A generic approach to preparing autologous cancer therapeutic vaccines
Published in Bioengineering & Biotechnology
Vaccines, the medical formulations that generally contain specific antigens and adjuvants, help the body fight against disease. They can educate the immune system to recognize and destroy pathogens (bacteria, viruses, cancer cells, etc.) in the body. Back in the 1790s, Edward Jenner developed the world’s first vaccine by using cowpox to trigger immunity against smallpox. In 1885, Louis Pasteur successfully tested his rabies vaccine on a human subject. Since then, a lot of vaccines against diphtheria, tetanus, anthrax, cholera, tuberculosis, and more were successfully developed and made a positive impact on human life.1 Mounting evidence has validated the efficacy of vaccines in preventing diseases caused by viruses and bacteria.
Unfortunately, in the case of cancer, the situation is much more complicated and this has greatly hindered the development of cancer vaccines, especially those therapeutic vaccines which are administered in patients already afflicted with cancer. Unlike the foreign viruses and bacteria, cancer cells are developed from the normal healthy cells in the body. Although they go through some abnormal changes, they still closely resemble the normal cells and the immune system cannot readily distinguish them from other cells in the body. Meanwhile, each individual’s cancer cells exhibit distinct genetic and phenotypic profiles, and even the cancer cells in the same tumour have their unique antigens. Furthermore, the immunosuppressive tumour microenvironment, a complex niche consisting of various immunosuppressive cells (M2-like macrophages, MDSCs, Tregs, etc.) and immunosuppressive cytokines (IL-10, TGF-β, etc.), is another great barrier to effector immune cells.
Generally, conventional cancer therapeutic strategies, such as radiotherapy, chemotherapy, and photodynamic therapy, trigger apoptosis of many cancer cells. These apoptotic cells are silently cleared by phagocytes and cannot stimulate the immune system. Meanwhile, phagocytic clearance of the apoptotic cells by macrophages also triggers the release of immunosuppressive cytokines2 and further enhances the immunosuppression of tumour microenvironment. One day, an idea came into view: “why don’t we exploit these apoptotic cells to initiate anti-tumour immune responses?”
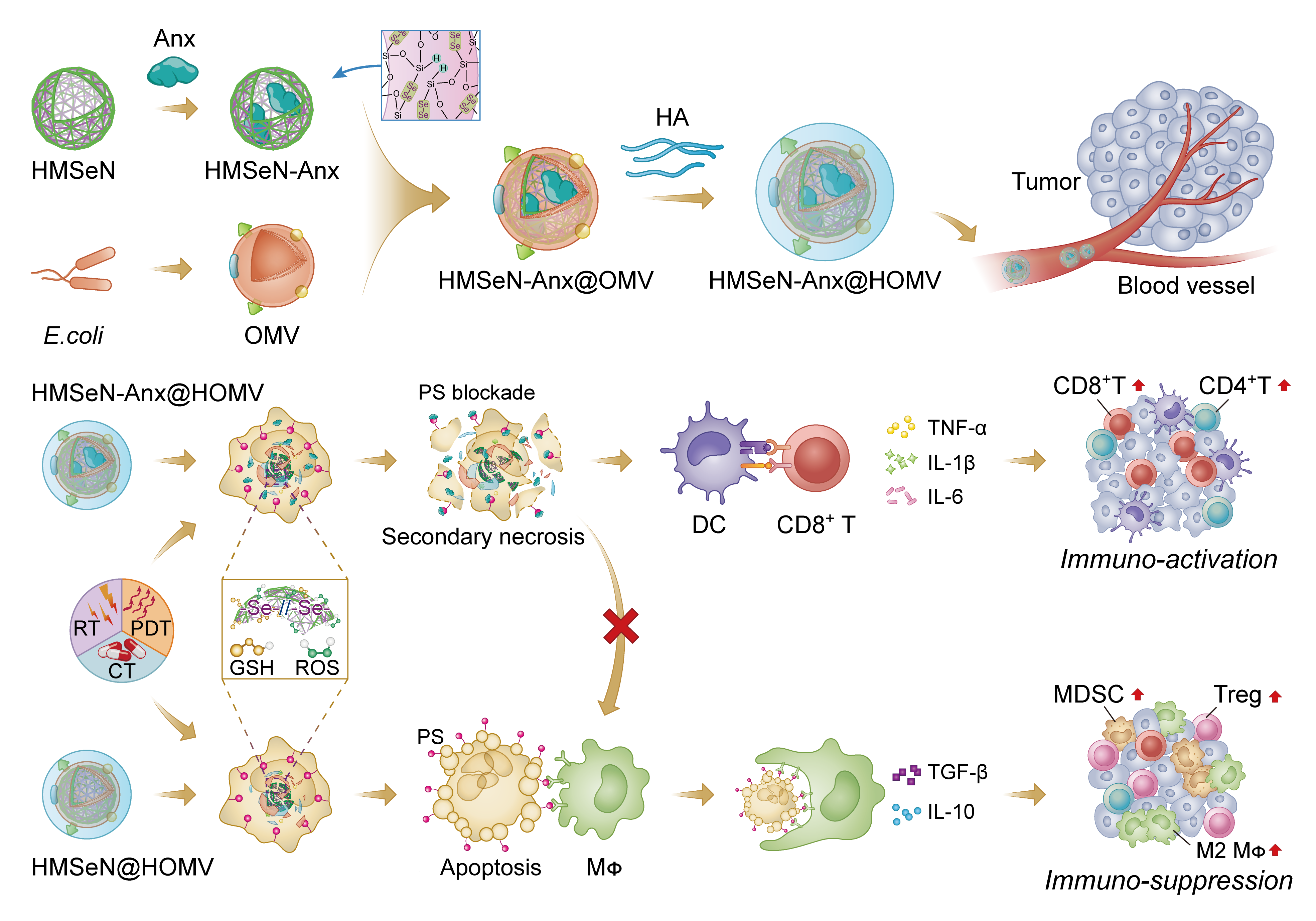
In our most recent study in Nature Biomedical Engineering, we developed a generic approach for preparing autologous tumour therapeutic vaccines through rewiring immunosuppressive apoptosis signalling into immunostimulatory secondary necrosis to provoke personalized anti-tumour immune responses (Figure 1). In the method, a biodegradable diselenide-bridged hollow mesoporous organosilica nanocapsule (HMSeN) was prepared for the delivery of Annexin A5 (Anx) protein, a commonly used molecular imaging agent for apoptosis detection.3 The resulting nanocapsule achieved “on-demand” burst release of Anx due to oxidation/reduction-responsive diselenide bond cleavage4 in oxidative tumour microenvironment or bioreductive intracellular environment. In this way, the released Anx blocked phosphatidylserine (PS) exposure on dying tumour cells, prevented the recognition and the subsequent phagocytosis of macrophages. These dying tumour cells would further undergo secondary necrosis, which simultaneously rendered the tumour immunogenic and inflamed the tumour microenvironment.
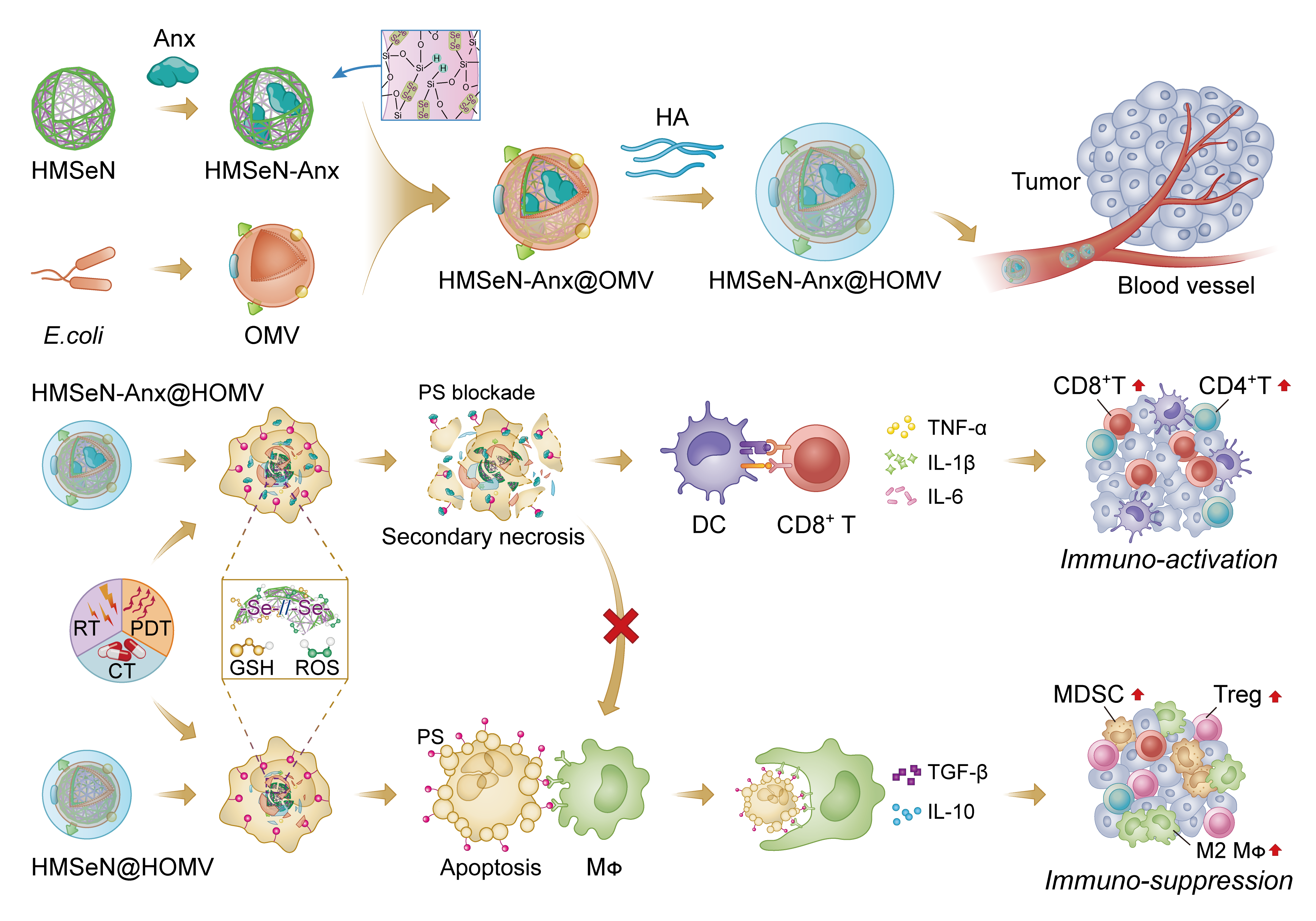 Figure 1: The process of preparing autologous cancer therapeutic vaccines Source file: https://doi.org/10.1038/s41551-020-0599-5
Figure 1: The process of preparing autologous cancer therapeutic vaccines Source file: https://doi.org/10.1038/s41551-020-0599-5
Given that many of the current cancer treatments can induce apoptosis, our approach enables effective targeting of each patient’s tumour with vaccines produced “just in me” by exploiting the treatment-induced apoptosis. Hopefully, with the optimization of this technology, our proposed approach can be applied to cancer patients in the near future. A patent describing this autologous cancer therapeutic vaccine has been filed.
References
1. Rey-Jurado, E. et al. Assessing the importance of domestic vaccine manufacturing centers: an overview of immunization programs, vaccine manufacture, and distribution. Front Immunol. 9, 26 (2018).
2. Huynh, M. -L. N., Fadok, V. A. & Henson, P. M. Phosphatidylserine-dependent ingestion of apoptotic cells promotes TGF-β1 secretion and the resolution of inflammation. J. Clin. Invest. 109, 41-50 (2002).
3. Logue, S. E., Elgendy, M. & Martin, S. J. Expression, purification and use of recombinant annexin V for the detection of apoptotic cells. Nat. Protoc. 4, 1383-1395 (2009).
4. Shao, D. et al. Bioinspired diselenide-bridged mesoporous silica nanoparticles for dual-responsive protein delivery. Adv. Mater. 30, 1801198 (2018).
Written by Ling Li and Xiaoyuan (Shawn) Chen.
Follow the Topic
-
Nature Biomedical Engineering
This journal aspires to become the most prominent publishing venue in biomedical engineering by bringing together the most important advances in the discipline, enhancing their visibility, and providing overviews of the state of the art in each field.
Related Collections
With Collections, you can get published faster and increase your visibility.
Implantable wireless communication technologies
Publishing Model: Hybrid
Deadline: Nov 28, 2026
Medical Ultrasound: Emerging Techniques and Applications
Publishing Model: Hybrid
Deadline: Jan 29, 2027




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in