Activation of P2RX7 boosts antitumor immune responses and sensitizes tumors to immunotherapy
Published in Cancer
Like
Be the first to like this
Beside classical chemotherapeutic approaches and targeted therapies, the emergence of immunotherapy has revolutionized cancer treatment in the last few years. However, many patients are unresponsive or become resistant to treatment, highlighting the need of new therapeutic strategies to improve patients’ outcome.
Since chronic inflammation is a cancer-promoting factor, we thought that the purinergic receptor P2RX7 would favor tumor growth given its prominent role in the release the pro-inflammatory cytokine IL-1β. However, we have shown that P2RX7 restrains tumor growth (Hofman et al., 2015).
Unlike other receptors of the P2X family, P2RX7 is activated by high extracellular ATP (eATP) levels found in inflamed and cancerous tissues. Its activation leads to calcium influx, potassium efflux and the assembly of the NLRP3 inflammasome and consequently the release of IL-1β and IL-18. Moreover, its activation leads to cell permeabilization that ultimately leads to cell death.
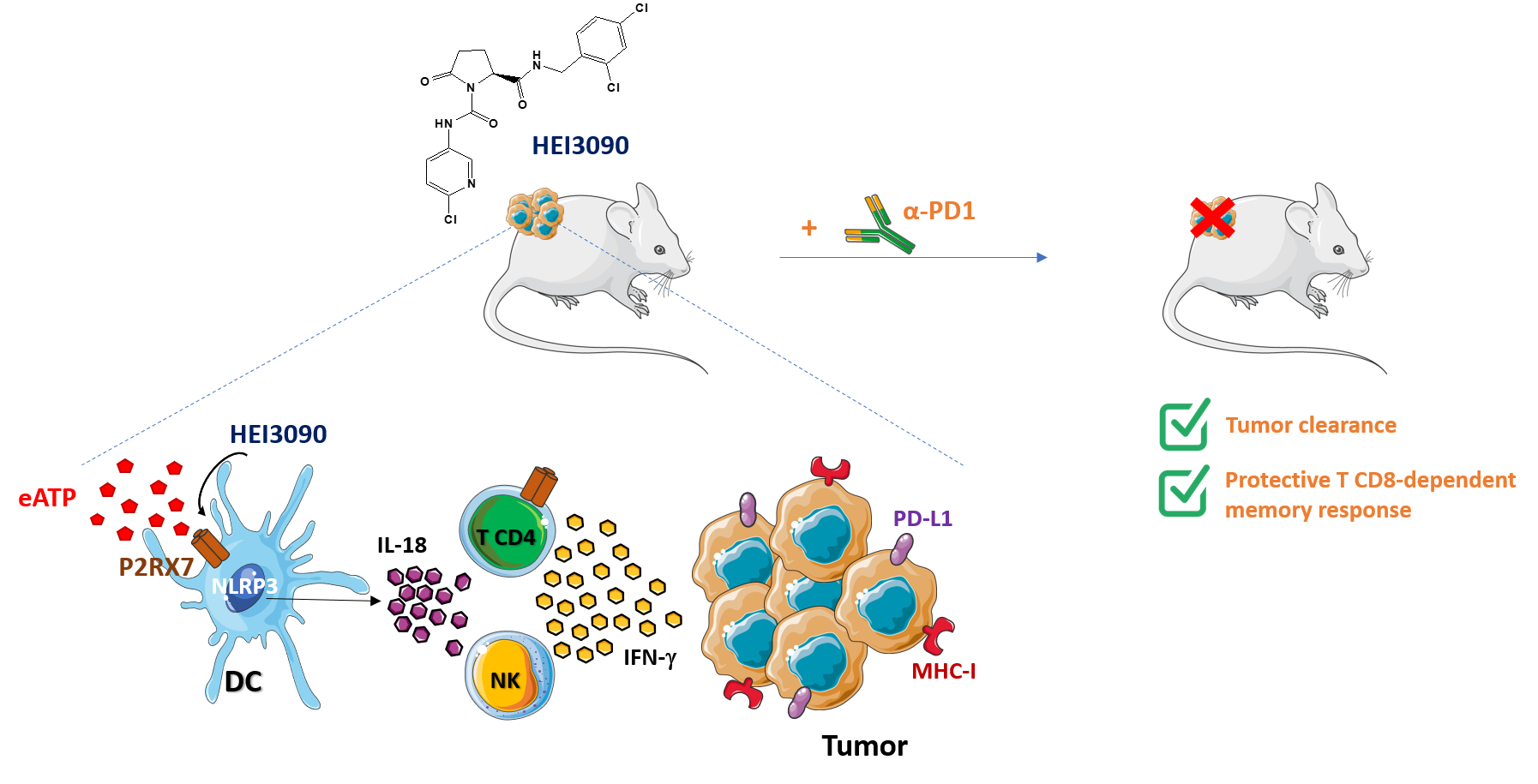
Given its antitumor proprieties and its ability to modulate the immune response, we proposed that enhancing P2RX7's activation would be a promising therapeutic approach. We developed a series of molecules and screened them in vitro for their ability to (i) enhance P2RX7-mediated calcium influx during eATP exposure and (ii) show no toxicity in the absence of P2RX7 or eATP. Our goal was to select molecules that would show antitumor proprieties highly dependent on P2RX7’s expression in an ATP-rich tumor microenvironment without any toxicity in normal tissues.
The most promising and efficient molecule is named HEI3090 (patent WO2019185868A1) and was selected to test our hypothesis. HEI3090 is a positive modulator of P2RX7 and requires the presence of eATP to enhance P2RX7’s activities, namely calcium influx and cell permeabilization.
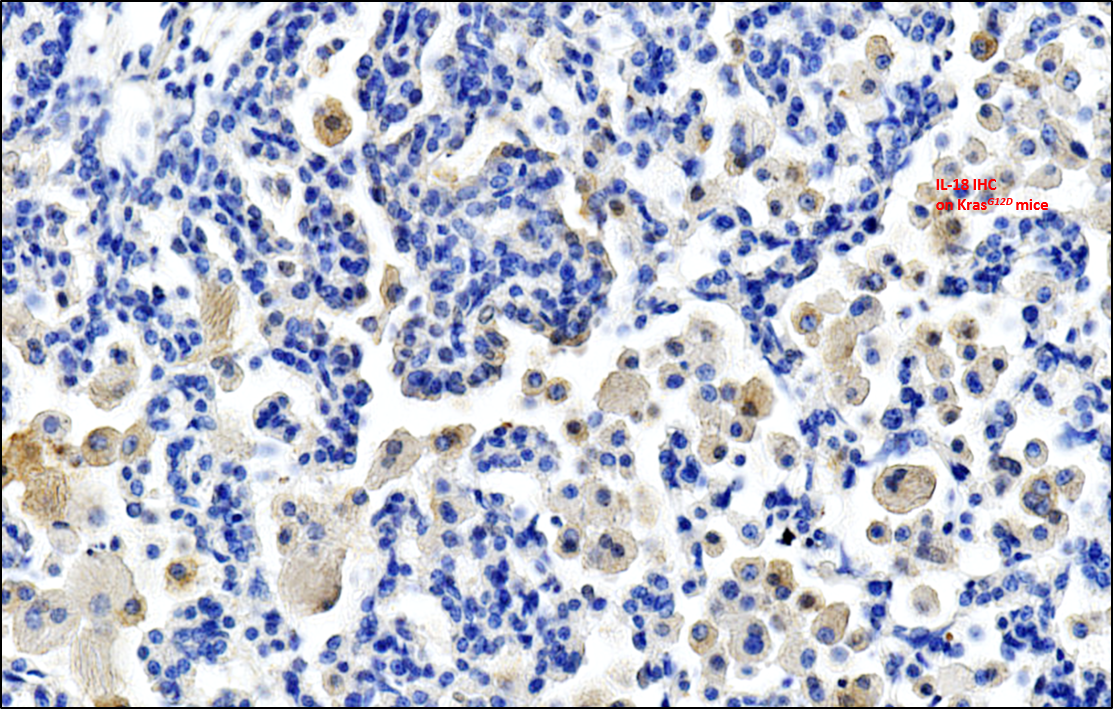
In order to test the antitumoral proprieties of HEI3090, we used a transplantable syngeneic immunocompetent mouse model of lung tumor cells (LLC, Lewis Lung Carcinoma). We have shown that HEI3090 inhibits efficiently tumor growth and targets P2RX7’s expressing immune cells to trigger an antitumor immune response and enhance tumor immunogenicity. Therefore, combining HEI3090 with immune checkpoint inhibitors was a very interesting approach. Indeed, the combination of HEI3090 with αPD-1 cured 80% of mice transplanted with LLC cells and decreased tumor burden by 60% in an oncogene-induced mouse model of lung cancer (LSL KrasG12D). Cured mice were also protected against a tumor re challenge, highlighting the ability of HEI3090 to induce a robust immune memory response.
HEI3090’s antitumor effect relies on the release of IL-18 by the NLRP3 inflammasome. IL-18 (originally named IFN-γ-inducing factor) is a cytokine that drives IFN-γ production by T cells and NK cells, and enhances therefore their cytotoxicity against tumor cells. However, activation of P2RX7 with HEI3090 does not enhance IL-1β release. This finding was very surprising but reassuring since IL-1β a pro-inflammatory cytokine involved in immune escape. Therefore, only the antitumoral effect of the P2RX7/NLRP3 pathway was boosted using HEI3090, with an increased release of IL-18.
HEI3090’s effect was mainly studied in lung cancer since it is the focus of our laboratory. We have also shown that activation of P2RX7 was promising in a melanoma mouse model. Indeed, HEI3090 efficiently inhibits melanoma tumor growth by triggering antitumor immunity. Moreover, the combination of HEI3090 and αPD-1 also increased αPD-1 efficacy. These results demonstrate that the antitumor activity of HEI3090 follows the same rule in all tumor models tested.
By activating P2RX7 with HEI3090, we highlighted the importance of the P2RX7/NLRP3/IL-18 pathway in tumor growth inhibition. IL-18 acts as a key factor in activating antitumor immune responses and in sensitizing tumors to immunotherapy. Boosting this pathway is a very promising strategy since it could increase the number of patients eligible for immunotherapies and have synergistic effects with immunotherapies. In fact, in silico analyses showed that lung cancer patients expressing high levels of P2RX7 and PD-L1 would highly benefit from a combo therapy (HEI3090+immunotherapy).
Collectively, our results unravel a new therapeutic strategy in lung cancer treatment by increasing the number of patients eligible for immunotherapies and boosting their efficacy.
Read the article
Follow the Topic
Cancer Biology
Life Sciences > Biological Sciences > Cancer Biology
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
This collection welcomes submissions based on studying preclinical models, as well as population-wide and clinical studies. Studies that advance our understanding of mechanisms behind healthy aging are also welcomed. Clinical research of interest will include epidemiological studies, observational studies, longitudinal cohort studies, systematic reviews and clinical trials.
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
A selection of recent articles that highlight issues relevant to the treatment of neurological and psychiatric disorders in women.
Publishing Model: Hybrid
Deadline: Ongoing



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in