Buyer’s remorse: how OAS1 genetic variants acquired by modern humans make them vulnerable to severe COVID-19
Published in Microbiology
Our lab explores molecular mechanisms of genetic associations, and some emerging results from genome-wide association studies (GWAS) for SARS-CoV-2 infection caught our attention. The signal within the 12q24.13 region was particularly intriguing as this genomic locus encodes a family of OAS proteins, with substantial literature on its role in immunity. These results were primarily based on the genetic association of a single nucleotide polymorphism (SNP) rs10774671, which affects the splice acceptor for the last exon of OAS1. So, we started our journey into exploring molecular mechanisms of this genetic association, expecting that the same SNP could account for the association both with SARS-CoV-2 infection and disease severity. Indeed, through genetic, bioinformatic and functional studies, we concluded that rs10774671 is one of the functional variants responsible for this association. However, another variant, rs1131454, located within exon 3 of OAS1, is also important. Both these variants affect nonsense-mediated decay (NMD) of OAS1 and thus control how much of OAS1 protein is produced. In turn, OAS1 protein would sense and activate the mechanism resulting in the degradation of intracellular RNA, and specifically of RNA viruses, such as SARS-CoV-2. Our findings also help to identify individuals carrying the risk alleles of the two OAS1 SNPs who would most benefit from early treatment with interferons to prevent progression to COVID-19 that is severe enough to require hospitalization.
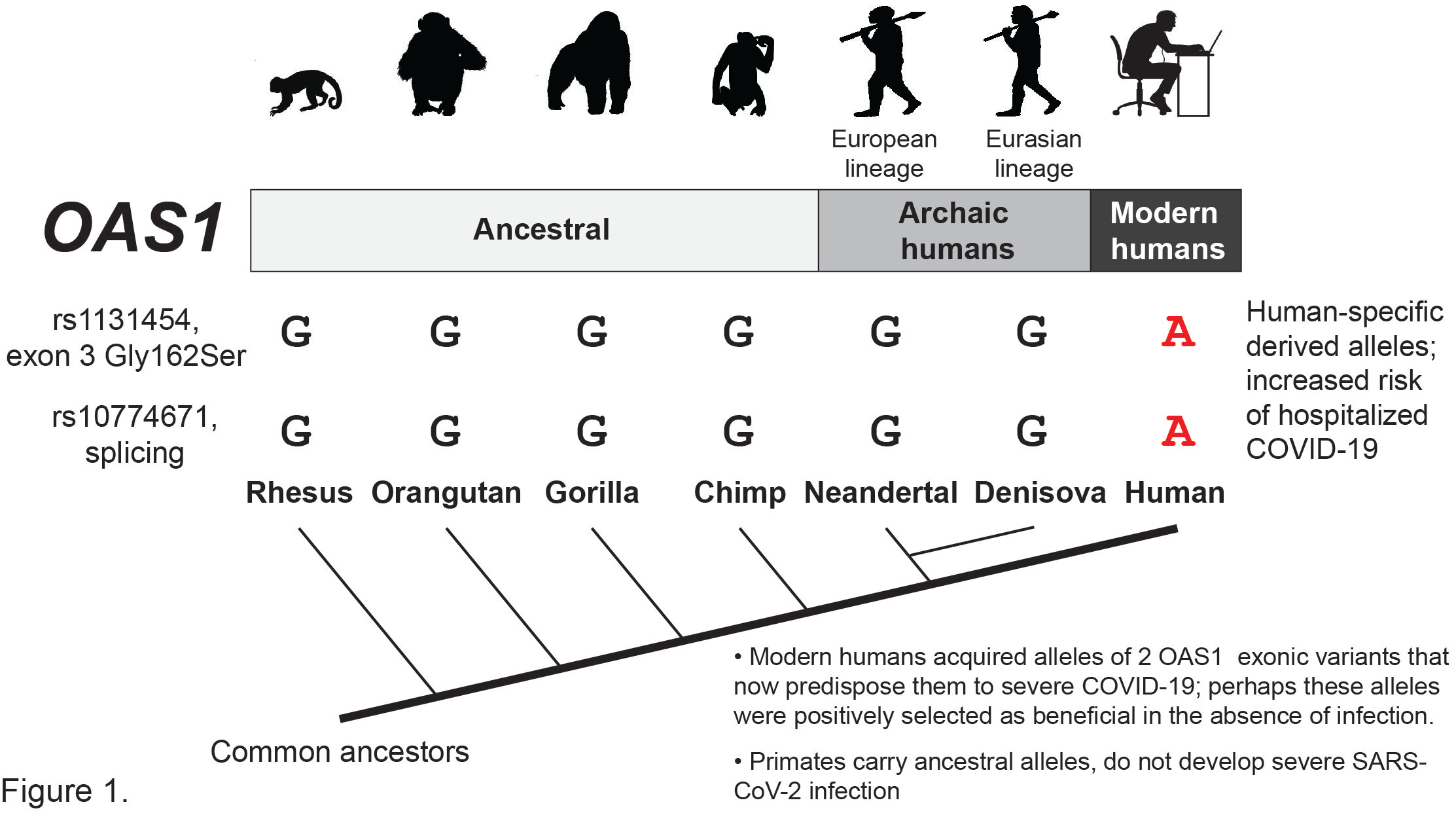
Notably, the risk alleles of both variants, rs10774671-A and rs1131454-A, emerged only in modern humans (Figure 1) but became very common. Specifically, in populations of the 1000 Genomes Project, rs10774671-A has 65-75% allele frequencies in non-Africans and 36% in Africans, and rs131454-A has 57-63% in non-Africans and 15% in Africans.
It is unclear why the derived alleles that now increase the risk of severe COVID-19 were selected to be retained in human populations at such high frequency. It has been suggested that genomic fragment inherited from Neandertals and represented by rs10774671-G allele protects from SARS-CoV-2 infection and severe COVID-19. But it looks like protection against severe COVID-19 could be provided by any ancestral or archaic human (Neandertal or Denisova) version of OAS1 (Figure 1). Why did the more degradable version of OAS1 that carries the risk alleles and impairs the immune response to SARS-CoV-2 become the dominant version in modern humans? Perhaps this OAS1 version provided more important benefits in the absence of infection that outweighed its inferiority at the time of infection? The degradation through the NMD pathway could provide a more fine-tuned regulation of the OAS1 activity, which results in the elimination of intracellular RNA, both of the pathogens and the host, and likely cell death. If not tightly and timely regulated, this function could be very damaging, thus favoring the selection of genetic variants acquired by modern humans to produce the more controllable version of OAS1. However, it feels like a “buyer’s remorse” at the time of the SARS-CoV-2 pandemic when having the ancestral or archaic version of OAS1 appears to protect us from severe COVID-19 better than the derived version produced by the majority of the population. https://www.nature.com/articles/s41588-022-01113-z
Follow the Topic
-
Nature Genetics
This journal publishes the very highest quality research in genetics, encompassing genetic and functional genomic studies on human and plant traits and on other model organisms.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in