Co(CN)3 Micro-Crystals with Well-Defined Coordination Structure: A Model Catalyst Beyond Co-N-C for the Oxygen Reduction Reaction
Published in Chemistry
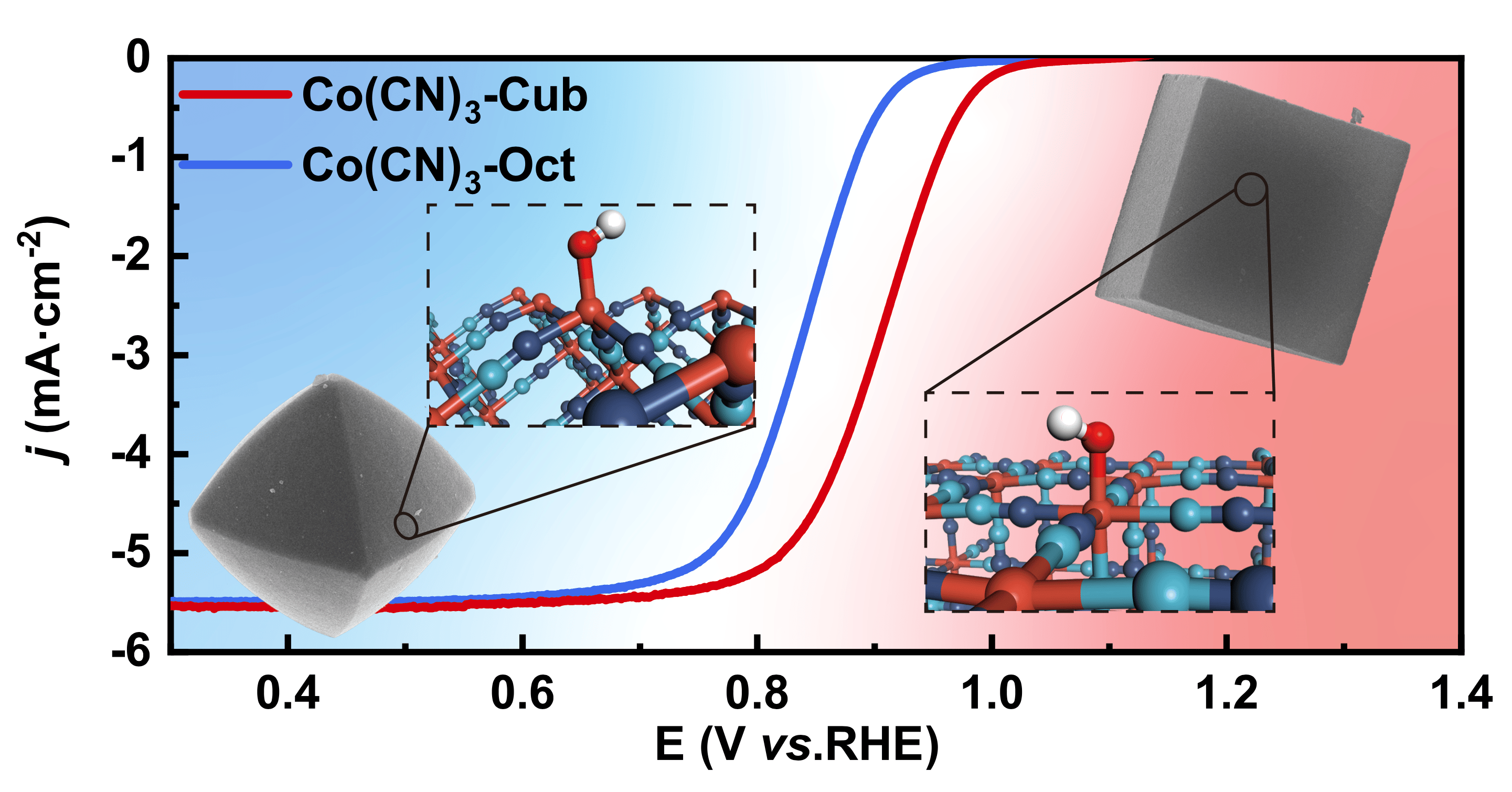
Fuel cells transcend the constraints of traditional heat engines, offering a more efficient and eco-friendly way of generating clean electricity. Platinum group metals have exhibited exceptional catalytic prowess in ORR, yet their exorbitant cost and limited availability have curtailed their widespread utilization. Carbon-based single metal atom catalysts, specifically the M-N-C catalysts, have shown great promise in anion exchange membrane fuel cells (AEMFCs). However, presently synthesized M-N-C catalysts often exhibit irregular coordination structures, stemming from uncontrollable pyrolysis processes, resulting in haphazardly configured metal site coordination environments. Furthermore, to attain the desired "isolated" states of metal sites in M-N-C type catalysts, intentional site density reduction is required, inadvertently increasing mass transport resistance and, consequently, diminishing catalyst performance in fuel cells. This limitation impedes their widespread adoption. Hence, the development of catalysts boasting well-defined active sites and maintaining high active site densities stands as an essential yet formidable challenge.
In this study, Prof. Xiaoming Sun's team, the Nanochem Group at Beijing University of Chemical Technology, reports a new type of catalyst: Co(CN)3 micro-crystals with both well-defined active sites and high site densities, addressing the issues in traditional M-N-C catalysts. The presence of cyanide ligands in Co(CN)3 allows us to view the Co center as analogous to M-N-C configurations. The micro-crystal exhibits ordered crystalline structures with controlled cubic and octahedral morphologies, ensuring precise coordination of Co sites on the surface. We conducted a detailed investigation into the relationship between coordination number and catalytic performance based on the two facets of Co(CN)3. The cubic Co(CN)3 (referred to as Co(CN)3-Cub) with affirmatively 5-coordinated Co sites exhibits outstanding ORR half-wave potential and AEMFC peak power density.
Structural identification
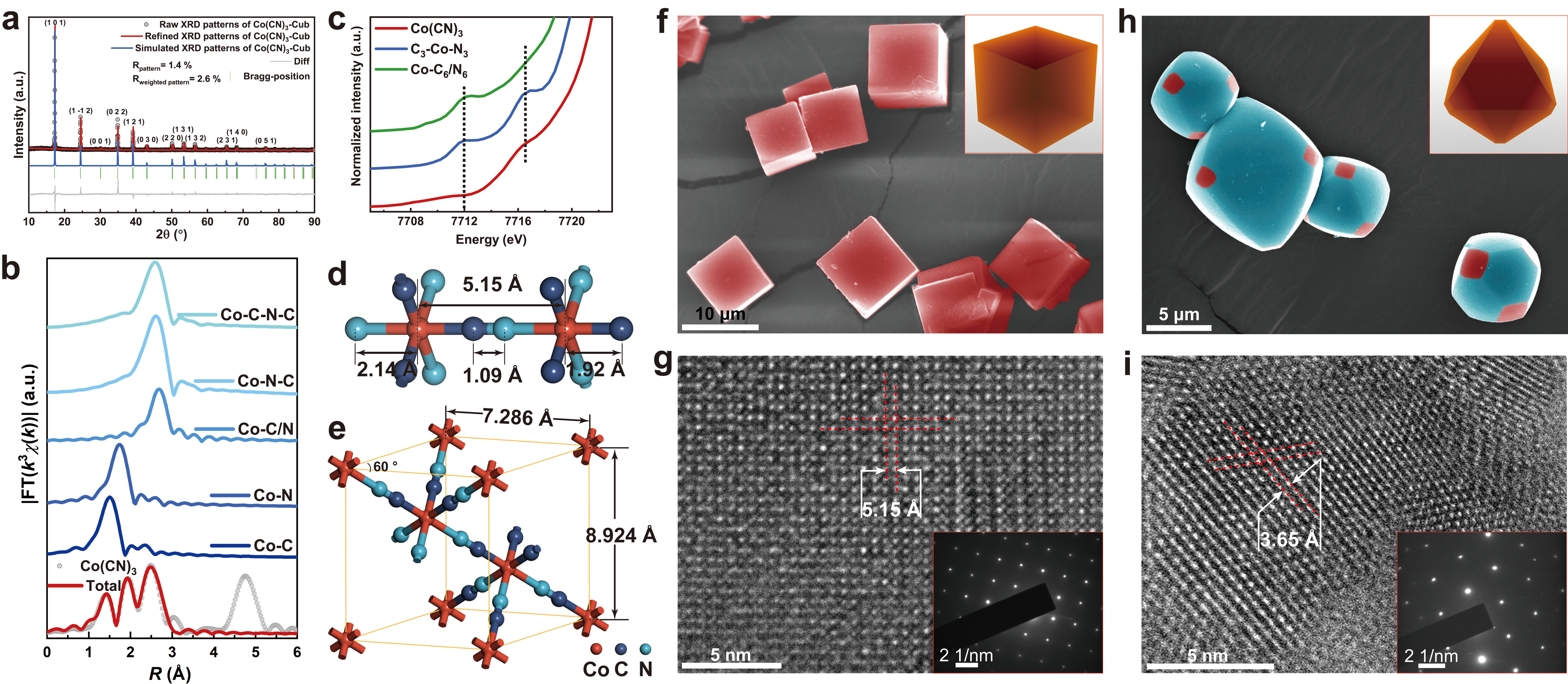
Concerning the bulk structure of Co(CN)3, we conducted the Single Crystal X-ray Diffraction characterization of Co(CN)3 and validated the asymmetric coordination environment within Co(CN)3 using X-ray Absorption Near-Edge Structure (XANES) spectra, which are highly sensitive to symmetric structures. Thanks to the well-ordered nature of the crystalline material, we were able to investigate the surface structure with precision. High-Angle Annular Dark-Field Scanning Transmission Electron Microscopy (HAADF-STEM) confirmed the arrangement of Co atoms on crystal faces with varying morphologies, allowing us to identify 5-coordinated and 3-coordinated Co sites on cubic and octahedral Co(CN)3 surfaces, respectively.
In addition, we wish to convey our profound gratitude to Associate Research Fellow Juncai Dong at the Institute of High Energy Physics, Chinese Academy of Sciences. His formidable grasp of the theoretical underpinnings of X-ray Absorption Spectroscopy (XAS) technology, coupled with his distinctive technical prowess, has been of invaluable assistance to this work.
Coordination dependent ORR performance studies
The synthesized Co(CN)3 was employed to investigate the impact of coordination numbers on ORR performance. Co(CN)3-Cub, with 5-coordinated Co sites, exhibited a higher onset potential of 0.99 V and half-wave potential of 0.90 V compared to Co(CN)3-Oct, underscoring the influence of Co site coordination on ORR activity. Notably, Co(CN)3-Cub surpassed commercial Pt/C catalysts, demonstrating outstanding ORR performance. Moreover, crystalline surfaces and robust chemical bonds conferred enhanced stability, as evidenced by a mere 20 mV loss for Co(CN)3-Cub after 10,000 cycles.
It is beneficial for minimizing the diffusion distance of oxygen in the real fuel cell applications, while the active site density on the surface of the crystals is much higher than that of the M-N-C catalysts. Harnessing the high activity and Co site density of Co(CN)3, we fabricated an AEMFC membrane electrode assembly (MEA) employing Co(CN)3-Cub as the cathode catalyst. The Co(CN)3-Cub MEA showed a peak power density of 1.67 W·cm-2 at the current density of 3.84·A cm-2 in H2-O2 cell, which surpassed the MEA using the 60 wt% commercial Pt/C and ranked forefront in the reported AEMFCs. Furthermore, it is imperative to acknowledge the invaluable contributions of Dr. Xingdong Wang, whose diligent efforts in AEMFC testing played a pivotal role in advancing our research endeavors.
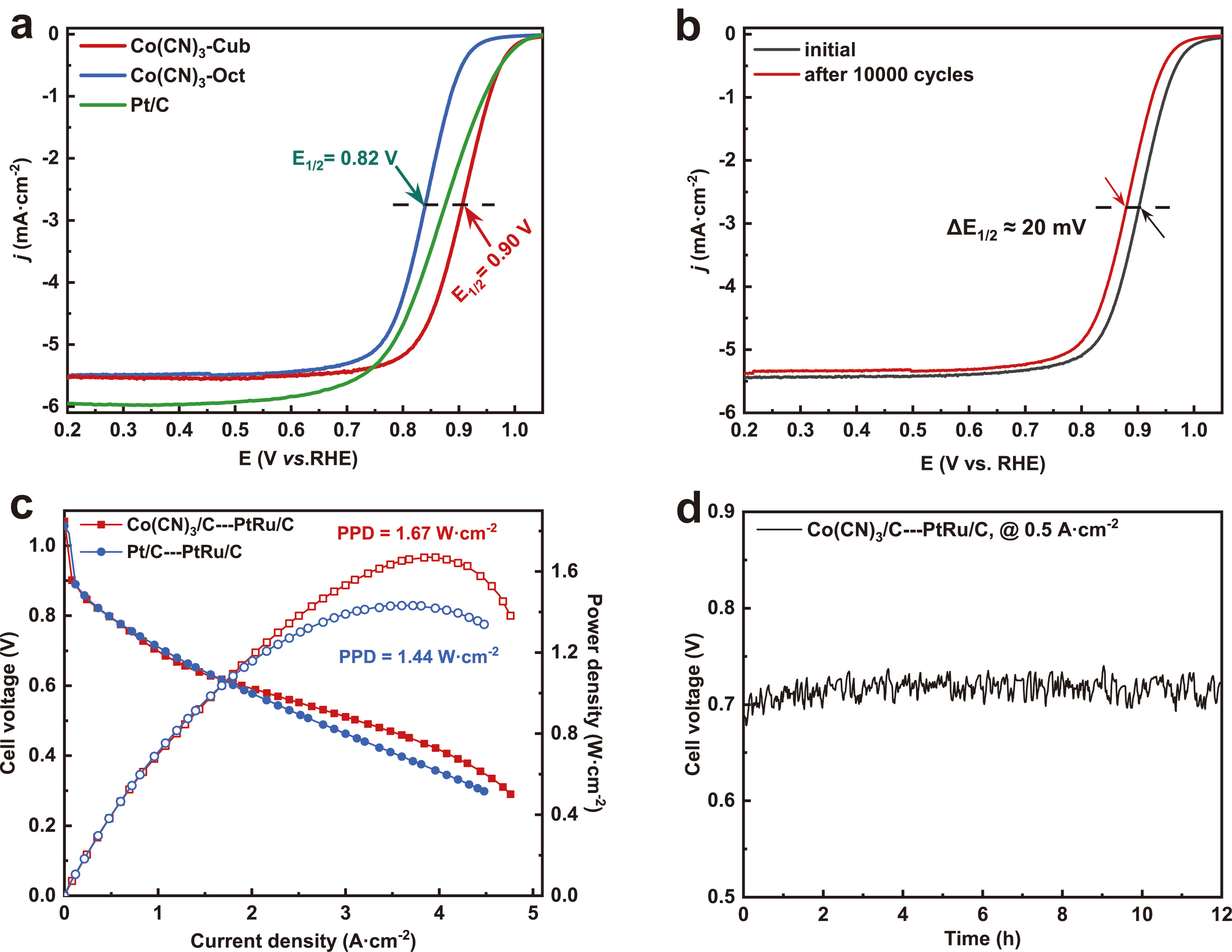
Fig. 2. Electrocatalytic ORR performance of the Co(CN)3 catalysts. a, LSV polarization curves. b, LSV curves of the Co(CN)3-Cub before and after 10000 CV cycles. c, H2-O2 AEMFC polarization and power density curves. d, The stability of the MEA using Co(CN)3-Cub as cathode.
Mechanism studies
We observed the ORR reaction processes occurring on the Co(CN)3-Cub and Co(CN)3-Oct crystalline facets, i.e., the 5- and 3-coordinated Co sites, using in-situ attenuated total reflectance surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) and confirmed OH desorption as the potentially determining step with the aid of DFT calculations. 5-coordinated Co sites exhibited weaker OH adsorption energies by featuring d-band centres further away from the Fermi energy level, and consequently made it easier to desorb, thus leading to an increase in activity. We wish to extend our heartfelt appreciation to Hao Sun and Jinjie Fang for their exemplary contributions in the DFT computations and the in-situ ATR-SEIRAS tests.
Bigger picture
In this work, Dr. Kai Sun, Prof. Zhongbin Zhuang, Assoc. Prof. Shubo Tian, Prof. Xiaoming Sun and co-authors synthesised and accurately characterised the novel Co(CN)3 micro-crystals and utilized it as ORR catalyst. The primary benefit of crystalline materials over amorphous nitrogen-doped carbon materials lies in their well-defined, long-range order. This characteristic allows us to deduce surface coordination environments directly from the crystal structure, avoiding the fact that the metal coordination number and configurations are buried by the averaging, which is associated with XAS and its fitting techniques. This work not only illustrates the promising of Co(CN)3 micro-crystals in fuel cells, but also, benefit from the incredibly precise surface structure, provides a reference system for the coordination number-performance relationship experimentally, which may be of great research interest in the design and development as well as the standardization of the similar M-N-C materials.
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in