Blocking cancer stem cell functions in colon tumors using a drug initially developed to treat cocaine addiction
Published in Cancer and Pharmacy & Pharmacology
Our group previously reported G9a as an essential epigenetic regulator maintaining pluripotent-like gene signatures and CSC-associated functions in colorectal cancer 1. Such a role for G9a is certainly not exclusive to colorectal cancer, as highlighted in our review article covering the latest findings on this chromatin writer in different types of tumors 2. Despite the development of several small molecule inhibitors directly blocking G9a histone methyltransferase activity, including BIX-01294, UNC0642, and A-366, no clinical trials and very few in vivo experiments using these compounds were reported. This void is likely due to the harmful impact of systemic, untargeted alteration of G9a activity, which is essential to the integrity of healthy differentiated tissues 2,3. To cite the Nobel Prize awardee Sir James Black, “The most fruitful basis for the discovery of a new drug is to start with an old drug.” Thus, we applied the concept of drug repurposing to identify “clinically safe” compounds that could serve as context-specific blockers of G9a activity in colorectal cancer. As a postdoc, Yannick was mentored by Dr. Mickie Bhatia at McMaster University, who developed a phenotypic drug screening method involving a transformed variant of human embryonic stem cells 4. In our adaptation of this assay, we quantified variations in CSC hallmarks, such as the pluripotency marker Oct4 and the main epigenetic mark catalyzed by G9 (H3K9me2), using high-content imaging following drug treatments.
This target-agnostic approach identified the dopamine transporter antagonist vanoxerine as a potent inhibitor of cancer stem cell (CSC) functions. In vivo serial tumor transplantation, patient-derived clonogenic organoid formation, and lineage-tracing experiments validated the suppression of CSCs by vanoxerine in colorectal cancer. Molecular experiment in human colon cancer cells revealed that vanoxerine blocks the expression of G9a (EHMT2) by downregulating Akt signaling, which, in turn, reduces the recruitment of the transcription factor Nur77 at the EHMT2 promoter. A similar mechanism for G9a inhibition, relying on Nur77 processing, was independently reported in rhabdomyosarcoma cells treated with flavonoids like quercetin and kaempferol 5.
Vanoxerine is a dopamine reuptake inhibitor that was initially developed to treat cocaine addictions. It has a slower dissociation rate and superior affinity for the dopamine transporter (DAT) compared to cocaine itself 6. Vanoxerine was deemed safe for human patients through different clinical trials outside the field of oncology 6-8. Such toxicology findings align with the absence of notable effects on normal human colonic organoids and mouse healthy tissue integrity observed in our preclinical work upon vanoxerine treatments.
Following the identification of vanoxerine, we were puzzled by having a DAT inhibitor as a frontrunner candidate. One big question was whether vanoxerine affected G9a expression and CSC functions by engaging its primary target. After all, phenotypic-based drug discovery can become very challenging at the target deconvolution stage and through deciphering the mechanism of action for a candidate 9. Still, we were aware of a study from the Nestler lab from 2010, reporting that cocaine consumption represses G9a expression in the nucleus accumbens and leads to behavioral plasticity 10. Considering we were also observing important epigenetic and transcriptional re-wiring in cells treated with vanoxerine, we started looking for the presence of DAT in colorectal tumors. We used different approaches to confirm the presence of DAT mRNA (SLC6A3) and protein in colon cancer cell lines and primary patient samples. Instead of a plasma membrane distribution, immunostaining revealed a DAT distribution pattern within intracellular vesicles, just like what was reported in the Human Protein Atlas for another type of human cancer cells (U2OS). Moreover, single-cell protein analysis showed that ~5% of the cells from primary colon adenocarcinoma were positive for DAT. Just for a sanity check, Chris searched the literature for other instances where the dopamine transporter is expressed in the context of colorectal cancer (Figure 1). Different studies consistently detected DAT mRNA (SLC6A3) in colon cancer cell lines, patient tumors, metastases, and organoid systems.
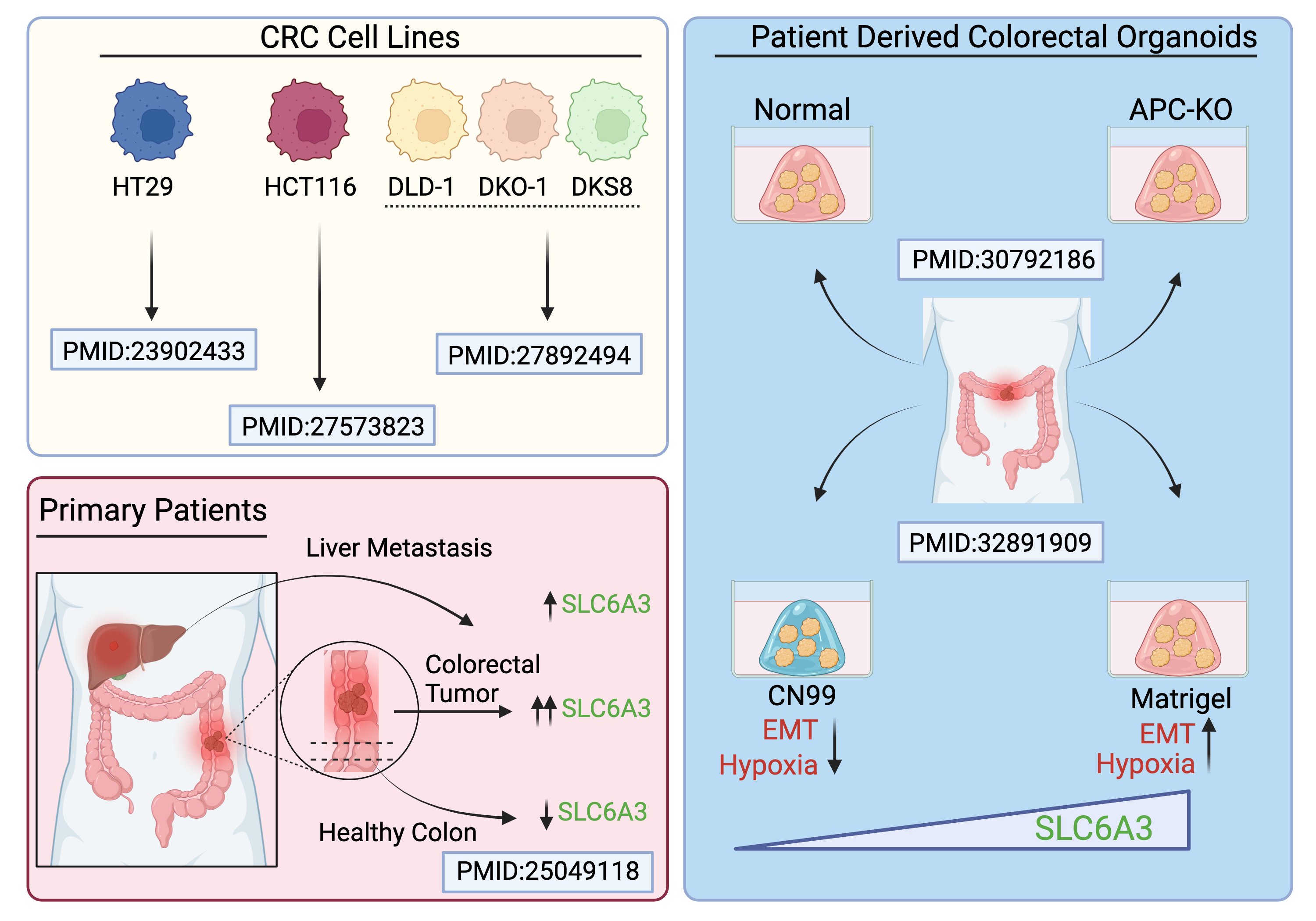
Figure 1: Summary of SLC6A3 expression in human colon cancer cell lines, tumor tissues, and colonic organoid systems.
As surprising as finding DAT in colon tumors may sound, we must remember that other dopamine signaling components were observed in different cancers and associated with CSC functions 4,11-13. By knocking down SLC6A3 in colon cancer cells, we recapitulated the loss of self-renewal and tumor-initiating capacities we observed upon vanoxerine treatment. Integrative transcriptomic analyses also supported correlated alterations in pluripotency networks and genes typically upregulated in colorectal tumors in DAT knockdown, vanoxerine-treated, and BIX-01294-treated cells. This reassured us of the relevance of DAT in response to vanoxerine and, overall, supported the fact that our phenotypic drug discovery approach identified an unanticipated molecular axis in colorectal tumors.
In a previous study, we observed that G9a inhibition was associated with increased expression of endogenous transposable elements such as the Long Interspersed Nuclear Element-1 (LINE-1) and Human Endogenous Retrovirus (hERV) families in transformed embryonic stem cells 1. These ancient viral DNA fragments integrated into our genome throughout evolution are usually kept silent by epigenetic marks, including 5-methyl cytosine and histone lysine methylation in healthy somatic tissues 14. In cancer, the “reactivation” of these elements, e.g., following treatments with a demethylating agent, increases transposable element transcripts, stimulating the antiviral innate immune response 14. In two recent studies, altering H3K9me2 levels via G9a inhibition or blockade of SETDB1 chromatin recruitment enhanced anti-tumor immunity in melanoma 15,16. The de-repression of transposable elements was found at the center of such a restoration of immune susceptibility 15. Accordingly, both vanoxerine treatments and DAT knockdown increased the expression of transposable elements in colorectal cancer cells. We also observed a substantial increase in tumor-infiltrating lymphocytes in colorectal tumors treated with vanoxerine in vivo. Overall, these observations support that our drug candidate is restoring anti-tumor immune susceptibility, which is quite significant considering that colorectal cancers tend to show poor response to standard immunotherapy. Considering our results in colorectal cancer and the other studies in melanoma, we can expect this phenomenon to occur in different types of tumors following G9a inhibition.
Our target-agnostic approach that led to identifying vanoxerine as a new CSC-targeting agent revealed the unanticipated presence of DAT in the most aggressive cells within colorectal tumors. Why is this transporter being there, and what is it doing exactly? Although we depicted the presence of DAT in human primary colon tumors, we feel we only scratched the surface so far. We don’t know much yet about DAT trafficking or the mechanism linking it to Akt in tumor cells. Also, we do not fully understand the transcriptional regulation of SLC6A3 expression in this context. Although dopamine itself did not appear crucial to vanoxerine’s effect on CSC functions, we cannot rule out its implication in potential pro-oncogenic functions of DAT in colon tumors. Hence, we want to understand everything about DAT in cancer stemness, which will keep us busy for the years to come. But we also hope that other groups around the community, via their expertise, will shed light on the fundamental role of this transporter in cancer and help design new treatments benefiting patients.
References:
1 Bergin, C. J. et al. G9a controls pluripotent-like identity and tumor-initiating function in human colorectal cancer. Oncogene 40, 1191-1202 (2021). https://doi.org/10.1038/s41388-020-01591-7
2 Haebe, J. R., Bergin, C. J., Sandouka, T. & Benoit, Y. D. Emerging role of G9a in cancer stemness and promises as a therapeutic target. Oncogenesis 10, 76 (2021). https://doi.org/10.1038/s41389-021-00370-7
3 Padeken, J., Methot, S. P. & Gasser, S. M. Establishment of H3K9-methylated heterochromatin and its functions in tissue differentiation and maintenance. Nat Rev Mol Cell Biol 23, 623-640 (2022). https://doi.org/10.1038/s41580-022-00483-w
4 Sachlos, E. et al. Identification of drugs including a dopamine receptor antagonist that selectively target cancer stem cells. Cell 149, 1284-1297 (2012). https://doi.org/10.1016/j.cell.2012.03.049
5 Shrestha, R. et al. Flavonoids kaempferol and quercetin are nuclear receptor 4A1 (NR4A1, Nur77) ligands and inhibit rhabdomyosarcoma cell and tumor growth. J Exp Clin Cancer Res 40, 392 (2021). https://doi.org/10.1186/s13046-021-02199-9
6 Preti, A. Vanoxerine National Institute on Drug Abuse. Curr Opin Investig Drugs 1, 241-251 (2000).
7 Piccini, J. P. et al. Randomized, double-blind, placebo-controlled study to evaluate the safety and efficacy of a single oral dose of vanoxerine for the conversion of subjects with recent onset atrial fibrillation or flutter to normal sinus rhythm: RESTORE SR. Heart Rhythm 13, 1777-1783 (2016). https://doi.org/10.1016/j.hrthm.2016.04.012
8 Kadric, S., Mohler, H., Kallioniemi, O. & Altmann, K. H. A Multicenter, Randomized, Placebo-Controlled Study to Evaluate the Efficacy and Safety of Long-Acting Injectable Formulation of Vanoxerine (Vanoxerine Consta 394.2 mg) for Cocaine Relapse Prevention. World Journal of Neuroscience 9, 113-137 (2019). https://doi.org/10.4236/WJNS.2019.93008
9 Vincent, F. et al. Phenotypic drug discovery: recent successes, lessons learned and new directions. Nat Rev Drug Discov (2022). https://doi.org/10.1038/s41573-022-00472-w
10 Maze, I. et al. Essential role of the histone methyltransferase G9a in cocaine-induced plasticity. Science 327, 213-216 (2010). https://doi.org/10.1126/science.1179438
11 Aslostovar, L. et al. Abnormal dopamine receptor signaling allows selective therapeutic targeting of neoplastic progenitors in AML patients. Cell Rep Med 2, 100202 (2021). https://doi.org/10.1016/j.xcrm.2021.100202
12 Jandaghi, P. et al. Expression of DRD2 Is Increased in Human Pancreatic Ductal Adenocarcinoma and Inhibitors Slow Tumor Growth in Mice. Gastroenterology 151, 1218-1231 (2016). https://doi.org/10.1053/j.gastro.2016.08.040
13 Borcherding, D. C. et al. Expression and therapeutic targeting of dopamine receptor-1 (D1R) in breast cancer. Oncogene 35, 3103-3113 (2016). https://doi.org/10.1038/onc.2015.369
14 Bergin, C. J., Mendes da Silva, A. & Benoit, Y. D. Where to Draw the LINE-Are Retrotransposable Elements Here to Stay? Cancers (Basel) 15 (2023). https://doi.org/10.3390/cancers15164119
15 Zhang, S. M. et al. KDM5B promotes immune evasion by recruiting SETDB1 to silence retroelements. Nature 598, 682-687 (2021). https://doi.org/10.1038/s41586-021-03994-2
16 Kato, S. et al. Gain-of-function genetic alterations of G9a drive oncogenesis. Cancer Discov (2020). https://doi.org/10.1158/2159-8290.CD-19-0532
Follow the Topic
-
Nature Cancer
This journal aims to provide a unique forum through which the cancer community will learn about the latest, most significant cancer-related advances across the life, physical, applied and social sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Cancer Neuroscience: from mechanisms to therapy
Publishing Model: Hybrid
Deadline: Jan 30, 2027


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in