Cobalt(III)-catalyzed asymmetric ring-opening of 7-oxabenzonorbornadienes via indole C–H functionalization
Published in Chemistry
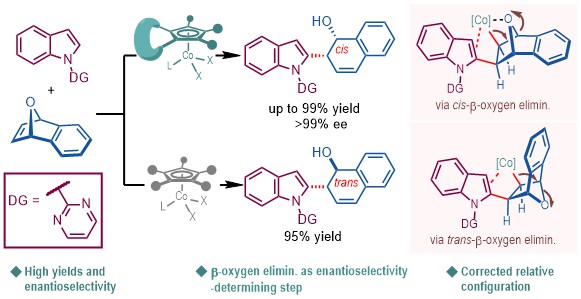
Asymmetric ring-opening (ARO) of strained bicyclic olefins is an important platform for building-up molecular complexity. Merging ARO reactions with transition-metal-catalyzed C–H activation has become an area of recent interest (Chem. Soc. Rev. 2021, 50, 3013-3093). However, the C–H activation-induced ARO reactions of 7-oxabenzonorbornadienes via β-oxygen elimination remained elusive. Mechanistically, it was regarded that the ARO reactions proceeded via the exo-coordination of a carbon–metal species to the olefin moiety, migratory insertion and cis-β-heteroatom elimination. And the migratory insertion was speculated as the enantioselectivity-determining step in the ARO reactions (Fig. 1).
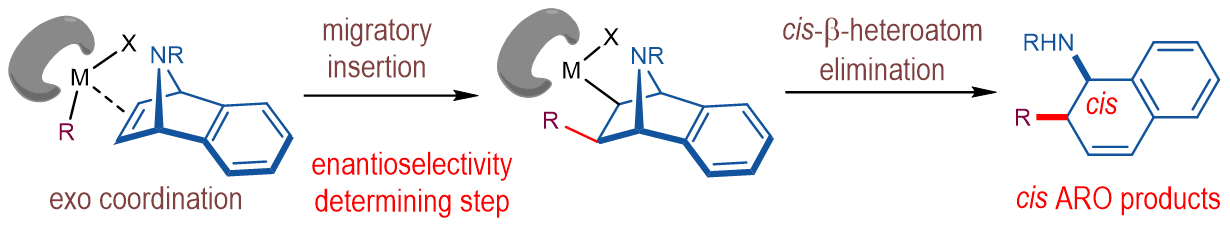
Fig. 1. Common mechanistic manifold of asymmetric ring-opening of bicyclic alkenes
In this work, high yields and excellent stereoselectivity were obtained for a wide range of cis-ring-opening products under the optimal conditions. More interestingly, the utilization of Cp*Co(CO)I2
as the catalyst led to reversed diastereoselectivities favoring the trans ring-opening products (Fig. 2).
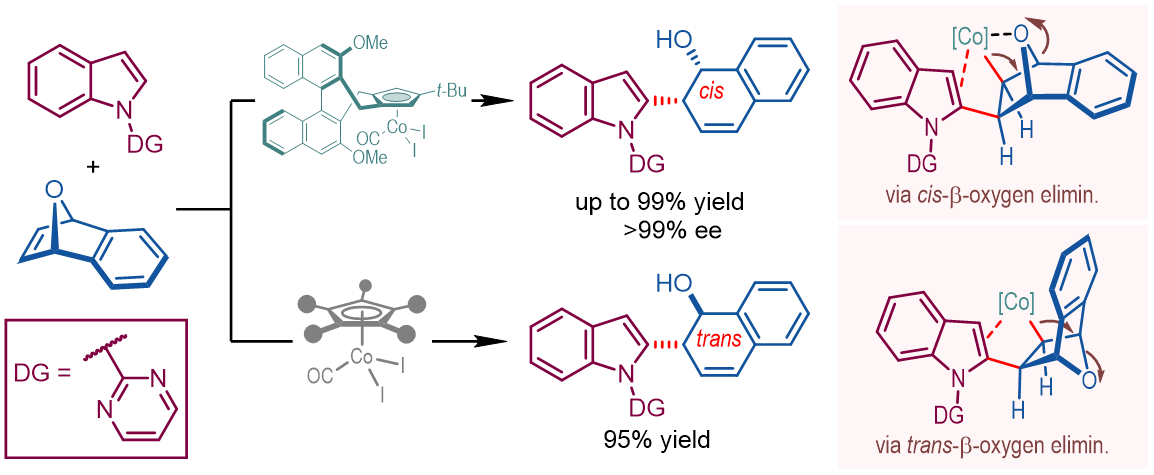
Fig. 2. Co-cat. asymmetric ring-opening of 7-oxabenzonorbornadienes
Combined experimental and computational studies suggested the mechanistic pictures of the CpCo-catalyzed ARO reactions where cis- and trans-β-oxygen elimination work as the selectivity-determining step for enantioselective and racemic reactions, respectively (Fig. 3). Such results are different from the previous cognition (Angew. Chem. Int. Ed. 2019, 58, 322-326; Angew. Chem., Int. Ed. 2019, 58, 2514-2518).
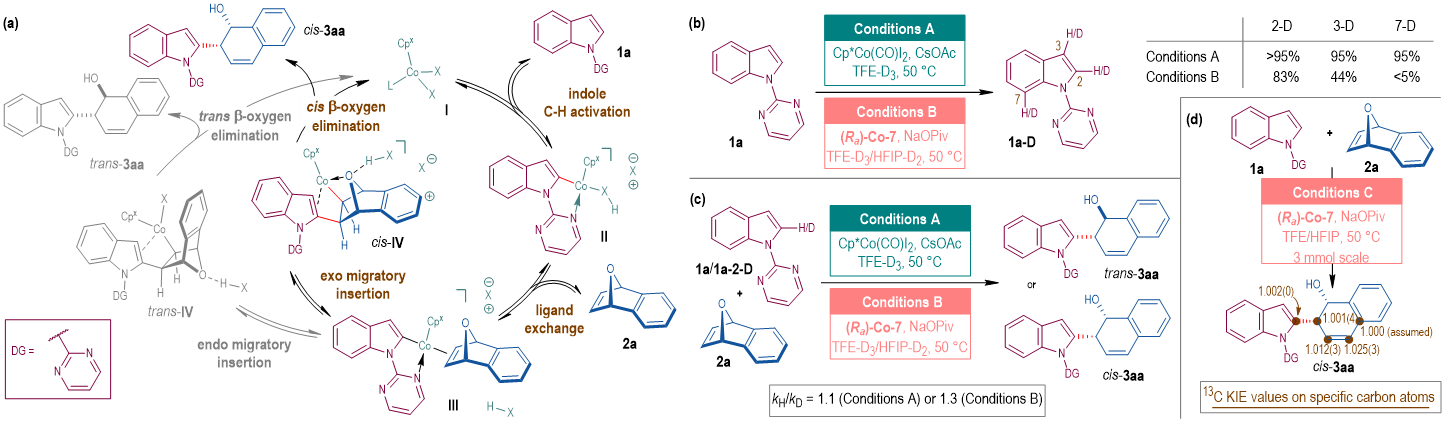 Fig. 3. Proposed catalytic cycle and mechanism experiment
Fig. 3. Proposed catalytic cycle and mechanism experiment
As an extension, 7-azabenzonorbornadiene could also be applied in the similar ARO reactions in a diastereodivergent manner (Fig. 4).
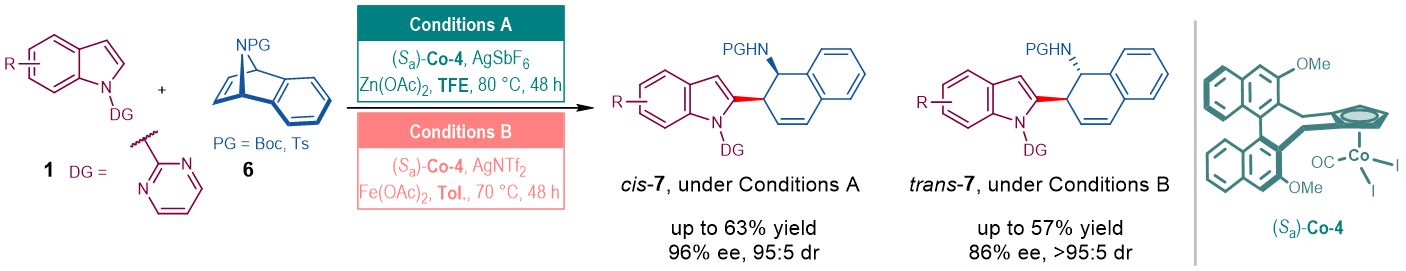 Fig. 4. ARO reactions of 7-azabenzonorbornadiene
Fig. 4. ARO reactions of 7-azabenzonorbornadiene
More details of this work could be found here: “Cobalt(III)-catalyzed asymmetric ring-opening of 7-oxabenzonorbornadienes via indole C–H functionalization” in Nature Communications.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in