Arginine-modified black phosphorus quantum dots with dual excited states for enhanced electrochemiluminescence in bioanalysis
Published in Chemistry
Electrochemiluminescence (ECL) is a light-emitting process, in which the excited state species (R*) are generated via exergonic electron transfer and exchange between the electrogenerated intermediates, following the radiative transitions to the ground state (S0). Generally, the produced R* can be either the lowest excited singlet state (S1) species (1R*) or the triplet state (T1) species (3R*). In this work, Ju’s group found a dual excited states-mediated ECL mechanism with the transitions from both S1 and T1 by using black phosphorus quantum dots (BPQDs) as a nanoemitter, and proposed a strategy to modulate the excited states via arginine (Arg or R) modification of BPQDs (R-BPQDs) to enhance the ECL efficiency.
The BPQDs can be conveniently modified with Arg via both the electrostatic interaction of the surface defects with delocalized positive charge of guanidine group and the hydrogen bond interaction between the electron-withdrawing guanidine group and PxOy moiety. The presence of Arg on BPQDs passivates the oxidation defects of BPQDs and increases the negative surface charge due to the exposure of carboxyl group with a pKa value of 2.17, and thus endows BPQDs with better stability, solubility and modifiability for extending the bioanalysis application, which were demonstrated by the Raman spectra, X-ray photoelectron spectroscopic spectra and the Fourier transform infrared spectra (Fig. 1).
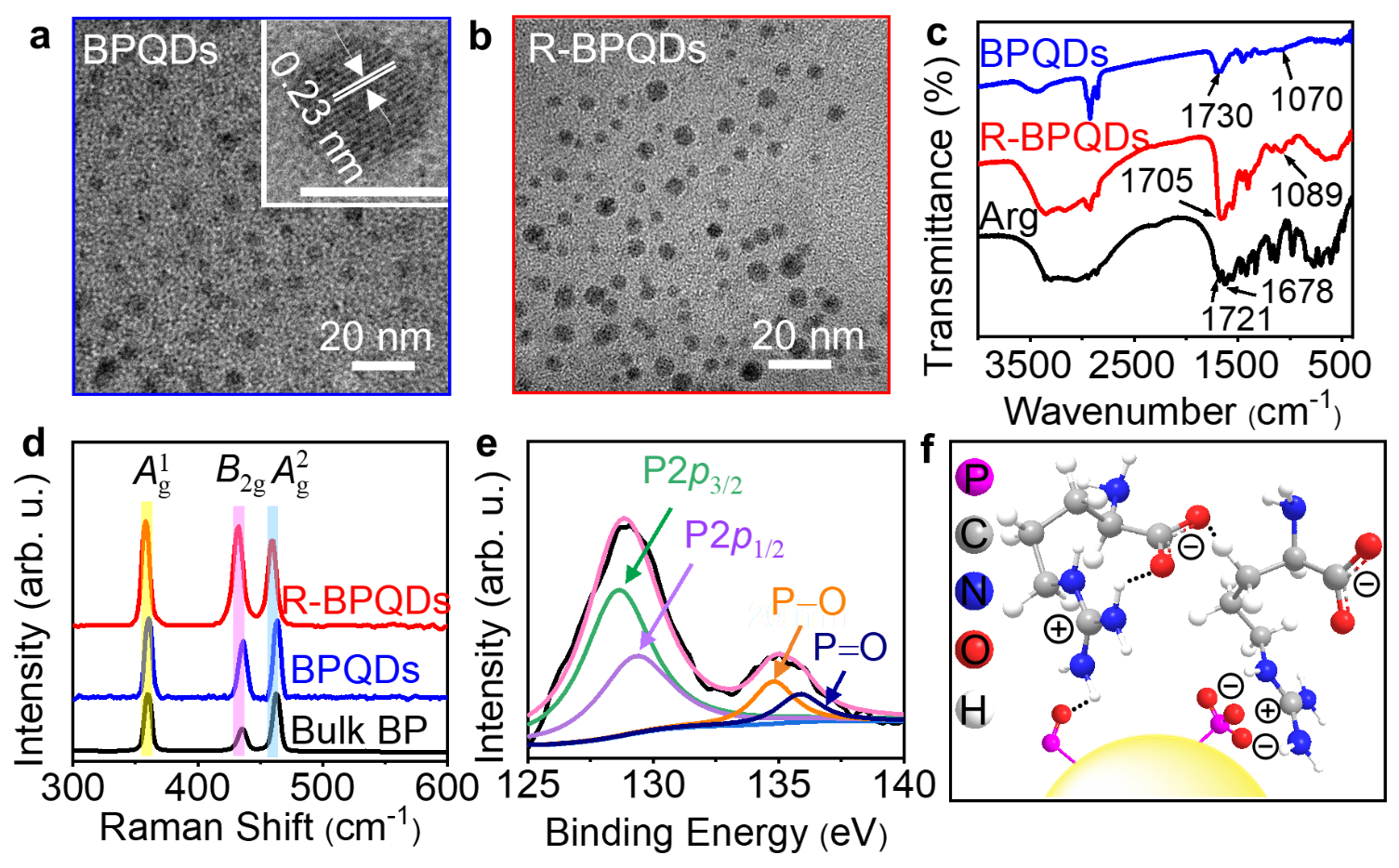
Fig. 1 Structural and chemical characterization before (BPQDs, blue) and after Arg modifification (R-BPQDs, red).
Compared to bare glassy carbon electrode (GCE), the cathodic electrochemical process of BPQDs/GCE showed a weak reduction peak of BPQDs at around −1.22 V, which shifted to −1.15 V and became more distinct after introducing the electron-withdrawing guanidine group by Arg modification (Fig. 2). In the presence of cathodic co-reactant K2S2O8, R-BPQDs/GCE showed a cathodic ECL emission at −1.20 V, which was 25 folds stronger than that of BPQDs/GCE. The cathodic ECL efficiency of R-BPQDs was 3.2 times higher compared to BPQDs.
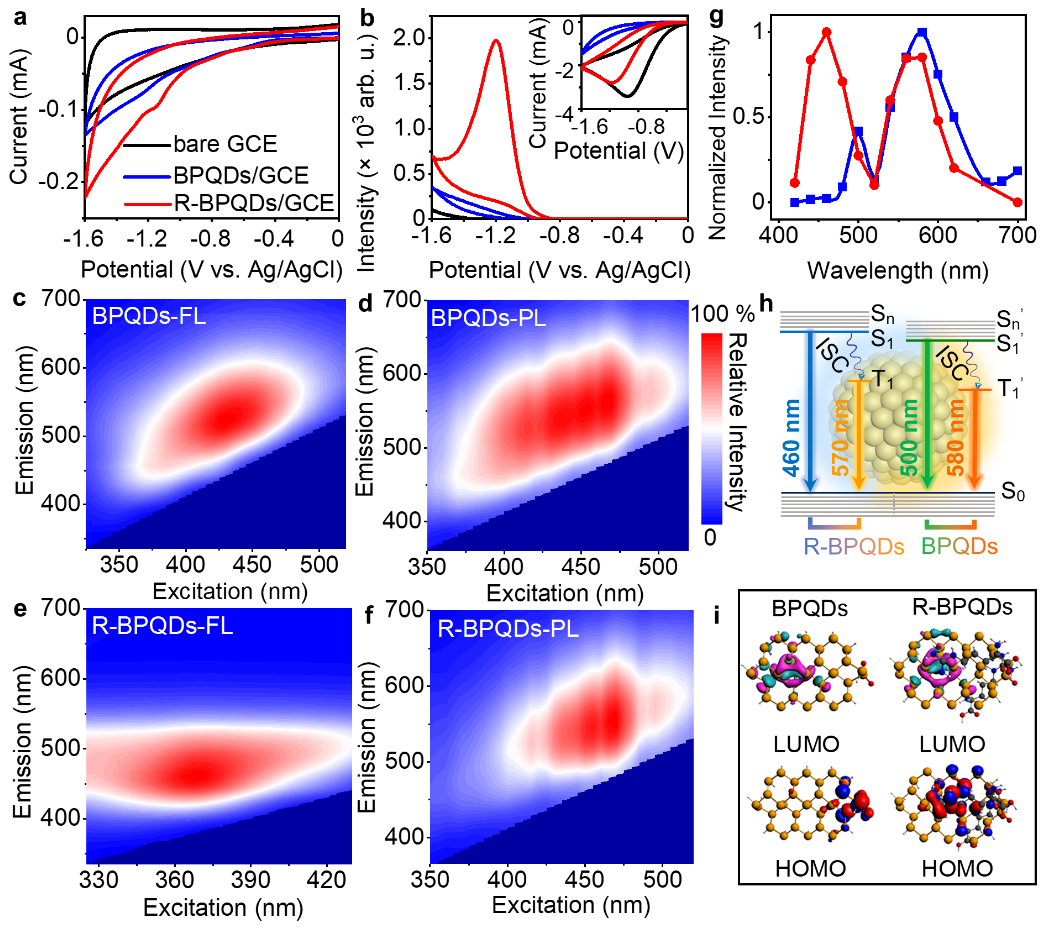
and after Arg modifification (R-BPQDs/GCE, red).
The ECL mechanism containing two radiative transitions from both S1 and T1 to S0 was demonstrated by the ECL spectra with two emission peaks as observed from the fluorescence (FL) and phosphorescence (PL) emission of BPQDs, and the photoluminescence decay spectra showing both ns- and μs-level decay lifetimes, and also proved by two experimental band gaps calculated from the Tauc plots of the UV-vis-NIR diffuse reflectance spectra. Besides, the photoluminescence lifetimes did not obviously change at temperatures from 170 to 310 K, which excluded the possibility of thermally activated delayed fluorescence process, and further indicated the existence of two radiative transitions from both S1 and T1 for ECL emission.
According to the electrostatic and hydrogen bond interactions between Arg and oxidation defects of BPQDs, time-dependent density functional theory computation was implemented to rationalize above conclusion. The existence of oxidation defects resulted in the localized highest occupied molecular orbital (HOMO) of BPQDs at the defect sites, which hindered the charge transfer as a trap state, and thus weakened the emission intensity. The Arg modification passivated the surface oxidation defects, accordingly leading to the delocalization of HOMO of R-BPQDs to the central zone, and thus the change of the electron transition channel and the greater spatial overlap of the HOMO with the lowest unoccupied molecular orbital (LUMO), which significantly improved the emission oscillator strength and the charge transfer capability. By comparing the ECL changes upon different amino acid modifications, it was concluded that the cathodic ECL enhancement of R-BPQDs was attributed to the presence of electron-withdrawing guanidine group, which stabilized the adjacent R-BPQDs•− anion radical after electrochemically injecting electron into LUMO of R-BPQDs. Thus the relative cathodic ECL efficiency of R-BPQDs vs. 1 mM [Ru(bpy)3]2+ with 0.1 M K2S2O8 reached 48%, which was much larger than those of thermally activated delayed fluorescence emitters reported previously, providing an alternative ECL nanoemitter for bioanalysis.
To demonstrate the application of R-BPQDs in ECL bioanalysis, the Arg attached on BPQDs has been replaced by Arg-containing peptide RRGDS, where RGDS is a peptide specifific to integrin. By coating RRGDS-BPQDs on multi-wall carbon nanotubes modified GCE, the integrin-rich cells were bound to the electrode surface via the recognition of RGDS to integrin, which led to a sensitive ECL method for the evaluation of integrin inhibitor. The obtained inhibiting efficiency to integrin on A549 cells demonstrated the practicability of the ECL of BPQDs and the modulating strategy. The new ECL mechanism and the modulation strategy open a new avenue to decipher more ECL systems for broadening the ECL applications of nanoemitters.
More details on this work can be found in “Arginine-modified black phosphorus quantum dots with dual excited states for enhanced electrochemiluminescence in bioanalysis” published in Nature Communications on November 26, 2022. Link: https://doi.org/10.1038/s41467-022-35015-9
Short biography
Huangxian Ju received his BS, MS and PhD degrees from Nanjing University during 1982–1992, and was a postdoc in Montreal University (Canada) in 1996–1997. He became an associate and full professor of Nanjing University in 1993 and 1999. He is currently the director of State Key Laboratory of Analytical Chemistry for Life Science, fellow of the International Electrochemical Society and fellow of the Royal Society of Chemistry. His research interests focus on analytical biochemistry, biosensing and molecular diagnosis. He has authored 84 patents (43 approved), 6 English books, 7 Chinese books and 20 chapters, and published 832 papers in different journals with h-index of 102 (Google Scholar h-index 111 with more than 46500 citations). He was awarded with a National Science Fund for Distinguished Young Scholars in 2003, Changjiang Scholar Professor by the Ministry of Education in 2007, the National Key Talents in The New Century and the chief scientist of the national 973 project in 2009, the special government allowance of the State Council in 2011, the first Leici Outstanding Achievement Award of Chinese Chemical Sensors in 2019, and 2022 Advances in Measurement Science Lectureship Awards of American Chemical Society, as well as 19 natural science or science and technology prizes from Ministry of Education or Province Government.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in