Behind the paper: Unraveling the molecular mechanism behind COVID-19 pulmonary pathologies associated with ferroptosis
Published in Chemistry, Microbiology, and Cell & Molecular Biology
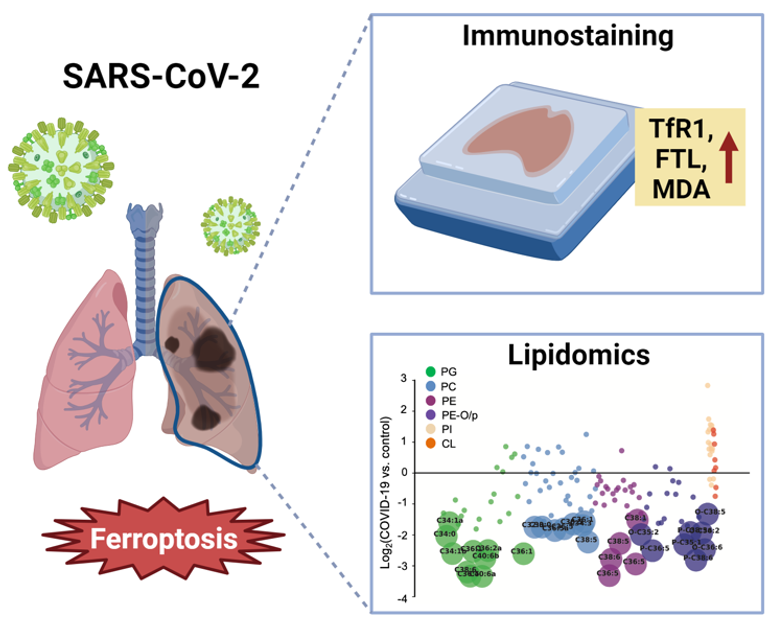
The COVID-19 pandemic caused by SARS-CoV-2 infection was a global health crisis. Future pandemics involving related coronaviruses are likely to emerge in the future. Upon infection, the virus primarily targets the respiratory tract, including acute respiratory distress syndrome (ARDS), leading to mortality in some individuals1. ARDS shows lung manifestation of acute lung injury (ALI) such as diffuse alveolar damage (DAD). Non-acute lung injury (non-ALI), such as vascular congestion and microthrombi, is also manifestation of COVID-192. COVID-19 pathology is thus characterized by a complex interplay between viral replication, immune responses, and tissue damage3.
Current treatments for COVID-19 alleviate symptoms. Antiviral drugs such as remdesivir have been authorized for use in treating COVID-194. These medications work by inhibiting viral replication and are beneficial when administered early in the course of the illness. Anti-inflammatory corticosteroids, such as dexamethasone and prednisone, reduce inflammation and improve outcomes in hospitalized patients, especially those with severe disease5. The molecular mechanism governing COVID-19 lung pathology remains unclear and is crucial for developing effective therapies for this aspect of the disease.
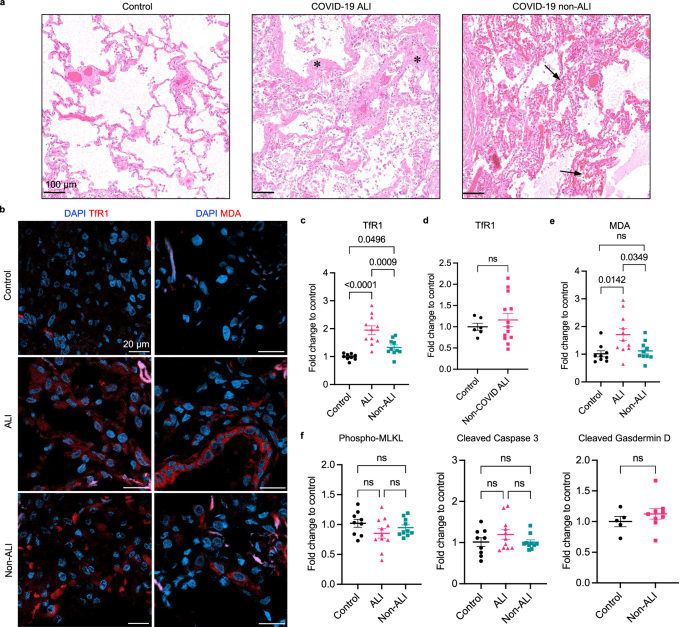
Our study was motivated by the need to understand the mechanism underlying COVID-19 lung damage during the initial outbreak of the pandemic in 2020. We obtained COVID-19 patient lung samples encompassing ALI and non-ALI pathologies. By examining cell death markers in human COVID-19 lung tissue, we observed molecular signatures of ferroptosis in severe lung pathologies, including both ALI and non-ALI. Ferroptosis markers, including transferrin receptor 1 (TfR1) and malondialdehyde adduct (MDA), were increased in human COVID-19 lung tissue, reflecting increased lipid peroxidation and ferroptosis. In comparison, markers for other forms of cell death, including apoptosis, necroptosis, and pyroptosis, were not elevated.
Since ferroptosis and lung function are regulated by lipids, we analyzed lipid profile of COVID-19 lung tissues and found significant alterations in lipid composition in comparison to normal lung. We found depletion of phospholipids containing polyunsaturated fatty acyl tails (PL-PUFAs) and accumulation of lysophospholipids (lysoPLs). PL-PUFAs are prone to oxidation, resulting in the production of lipid hydroperoxides. This process contributes to their depletion during ferroptosis6. Subsequently, the oxidized PUFA tails are cleaved by phospholipases, resulting in the buildup of lysoPLs. This lipid change pattern is a hallmark of ferroptosis and provides evidence of ferroptosis occurrence in the lungs of COVID-19 patients.
We investigated the driver of ferroptosis using a COVID-19 hamster model and a primary lung cell model. Our analysis of patient samples suggested dysregulation in iron homeostasis (e.g. increased serum ferritin, and upregulated TfR1 and ferritin light chain in infected lung tissue). We established an iron-overload model by treating primary lung epithelial cells with ferric ammonium citrate (FAC). FAC treatment induced FTL expression, lipid peroxidation accumulation, and sensitized lung cells to ferroptosis.
To establish a COVID-19 disease model, we collaborated with the Institute of Comparative Medicine at Columbia University Irving Medical Center and the Miorin group at Icahn School of Medicine at Mount Sinai. We found that Syrian hamsters infected with wild-type SARS-CoV-2 developed lung manifestations that recapitulate human lung pathologies. In assessing ferroptosis in infected lungs, we observed a positive correlation between TfR1 expression and lung injury severity throughout tissues. Additionally, levels of the lipid peroxidation peaked on day seven post-infection, accompanied by elevated ferritin light chain (FTL) expression. Hamsters treated with ferroptosis inhibitors liproxstatin-1 or the ferrostatin-1 analog Th-2-31 showed partially reduced lung injury. However, the effectiveness of these inhibitors was hindered by insufficient drug accumulation, biodistribution, and stability. These findings nonetheless underscore the association between ferroptosis and COVID-19 lung pathology.
Overall, we found a critical role for ferroptosis, driven by dysregulated iron metabolism, in severe COVID-19 lung pathology. Notably, the heterogeneity in gene expression and ferroptosis response among different alveolar cell types, such as alveolar macrophages, fibroblasts, and epithelial cells, suggests a complex interplay of iron metabolism and cell-specific responses. Investigating these varied responses may lead to treatment strategies tailored to specific cell populations.
Additionally, our study points to the promising therapeutic potential of ferroptosis inhibitors in mitigating COVID-19-related lung injury. By targeting key players in the ferroptosis pathway, compounds might be developed to reduce lung damage and improve patient recovery. Moreover, exploring the synergy between ferroptosis-targeted therapies and existing COVID-19 treatments can inform combination strategies that address multiple aspects of the disease. Long-term studies on the effects of ferroptosis modulation will provide insights into the sustained impact on lung function and overall recovery. Furthermore, the identification of ferroptosis protein and lipid markers as diagnostic tools offers promise for non-invasive, timely assessments of COVID-19 severity, enhancing clinical decision-making and patient management.
Reference
1 Cardinal-Fernandez, P., Lorente, J. A., Ballen-Barragan, A. & Matute-Bello, G. Acute respiratory distress syndrome and diffuse alveolar damage. New insights on a complex relationship. Ann Am Thorac Soc 14, 844-850, doi:10.1513/AnnalsATS.201609-728PS (2017).
2 De Michele, S. et al. Forty postmortem examinations in COVID-19 patients. Am J Clin Pathol 154, 748-760, doi:10.1093/ajcp/aqaa156 (2020).
3 Martines, R. B. et al. Pathology and pathogenesis of SARS-CoV-2 associated with fatal coronavirus disease, United States. Emerging Infectious Diseases 26, 2005-2015, doi:10.3201/eid2609.202095 (2020).
4 Eastman, R. T. et al. Remdesivir: A Review of Its Discovery and Development Leading to Emergency Use Authorization for Treatment of COVID-19. ACS Cent Sci 6, 672-683, doi:10.1021/acscentsci.0c00489 (2020).
5 Leligdowicz, A., Hardin, C. C., Harhay, M. O. & Calfee, C. S. Immune modulation in sepsis, ARDS, and Covid-19—the road traveled and the road ahead. NEJM Evidence 1, EVIDra2200118, doi:10.1056/EVIDra2200118 (2022).
6 Yang, W. S. & Stockwell, B. R. Ferroptosis: death by lipid peroxidation. Trends in Cell Biology 26, 165-176 (2016).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in