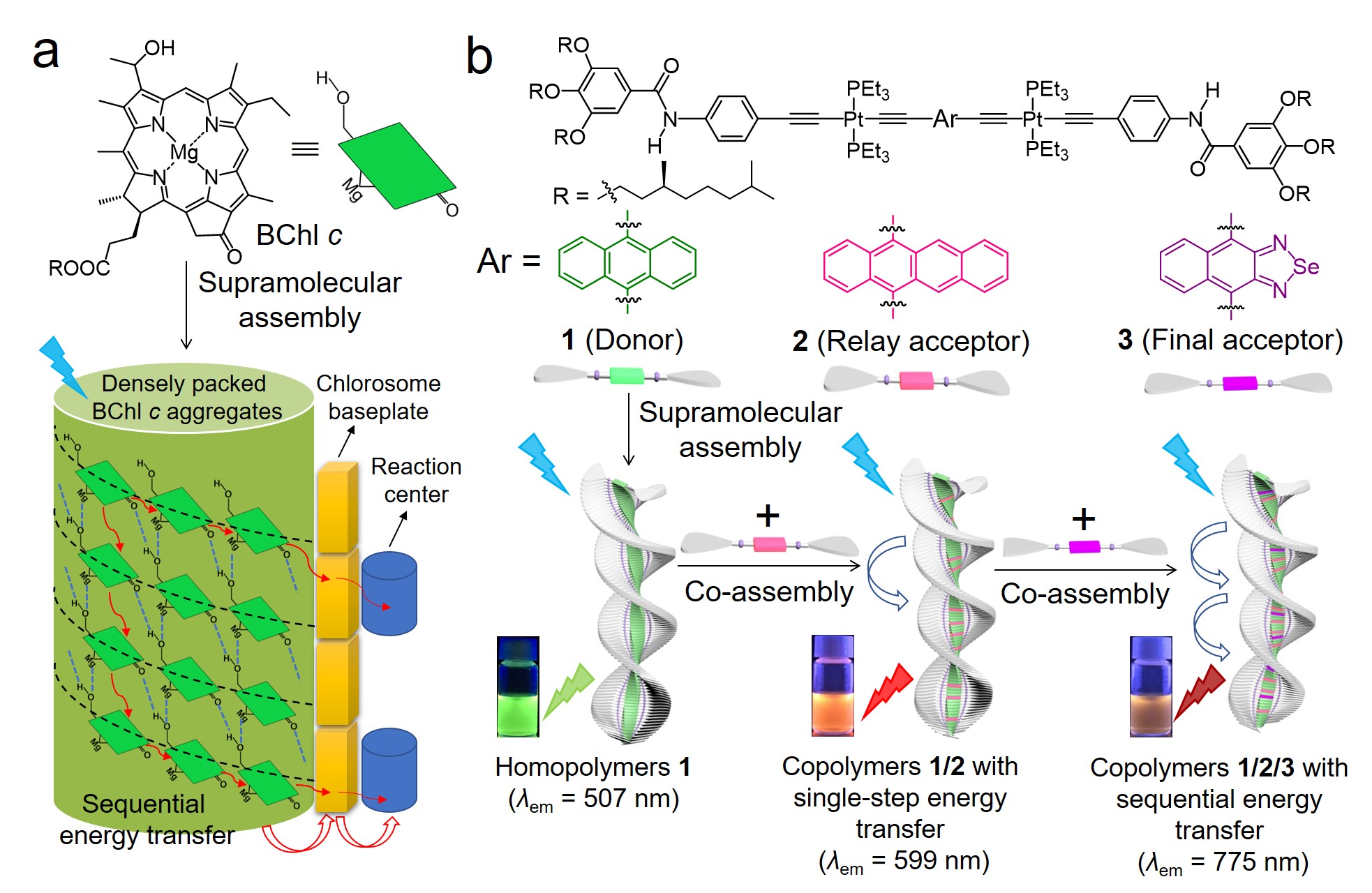
To simulate natural LHS, the first issue was to choose a suitable natural prototype for the newly designed artificial LHS. As classical prototypes, the LHSs of purple photosynthetic bacteria possess sequential energy transfer (SET), which show remarkably high overall energy transfer efficiencies (Φoverall ~ 95%).[1] However, most of artificial LHSs that mimic the prototype show Φoverall values less than 70% (see published paper for more examples).[2-4] For artificial systems, it is still challenging to perfectly mimic the functions of protein scaffolds in organizing bacteriochlorophyll (BChl) pigments via non-covalent binding[5] and promoting energy transfer via long-lasting quantum coherence[6]. On this account, we then paid attention to LHSs of green photosynthetic bacteria. Similar to higher plants, it contains hydrophobic cellular structures called chlorosomes. Direct supramolecular self-aggregation of BChl c pigments exists in chlorosomes without the aid of protein scaffolds.[7] Through cooperative self-assembly, densely packed nanostructures are generated with the involvement of 50000~250000 BChl c pigments.[8,9] Ascribing to high packing density and ordered packing mode of pigments, it efficiently collects light energy even under weak light intensity,[10] which transfer to reaction centers via SET.[11] Despite so many advantages, I feel puzzled that merely limited SET systems derived from BChl c have been reported.[12,13] The main cause probably lies in the fact that BChl c mimics have strong propensities to self-aggregate instead of co-assembling with other π-conjugated molecules.[14-16] Therefore, LHSs have been constructed via covalently attaching artificial antenna molecules to BChl c mimics.[12,13] It not only leads to the inevitably synthetic complexities, but also difficulties in the adjustment of donor/acceptor (D/A) ratios.
Considering that BChl c aggregates represent simpler yet efficient light harvesting antennae, we eventually decided to construct artificial LHS based on the natural prototype. As a PhD student major in supramolecular chemistry, I asked myself the second issue: whether green photosynthetic bacteria inspired SET system could be constructed purely via non-covalent strategy? To attain this objective, it is essential to seek for appropriate molecular basis. Fortunately, our group has developed a type of platinated (hetero)acene-based supramolecular monomers.[17-19] Similar to BChl c, they assemble into long-range-ordered helical aggregates via cooperative self-assembly (Figure 1). Intriguingly, by altering (hetero)acene units, spectral properties of supramolecular monomers can be regulated (Figure 1), which have potential to serve as D/A pairs. Accordingly, platinated (hetero)acene-based monomers are considered as suitable candidates. Once the molecular basis was decided, I encountered the third issue: how to determine (hetero)acene units of assembly monomers? With the aid of DFT/TDDFT calculations, spectral properties of supramolecular monomers possessing various (hetero)acenes were calculated. Through this method, I gained deeper insights into feasible combinations of D/A pairs. Of course, this method generally requires expensive computational cost while the results may sometimes less accurate. Recently, machine learning has been combined with DFT/TDDFT calculations to predict and study excitation energy transfer between D/A pairs in natural LHSs without the need of expensive computational costs.[6a,20] In the future, it maybe also a powerful tool for rational design of D/A pairs in artificial LHSs.
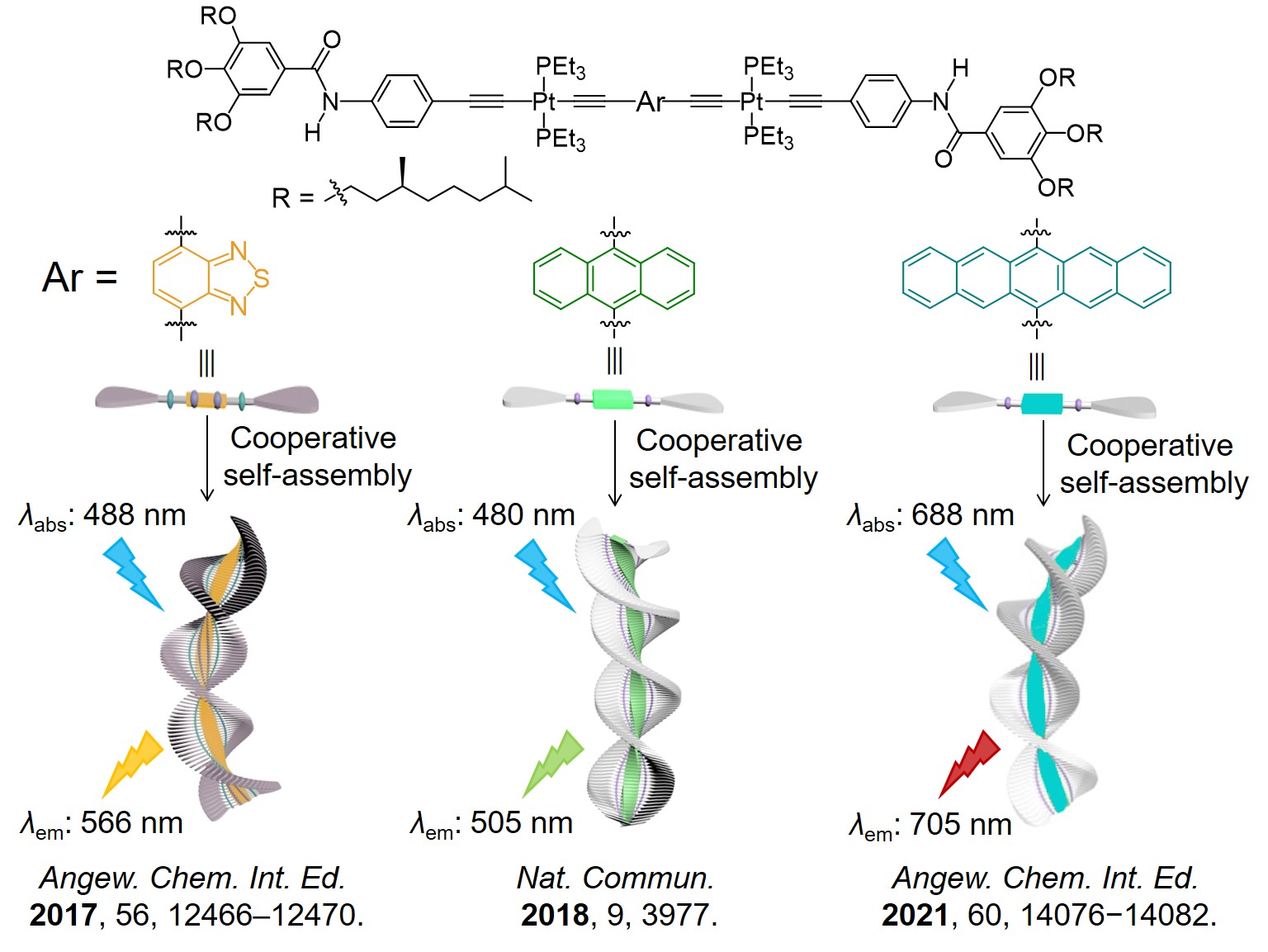
Figure 1. Platinated (hetero)acene-based supramolecular monomers developed by our group.
The last issue I concerned most was whether monomers could co-assemble into supramolecular copolymers? Through supramolecular copolymerization, the average distances of D/A pairs would be reduced, thus facilitating to excitation energy transfer. Our recently studies have confirmed that, due to the presence of bulky Pt(PEt3)2 moieties, hydrogen bonding interactions are the primarily driving forces to hold monomers together, whereas the close stacking of (hetero)acenes are prohibited.[17] Hence, supramolecular copolymerization is likely to occur via random mixing of monomers driven by hydrogen bonds.
Based on these considerations, a green photosynthetic bacteria inspired SET system is constructed via supramolecular copolymerization. Due to highly structural similarities, platinated (hetero)acene-based monomers 1–3 co-assemble into long-range-ordered supramolecular copolymers via replacing donors in light harvesting matrixes (Figure 2b). For binary copolymeric systems, they not only display excellent energy transfer efficiencies (75%~96%), but also high exciton migration rates (1014 orders of magnitude). The ternary supramolecular copolymers with a two-step SET show a Φoverall value of 87.4%, higher than most of the state-of-the-art supramolecular LHSs that mimic LHSs of purple photosynthetic bacteria (Φoverall < 70%). These superior properties originate from dense packing of co-assembled monomers in long-range-ordered supramolecular copolymers, mimicking pigment aggregation pattern in green photosynthetic bacteria (Figure 2a).
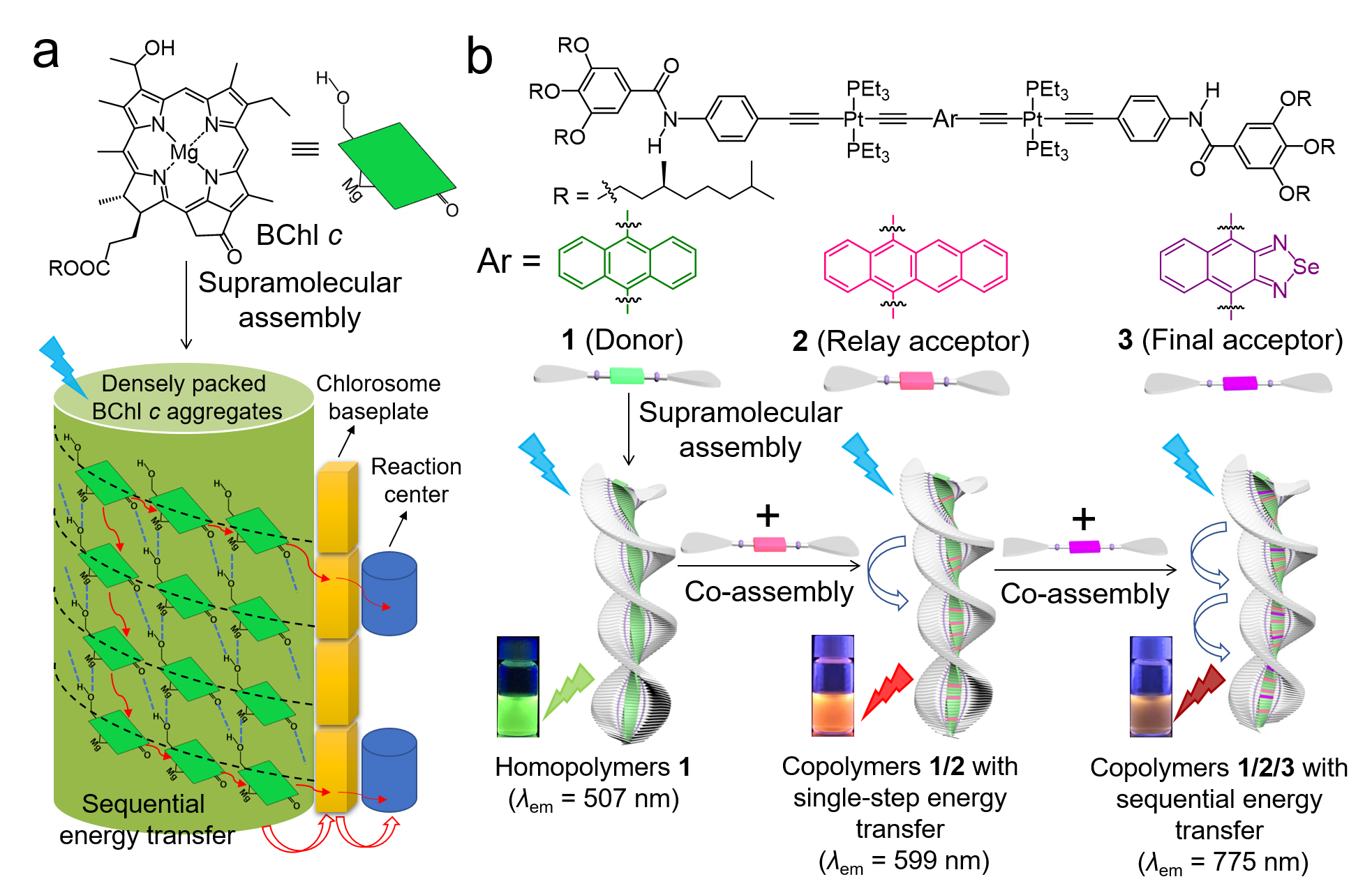
Figure 2. (a) Direct supramolecular self-aggregation of bacteriochlorophyll c (BChl c) into light harvesting antenna, together with sequential energy transfer in green photosynthetic bacteria. (b) Supramolecular copolymerization of 1–3 (cartoon symbols with green, pink, and purple color) with sequential energy transfer behaviors.
This study provides a rare example of bioinspired SET system constructed via supramolecular copolymerization. Notably, Meijer’s group has reported a SET system constructed via the same strategy.[13] However, supramolecular copolymerization merely occurs between porphyrin-based relay and Zn(II)-porphyrin-based final acceptors, whereas oligo(p-phenylene vinylene)-based donors are covalently linked to both acceptors. For the current study, all compositions (donors, relay and final acceptors) co-assemble purely via supramolecular copolymerization. This unique advantage can be ascribed to the introduction of platinated (hetero)acene-based monomers, which ensures spectral tunability without the need of major structural changes.
For photosynthetic organisms, light energy is transferred to reaction centers via SET and converted into chemical energy via photosynthesis processes. For artificial SET systems, they have been exploited to power photosynthesis in very limited studies.[2,21-23] In the final stage of this study, I attempted to combine the bioinspired SET system with chemical reactions. Since it primarily emits near infrared light from the final acceptor, it may be used to support near infrared light triggered polymerization.[24] However, the exploration was indeed unsuccessful. I think it is a pity for the study.
For further details, please read our article in Nature Communications: https://www.nature.com/articles/s41467-022-31094-w.
References:
[1] Hu, X., Damjanović, A., Ritz, T. & Schulten, K. Architecture and mechanism of the light-harvesting apparatus of purple bacteria. Proc. Natl. Acad. Sci. U.S.A. 95, 5935–5941 (1998).
[2] Hao, M. et al. A supramolecular artificial light-harvesting system with two-step sequential energy transfer for photochemical catalysis. Angew. Chem. Int. Ed. 59, 10095–10100 (2020).
[3] Sun, G. et al. A highly efficient artificial light-harvesting system with two-step sequential energy transfer based on supramolecular self-assembly. J. Mater. Chem. A 8, 9590–9596 (2020).
[4] Li, J.-J., Zhang, H.-Y., Dai, X.-Y., Liu, Z.-X. & Liu, Y. A highly efficient light-harvesting system with sequential energy transfer based on a multicharged supramolecular assembly. Chem. Commun. 56, 5949–5952 (2020).
[5] Otsuki, J. Supramolecular approach towards light-harvesting materials based on porphyrins and chlorophylls. J. Mater. Chem. A 6, 6710–6753 (2018).
[6] (a) Ullah, A. & Dral, P. O. Predicting the future of excitation energy transfer in light-harvesting complex with artificial intelligence-based quantum dynamics. Nat. Commun. 13, 1930 (2022); (b) Kong, F.-F. et al. Wavelike electronic energy transfer in donor–acceptor molecular systems through quantum coherence. Nat. Nanotechnol. 17, 729–736 (2022).
[7] Jochum, T. et al. The supramolecular organization of self-assembling chlorosomal bacteriochlorophyll c, d, or e mimics. Proc. Natl. Acad. Sci. U.S.A. 105, 12736–12741 (2008).
[8] Orf, G. S. & Blankenship, R. E. Chlorosome antenna complexes from green photosynthetic bacteria. Photosynth. Res. 116, 315–331 (2013).
[9] Jesorka, A., Balaban, T. S., Holzwarth, A. R., & Schaffner, K. Angew. Chem. Int. Ed. Engl. 35, 2861–2863 (1996).
[10] Chappaz-Gillot, C. et al. Anisotropic organization and microscopic manipulation of self-assembling synthetic porphyrin microrods that mimic chlorosomes: bacterial light-harvesting systems. J. Am. Chem. Soc. 134, 944–954 (2012).
[11] Mimuro, M. et al. Excitation energy flow in chlorosome antennas of green photosynthetic bacteria. J. Phys. Chem. 93, 7503–7509 (1989).
[12] Röger, C., Miloslavina, Y., Brunner, D., Holzwarth, A. R. & Würthner, F. Self-assembled zinc chlorin rod antennae powered by peripheral light-harvesting chromophores. J. Am. Chem. Soc. 130, 5929–5939 (2008).
[13] Hoeben, F. J. M. et al. Influence of supramolecular organization on energy transfer properties in chiral oligo(p-phenylene vinylene) porphyrin assemblies. J. Am. Chem. Soc. 129, 9819–9828 (2007).
[14] Matěnova, M. et al. Energy transfer in aggregates of bacteriochlorophyll c self-assembled with azulene derivatives. Phys. Chem. Chem. Phys. 16, 16755–16764 (2014).
[15] Miyatakea, T. & Tamiaki, H. Self-aggregates of natural chlorophylls and their synthetic analogues in aqueous media for making light-harvesting systems. 254, 2593–2602 (2010).
[16] Orf, G. S. et al. Polymer−chlorosome nanocomposites consisting of non-native combinations of self-assembling bacteriochlorophylls. Langmuir 33, 6427−6438 (2017).
[17] Han, Y., Yin, Y., Wang, F. & Wang, F. Single-photon near-infrared-responsiveness from the molecular to the supramolecular level via platination of pentacenes. Angew. Chem. Int. Ed. 60, 14076−14082 (2021).
[18] Gao, Z., Han, Y. & Wang, F. Cooperative supramolecular polymers with anthracene‒endoperoxide photo-switching for fluorescent anti-counterfeiting. Nat. Commun. 9, 3977 (2018).
[19] Wang, X. et al. Cooperative supramolecular polymerization of fluorescent platinum acetylides for optical waveguide applications. Angew. Chem. Int. Ed. 56, 12466–12470 (2017).
[20] (a) Häse, F. et al. Designing and understanding light-harvesting devices with machine learning. Nat. Commun. 11, 4587 (2020); (b) Häse, F., Kreisbeck, C. & Aspuru-Guzik, A. Machine learning for quantum dynamics: deep learning of excitation energy transfer properties. Chem. Sci., 8, 8419–8426 (2017).
[21] Zhang, D. et al. Artificial light-harvesting metallacycle system with sequential energy transfer for photochemical catalysis. J. Am. Chem. Soc. 143, 1313−1317 (2021).
[22] Jia, P.-P. et al. Orthogonal self-assembly of a two-step fluorescence-resonance energy transfer system with improved photosensitization efficiency and photooxidation activity. J. Am. Chem. Soc. 143, 399−408 (2021).
[23] Li, Y. et al. “On/off” switchable sequential light-harvesting systems based on controllable protein nanosheets for regulation of photocatalysis. ACS Nano 16, 8012−8021 (2022).
[24] Stafford, A. et al. Catalyst halogenation enables rapid and efficient polymerizations with visible to far-red light. J. Am. Chem. Soc. 142, 14733–14742 (2020).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in