Glioblastomas are a very aggressive type of tumor that grows exclusively in the brain. When a patient is diagnosed in the clinic, tumors have often already grown extensively and aggressively, making this a difficult to treat type of cancer with fatal outcome. When T cell immunotherapy and especially immune-checkpoint blockade proved successful in many cancer types, it provided a new hope for glioblastoma as well. Unfortunately, the hope was short lived, as the results of clinical studies1 (CheckMate 498, CheckMate 548) were disappointing. That was the start of our project: together with clinical oncologist Gerben Borst, we set out to understand why immunotherapy did not improve glioblastoma therapeutic response to standard of care treatment.
The dynamic and transient changes in response to treatment are key
We performed an extensive analysis of the changes in immune cells that infiltrate into the brain tumor in response to treatment. Mice which had developed genetically-engineered brain tumors were exposed to similar treatment regimen as those used in clinical trials in patients. We found that radiotherapy increases the number of T cells by tenfold, but that the increase was transient and the number of T cells quickly decreased again. A low T cell infiltration is considered one of the constraints to achieve effective immunotherapeutic results, so we hypothesized that this peak of T cells may provide a window of opportunity for therapeutic response. We decided to adjust the treatment schedule and started immunotherapy at the peak of T cell infiltration, just after radiotherapy, and found that indeed this yielded a (modest) improvement of survival. Our first conclusion was that since this adjuvant dosing of immunotherapy after radiotherapy has not been tried yet in patients, it may be a way forward in future clinical trials.
We next decided to dig deeper into the immune response post radio-immunotherapy to improve therapeutic outcome, which once again showed that the dynamic changes of immune cell content and phenotype post therapy was crucial. Using 24-color flow cytometry analyses and unbiased bioinformatic clustering of immune cells in the tumor microenvironment, we identified that a specific population of CD103+ regulatory T cells (Treg) increased in response to immunotherapy (Fig 1, population 1). The increase of these cells closely followed after the start of immunotherapy: if we started immunotherapy later, these CD103+ Tregs would peak at a later time as well. We further characterized these CD103+ Tregs and revealed their highly immunosuppressive features in comparison with other Tregs, which was associated with a high activity of lipid biosynthetic processes, a known indication of immunosuppressive activity2.
.jpg)
|
Figure 1: unbiased population analysis of regulatory T cells in the tumor microenvironment. (a) unbiased clustering of regulatory T cells results in 5 main populations present in all treatment regimens combined (b) density plots of regulatory T cells show that population 2 (in red) is the predominant population of Tregs after radiotherapy (RT) treatment, while population 1 (in orange) is the predominant population during combined radiotherapy and immunotherapy (RT+Conc.IT). |
Connecting the dots
The dynamic response of CD103+ Tregs post radio-immunotherapy combined with their immunosuppressive capacity and gene expression profile suggested these cells may play a functional role in suppressing an effector T cell response. Fortunately, we were able to partner with Dr. Sergio Quezada from the UCL Cancer Institute in London who had developed an antibody efficiently targeting and depleting Tregs. When we added this Treg depleting antibody to a radio-immunotherapy regimen, a subset of mice achieved long-term survival benefit with complete tumor regression (Fig 2a). We confirmed this effect was mediated by activation of effector CD8 T cells by depleting these cells, which completely reversed CD25-mediated survival benefit (Fig. 2b). By depleting these specific immune cells and studying the therapeutic response we showed that CD8 T cells can mount effective anti-tumor responses in glioblastoma, and that this response can be completely suppressed by the presence of regulatory T cells.
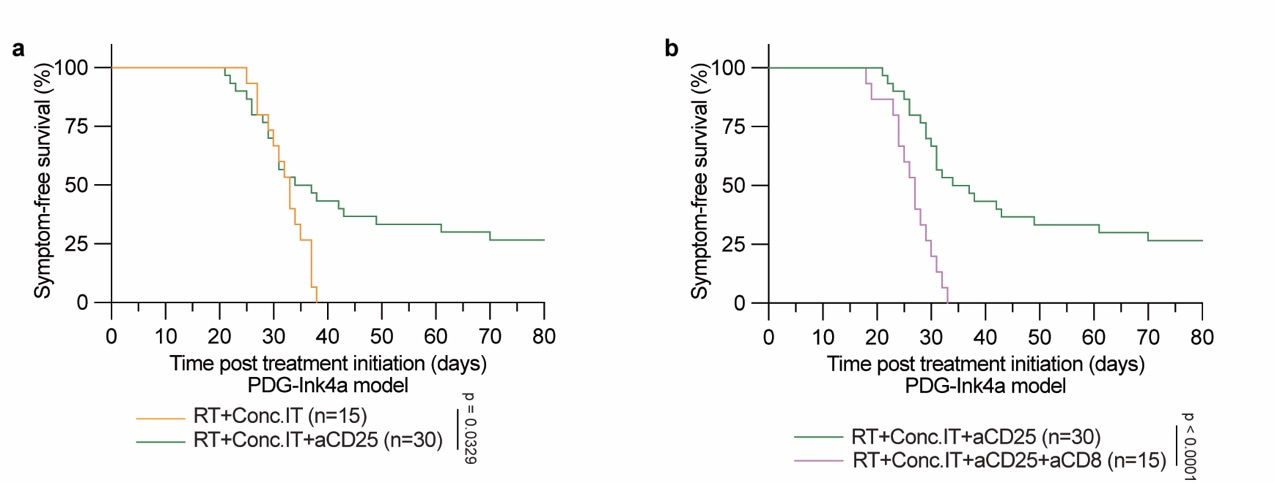
|
Figure 2: symptom-free survival of mice treated with combinations of radiotherapy and immunotherapy. (a) Mice treated with radiotherapy and combined immunotherapy (RT+Conc.IT) compared with mice treated with the same regimen and simultaneous depletion of regulatory T cells (RT+Conc.IT+aCD25). (b) Mice treated with radiotherapy, combined immunotherapy and Treg depletion (RT+Conc.IT+aCD25) compared with mice treated with the same regimen and simultaneous depletion of CD8+ T cells (RT+Conc.IT+aCD25+aCD8). |
Lessons learned and moving forward
One challenge in the quest for finding better treatments for glioblastomas is that the tumors are – by nature of growing in the brain – very hard to reach therapeutically and to analyze in translational research. Our results show this is a major obstacle, as our key results were transient changes in the early immune response to treatment, which would not be possible to analyze in patients. Our study underscores that research using mouse models is fundamental to understanding the complex immune response to different treatment strategies and remain critical for knowledge-guided design of future clinical trials.
Although our results provide hope for T cell-centric therapeutic approaches in what is considered a highly immunosuppressive type of tumor, efforts must continue. Indeed, our approach of targeting CD25+ cells did not deplete all Tregs, including a CD103+CD25- subset which was increased in response to immunotherapy and may be functionally important. Further work will need to focus on developing multiple ways of targeting immunosuppressive Tregs, taking into account the dynamic changes these cells may undergo in response to therapy.
References
- Reardon, D. A. et al. Effect of nivolumab vs bevacizumab in patients with recurrent glioblastoma: the CheckMate 143 phase 3 randomized clinical trial. JAMA Oncol 6, 1003–1010 (2020).
- Lim et al Lim, S. A. et al. Lipid signalling enforces functional specialization of Treg cells in tumours. Nature 591, 306–311 (2021).
Follow the Topic
-
Nature Cancer
This journal aims to provide a unique forum through which the cancer community will learn about the latest, most significant cancer-related advances across the life, physical, applied and social sciences.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in