Comparative Quantum-Classical Dynamics of Natural and Synthetic Molecular Rotors Show How Vibrational Synchronization Modulates the Photoisomerization Quantum Efficiency
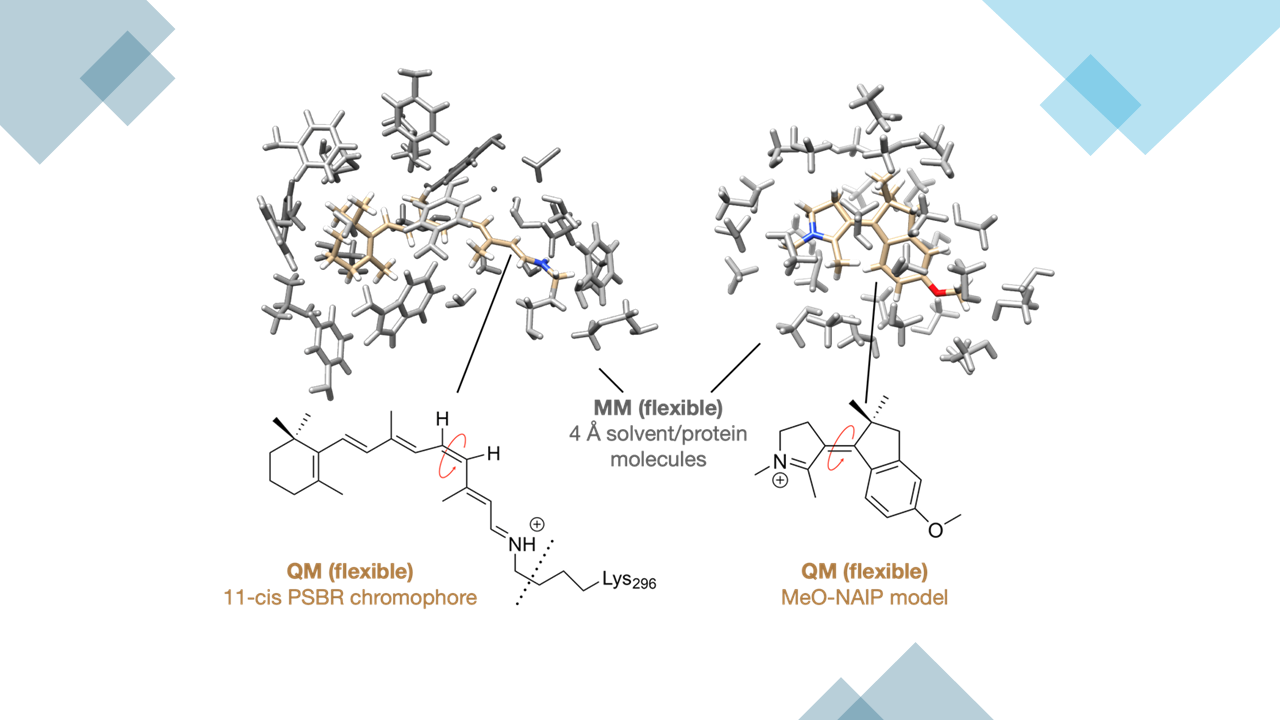
Our recent publication in Nature Communications presents a detailed examination of the quantum mechanical mechanisms underlying energy conversion in molecular rotors. This investigation brings to light significant findings regarding the quantum efficiency of synthetic molecular rotors in comparison with biological systems, such as Rhodopsin (Figure 1a). Here, we integrate critical results that underscore the novelty and scientific impact of our study.
Novel Insights into Promoter Modes
Our research has led to a significant discovery regarding the synthetic molecular rotor. We have identified a lower-frequency promoter mode that periodically enhances the magnitude of critical vibrational modes involved in isomerization (Figure 1b,c). This promoter mode operates on a different timescale of approximately 250 fs, which is distinct from the previously recognized 40 fs hydrogen out-of-plane wag mode associated with Rhodopsin. This finding suggests that photochemical systems can selectively use specific promoter modes to enhance the quantum efficiency of the energy conversion processes.
Advancements in Reactivity Conditions
Our study has extended the previously reported reactivity conditions that focus on the directional change of the vibrational mode τ-velocity (Figure 1d,e), where τ represents a dihedral angle proportional to the overlap between the p-orbitals pivotal in monitoring the progress of the isomerization double-bond-making-breaking process. We have found that reactivity depends not only on the directionality of τ-velocity (a necessary condition) but also on its magnitude (a sufficient condition). This nuanced understanding implies that both the rate and extent of τ change play a vital role in controlling the reactivity of molecular systems. This advancement provides a link between the effect of the promoter mode and the canonical Landau-Zener model, which is fundamental to photochemistry.
Solvent Effects on Quantum Efficiency
Additionally, we report an original finding concerning the solvent's (or counterion's) inhibitory effect on the quantum efficiency of the synthetic molecular rotor. The solvent affects the frequency of a vibrational mode, enabling it to act as a promoter. This insight sheds light on the complex interplay between the molecular environment and quantum efficiency, highlighting the role of specific vibrational mode frequency as a determinant in the efficacy of energy conversion processes.
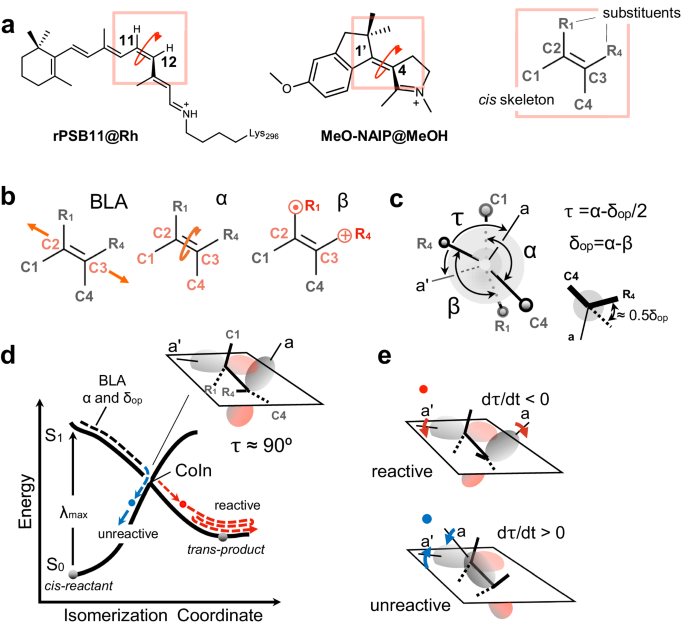
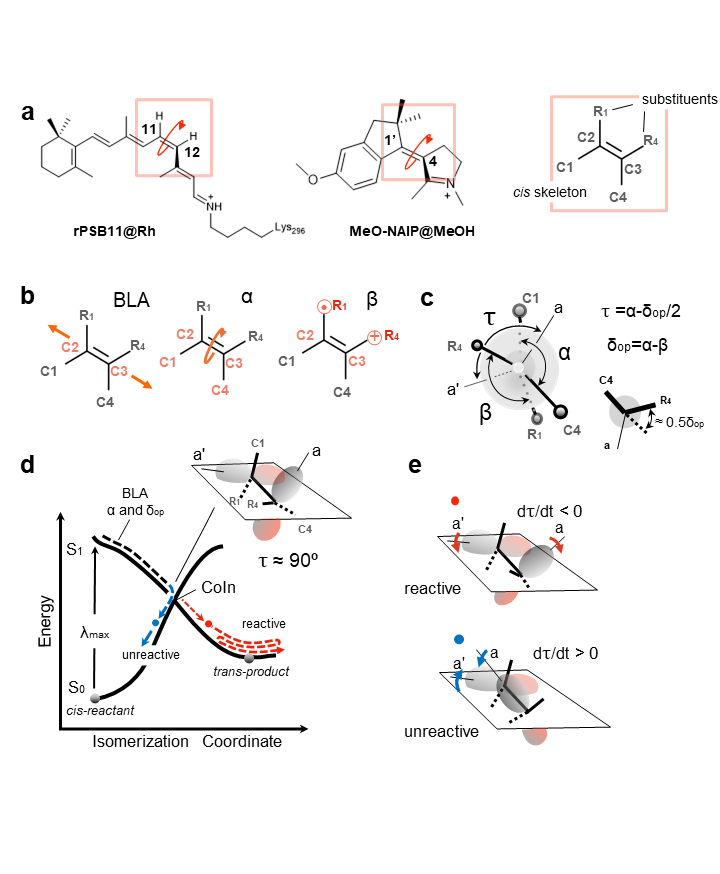 Figure 1. Structures and reactivity of systems under comparison. a) rPSB11 and MeO-NAIP structures and their common ethylenic framework and definition of the skeletal (C1, C4) and ethylenic (R1, R4) substituents. Notice the consistent out-of-plane counterclockwise (CCW) deformation in the two systems. b) Coordinates driving the photoinduced double bond isomerization: the skeletal bond-length-alternation (BLA), the double-bond twisting of the ethylenic fragment (α), dihedral describing the wag (β) of the ethylenic substituents. c) Definition of τ, α, and δop, where τ is the dihedral defining the overlap between the p-orbitals (represented by the axis a and a') at C2 and C3, and δop is a convenient function of β describing the substituent wag relative to the molecule conjugated framework. d) Schematic illustration of the potential energy surfaces (PESs) of both the ground state (S0) and the excited state (S1) (in solid black line). The trajectory represented through the evolution of double-bond isomerization coordinates BLA, α, and δop (in dashed lines). The red and blue circles represent unreactive and reactive decay points, respectively. e) Relationship between orbital overlap, dτ/dt phase, and reactivity. A reactive event can be associated with the π-bond breaking (dτ/dt < 0), and the unreactive event is associated with the π-bond making (dτ/dt > 0), defined by the p-orbitals (a and a') overlap at decay.
Figure 1. Structures and reactivity of systems under comparison. a) rPSB11 and MeO-NAIP structures and their common ethylenic framework and definition of the skeletal (C1, C4) and ethylenic (R1, R4) substituents. Notice the consistent out-of-plane counterclockwise (CCW) deformation in the two systems. b) Coordinates driving the photoinduced double bond isomerization: the skeletal bond-length-alternation (BLA), the double-bond twisting of the ethylenic fragment (α), dihedral describing the wag (β) of the ethylenic substituents. c) Definition of τ, α, and δop, where τ is the dihedral defining the overlap between the p-orbitals (represented by the axis a and a') at C2 and C3, and δop is a convenient function of β describing the substituent wag relative to the molecule conjugated framework. d) Schematic illustration of the potential energy surfaces (PESs) of both the ground state (S0) and the excited state (S1) (in solid black line). The trajectory represented through the evolution of double-bond isomerization coordinates BLA, α, and δop (in dashed lines). The red and blue circles represent unreactive and reactive decay points, respectively. e) Relationship between orbital overlap, dτ/dt phase, and reactivity. A reactive event can be associated with the π-bond breaking (dτ/dt < 0), and the unreactive event is associated with the π-bond making (dτ/dt > 0), defined by the p-orbitals (a and a') overlap at decay.
Implications for Molecular Machine Design
These discoveries have profound implications for the design and optimization of synthetic molecular machines. Our research paves the way for developing more efficient molecular rotors by elucidating the mechanisms that enhance or inhibit quantum efficiency. By understanding the nuanced role of promoter modes and the molecular environment conditions that influence these systems, we expect to advance our fundamental knowledge of molecular energy conversion and open new possibilities for utilizing molecular motion in future technologies.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in