Click Processes Involving Nitriles and Allenes are Orthogonal to CuAAC and SuFEx and Generate Fluorescent Linkages
Published in Chemistry
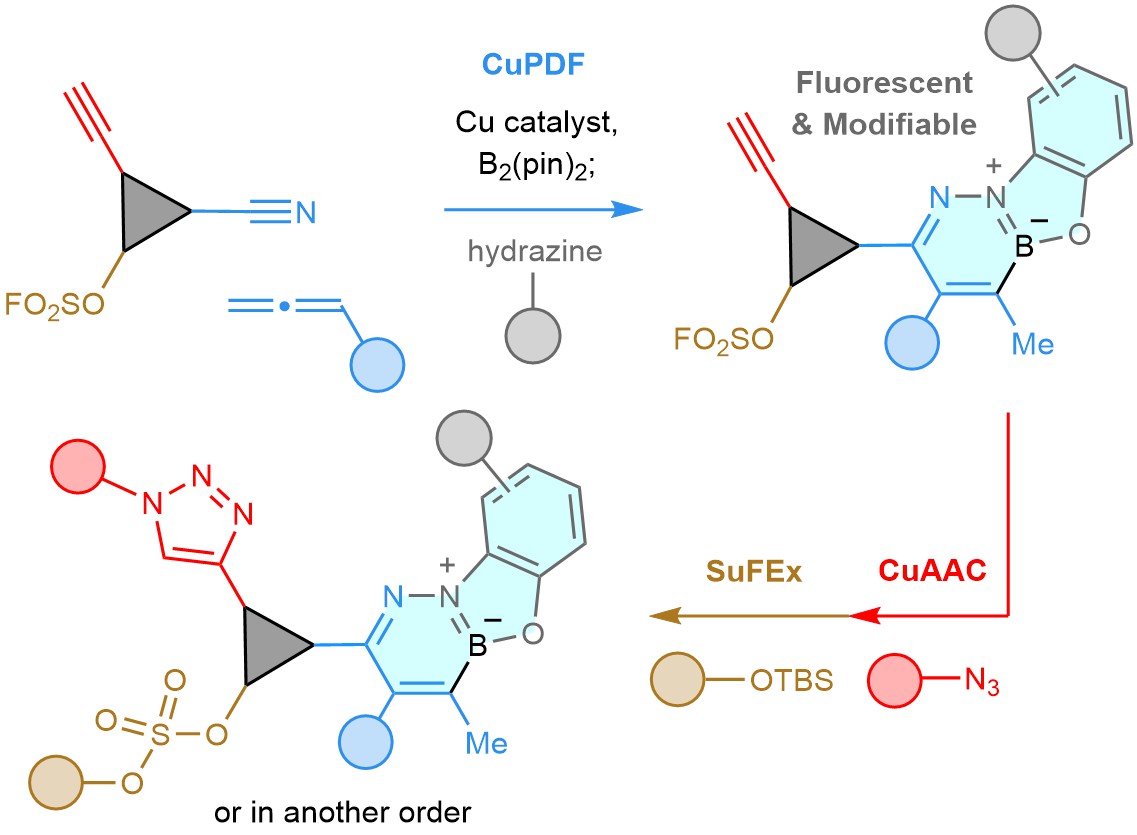
Several years ago, we wanted to see whether we could tap into our expertise in catalysis and make a meaningful contribution to click chemistry1. A reaction we had developed at the time was a copper-catalysed enantioselective reaction (Fig. 1a) that merged a nitrile, an allene, bis(pinacolato)diborane (B2(pin)2) and a silyl hydride (PMHS), that affords homoallylic amines with high diastereo- and enantioselectivity (II via I)2,3. This was a rare case of a catalytic C–C bond generation involving relatively inert nitriles (for example, acetonitrile is a commonly used solvent). We thought that α,β-unsaturated ketimine III, which we had worked hard to avoid, could be viewed as a modifiable linkage. Could it be then that nitriles and allenes are undiscovered clickable pairs, similar to an alkyne and an azide, partners in the legendary copper-catalysed azide alkyne cycloaddition (CuAAC)4-5?
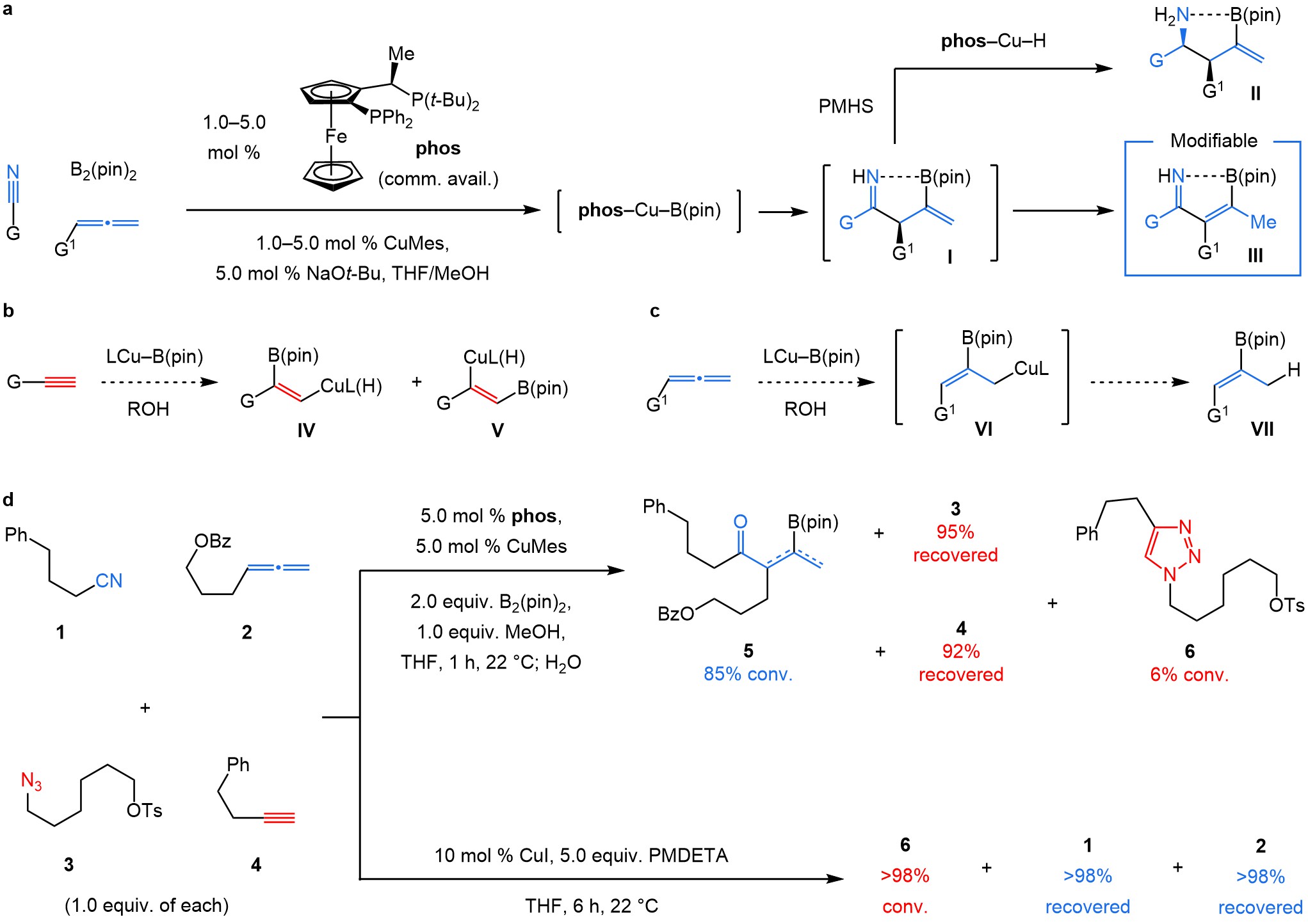
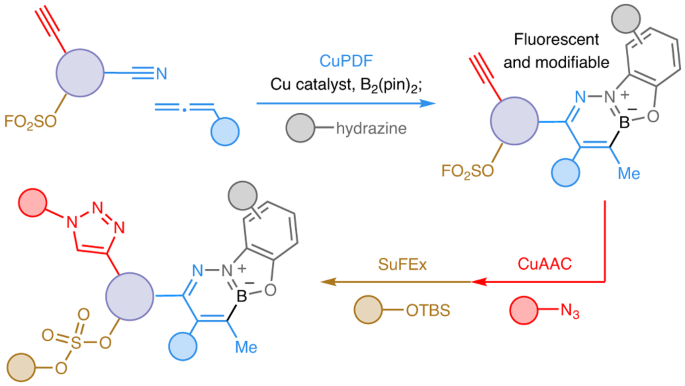
If the process were to be orthogonal to CuAAC, the two could be used sequentially without needing extra protection/deprotection steps, a requirement that can be particularly problematic in oligomer/polymer synthesis or preparation of acid- or base-sensitive organic molecules . But would two transformations, both Cu-catalysed, be orthogonal? For several reasons we thought this was a dicey proposition. LCu–B(pin) (L, a ligand) could easily react with an alkyne instead of an allene, affording IV and/or V (Fig. 1b)6, and many Cu complexes can promote CuAAC. Moreover, the in situ-generated allyl–Cu VI (Fig. 1c) could react with the alcohol, which is needed for catalyst release (converting C=NCuL to C=NH), affording VII. It was easy to find out and we had nothing to lose. When we submitted equimolar amounts of 1–4 to our reaction conditions (Fig. 1d), 5 (formed by ketimine hydrolysis) was formed and there was hardly any triazole (6) detectable. Conversely, the allene and nitrile remained intact under standard CuAAC conditions. Miraculously, we were in business!
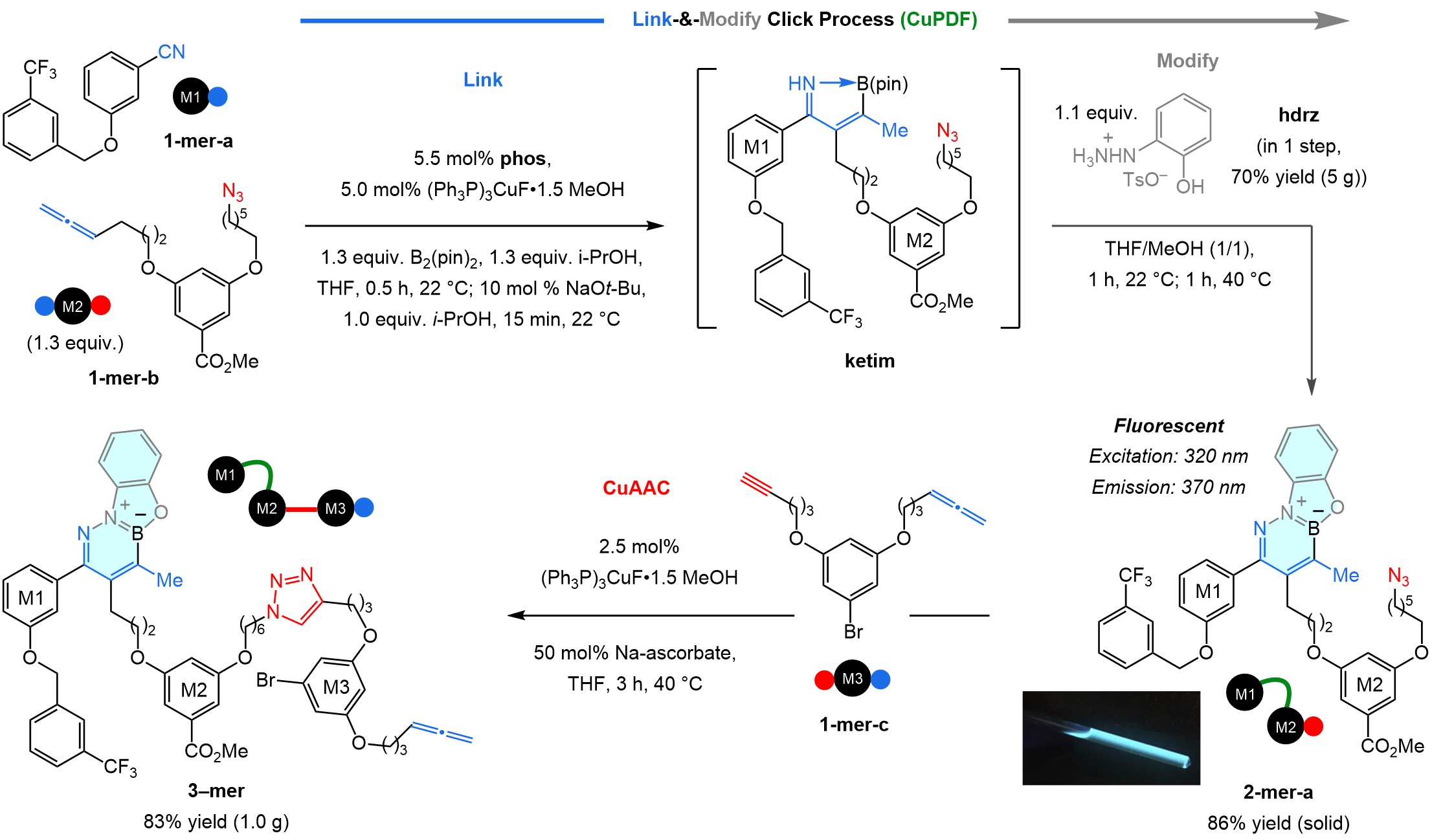
Looking for ways to modify the β-boryl ketimine, we were drawn to studies reported earlier by Gillingham7, a former group member, and his team in Basel. They had shown that diazaborinine rings can be obtained by reacting a hydrazine and an aldehyde with a nearby aryl–B(pin). There was a dazzling bonus to boot: the heterocyclic products were fluorescent. It did not take us long to find that linking 1-mer-a and 1-mer-b under conditions that ensures a priori alkene isomerization and formation of ketim (Fig. 2), followed by addition of hdrz delivered fluorescent diazaborinine (2-mer-a, 86% yield). We call this link-and-modify process CuPDF, short for Cu-catalysed phenoxydiazaborinine formation. The ensuing CuAAC involving 1-mer-c gave 3-mer.
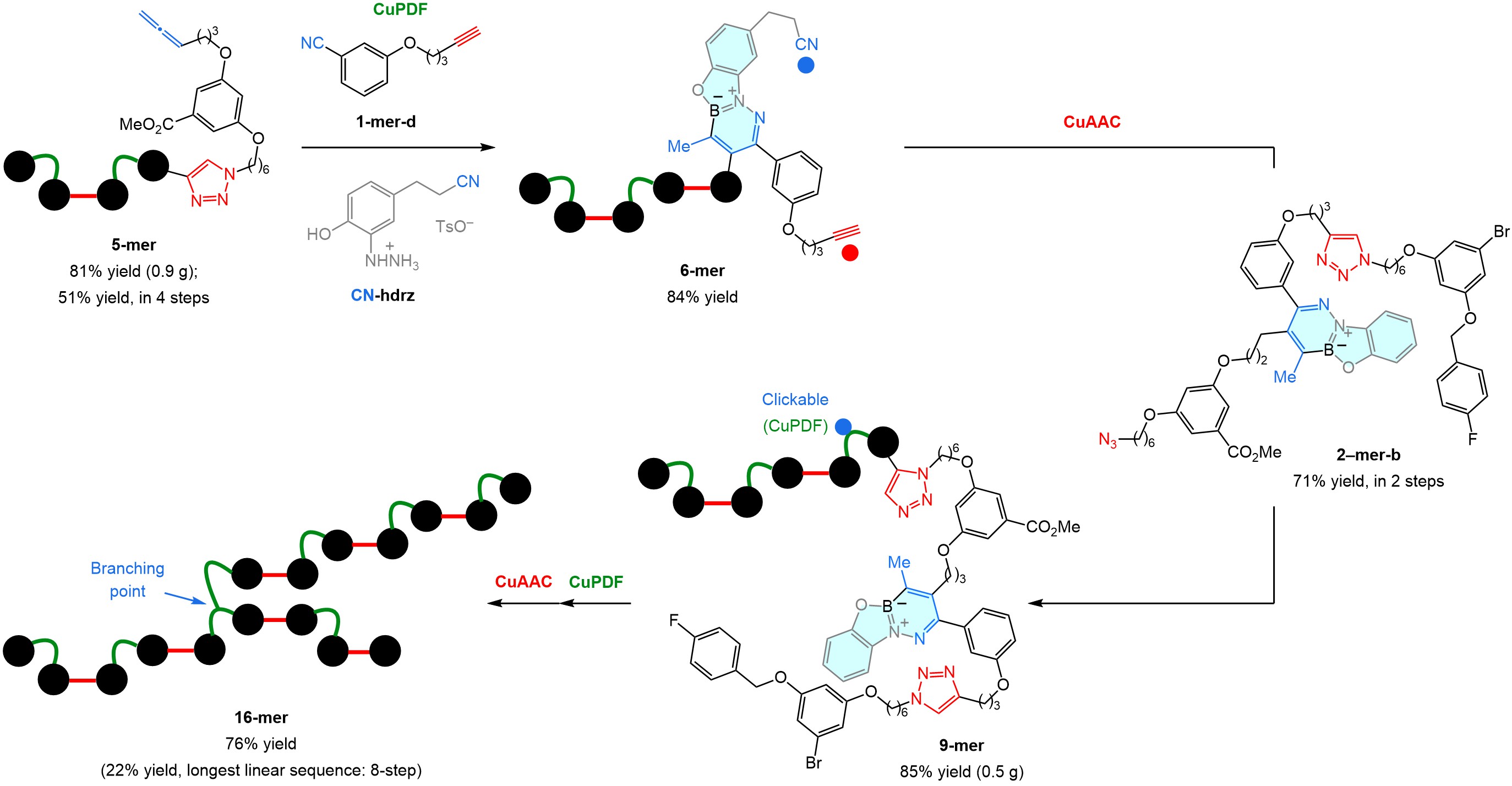
There are several advantages to a modifiable linker, one being that the hydrazine used may have a nitrile of its own. We probed this by using CN-hdrz to convert 5-mer to 6-mer (Fig. 3). After a CuAAC involving 2-mer-a, which could be prepared in parallel, we accessed 9-mer, a sequence-defined oligomer, equipped with an alterable monomer, making site-specific branching feasible. This was realized through a CuPDF/CuAAC sequence that delivered 16-mer.
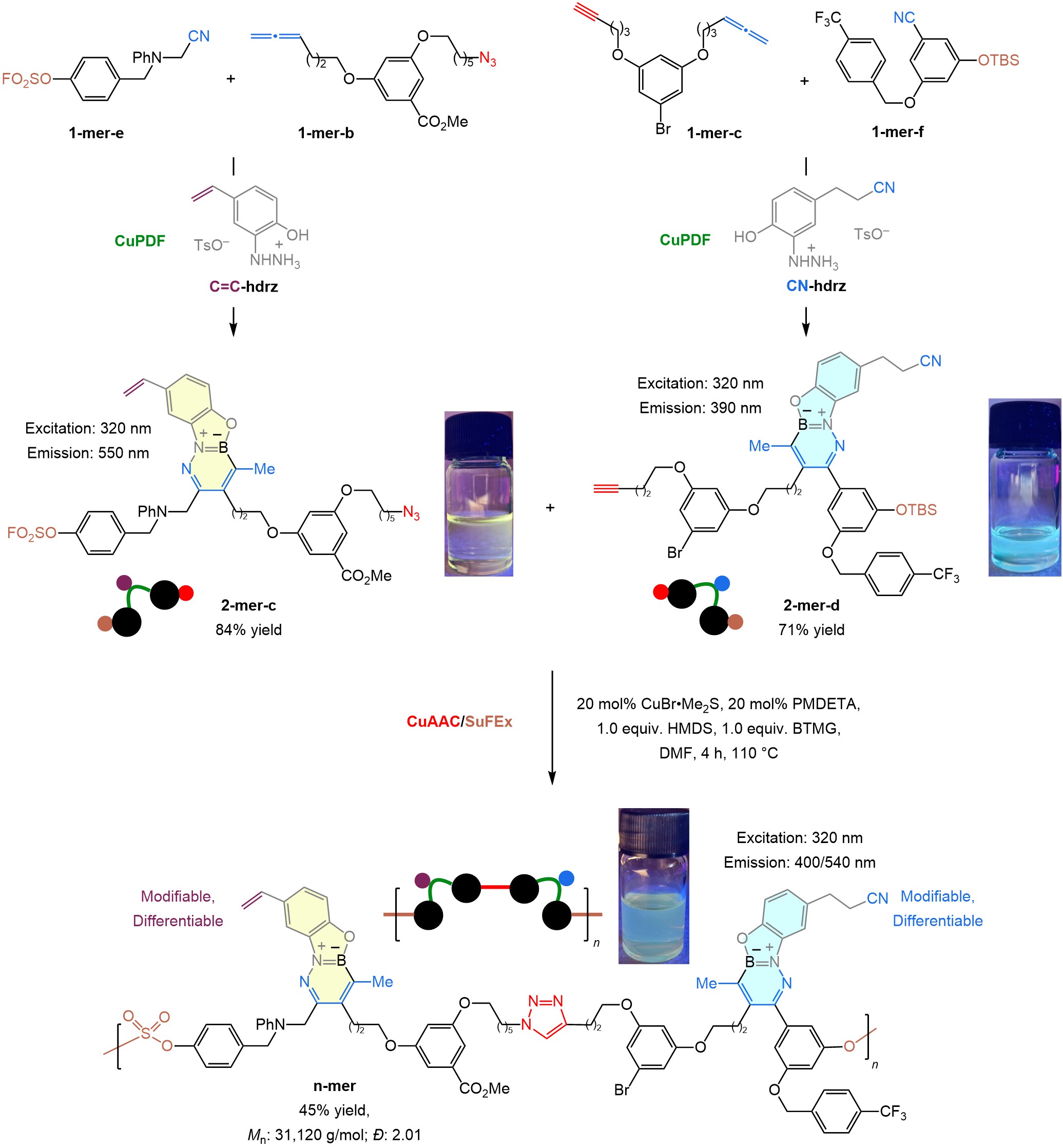
CuPDF is similarly orthogonal to SuFEx (sulfur-fluoride exchange)8, another widely used click method. The importance of having three catalytic and orthogonal click approaches can be seen in the context of the concise synthesis of a chemoselectively modifiable polymer (Fig. 4). Two pairs of monomers, 1-mer-b/1-mer-e and 1-mer-c/1-mer-f, each with two clickable units were linked with C=C-hdrz and CN-hdrz, affording two complementary dimers: 2-mer-c and 2-mer-d. The resulting dimers were simultaneously exposed to CuAAC and SuFEx conditions, resulting in the formation of a fluorescent and site-specifically modifiable polydisperse polymer (n-mer)9 (Fig. 4).
Next, we synthesised various multi-drug conjugates10. A cyano-substituted derivative of anti-cancer agent cilengitide11 (CN-Clng, Fig. 5a) was linked to several drugs, affording products such as Clng-Drug (with anti-inflammatory zaltoprofen). We leveraged the orthogonality of CuPDF/CuAAC and prepared Pent-Drug2 (Fig. 5b), consisting of cancer diagnostic agent pentagastrin, anti-bacterial sulbactam and breast cancer drug estradiol. It is noteworthy that CuPDF connects a drug to a host molecule while also imparting fluorescence, obviating the need for separate sets of transformations for accomplishing each task.
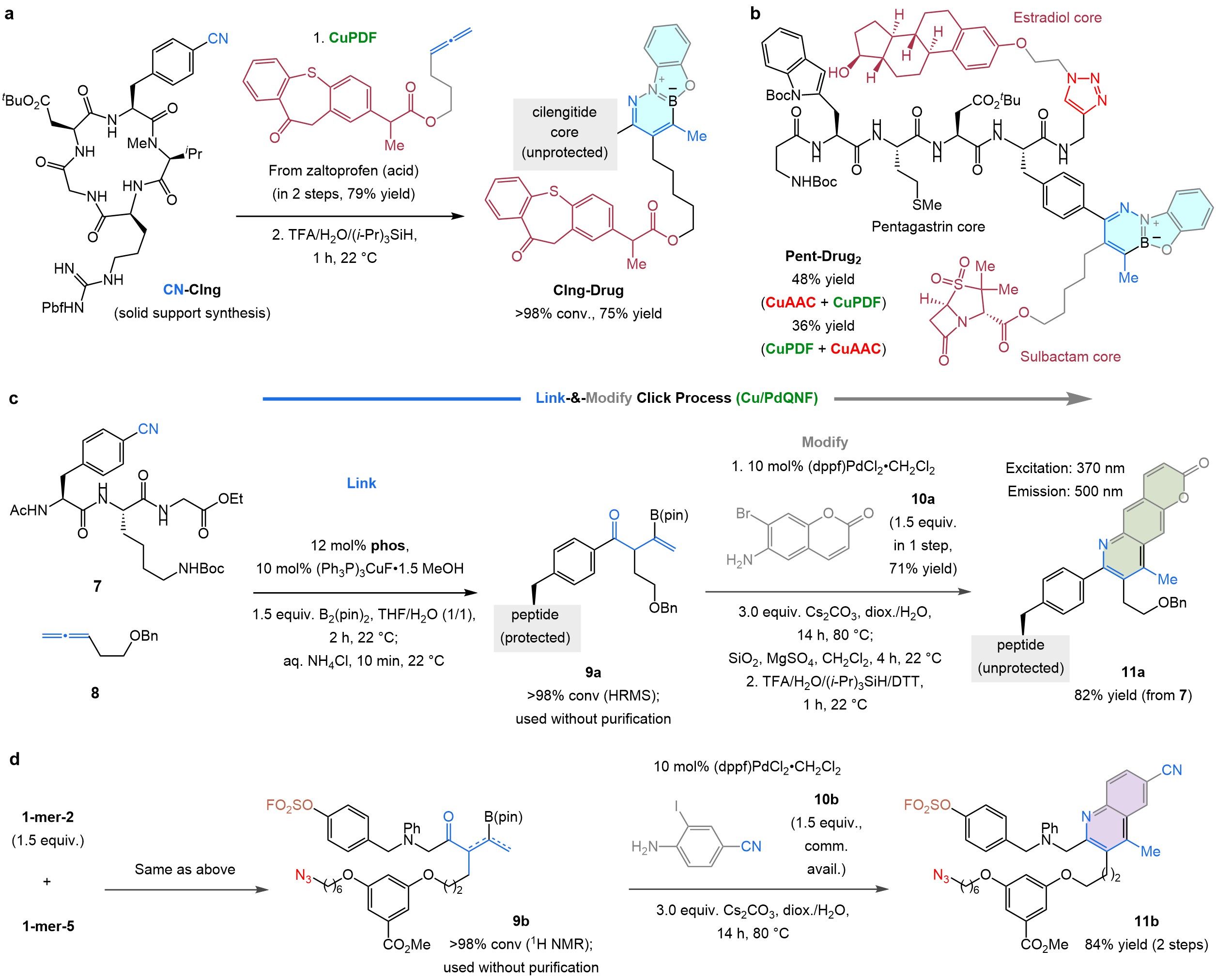
For applications in aqueous media, we developed Cu/PdQNF (Cu- and Pd-catalysed quinoline formation; Fig. 5c). Here, a b-boryl ketone, formed by ketimine hydrolysis, was coupled with a 2-haloaniline, a large assortment of which can be easily prepared (e.g., 10a) or are commercially available (e.g., 10b). Cu/PdQNF is also orthogonal to CuAAC and SuFEx (e.g., 11b, Fig. 5d).
Remarkably, our approach is just the second catalytic click strategy9 that generates modifiable linkers and the first and only existing approach for fabricating chemoselectively differentiable connectors. One can only imagine what would be possible with more orthogonal catalytic click options for forging adaptable linkers.
Contributors
Author
Amir H. Hoveyda
Director of the Laboratories of Catalytic Chemical Synthesis at the Institute for Supramolecular Science and Engineering, University of Strasbourg Patricia and Joseph T. ’49 Vanderslice Millennium Professor of Chemistry at Boston College
Contributing authors
Paulo H. S. Paioti
Group Leader at the Institute of Organic Chemistry and Biochemistry, IOCB Prague
Michele Formica
Postdoctoral Fellow at the Institute for Supramolecular Science and Engineering, University of Strasbourg
Meagan E. Hackey
Doctoral Student at the Department of Chemistry, Boston College
References
1. Kolb, H. C., Finn, M. G. & Sharpless, K. B. Click chemistry: diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. 40, 2004–2021 (2001).
2. Zhang, S. et al. Delayed catalyst function enables direct enantioselective conversion of nitriles to NH2-amines. Science 364, 45–51 (2019).
3. del Pozo, J. et al. Streamlined catalytic enantioselective synthesis of α-substituted β,γ-unsaturated ketones and either of the corresponding tertiary homoallylic alcohol diastereomers. J. Am. Chem. Soc. 142, 18200–18212 (2020).
4. Rostovtsev, V. V., Green, L. G., Fokin, V. V. & Sharpless, K. B. A Stepwise Huisgen Cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Ed. 41, 2596–2599 (2002).
5. Tornoe, C. W., Christensen, C. & Meldal, M. Peptidotriazoles on solid phase: [1,2,3]-triazoles by regiospecific copper(I)-catalyzed 1,3- dipolar cycloadditions of terminal alkynes to azides. J. Org. Chem. 67, 3057–3064 (2002).
6. Jang, H., Zhugralin, A. R., Lee, Y. & Hoveyda, A. H. Highly selective methods for synthesis of internal (α-) vinylboronates through efficient NHC–Cu-catalyzed hydroboration of terminal alkynes. Utility in chemical synthesis and mechanistic basis for selectivity. J. Am. Chem. Soc. 133, 7859–7871 (2011).
7. Gillingham, D. The role of boronic acids in accelerating condensation reactions of α-effect amines with carbonyls. Org. Biomol. Chem. 14, 7606–7609 (2016).
8. Li, S., Wu, P., Moses, J. E. & Sharpless, K. B. Multidimensional SuFEx click chemistry: sequential sulfur(VI) fluoride exchange connections of diverse modules launched from an SOF4 hub. Angew. Chem. Int. Ed. 56, 2903–2908 (2017).
9. Li, S. et al. SuFExable polymers with helical structures derived from thionyl tetrafluoride. Nat. Chem. 13, 858–867 (2021).
10. Yamazaki, C. M. et al. Antibody-drug conjugates with dual payloads for combating breast tumor heterogeneity and drug resistance Nat. Commun. 12, 3528 (2021).
11. Gilbert, M. R. et al. Cilengitide in patients with recurrent glioblastoma: the results of NABTC 03-02, a phase II trial with measures of treatment delivery. J. Neurooncol. 106, 147–153 (2012).
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in