Electrochemically reducing carbon dioxide (CO2RR) to carbon-based chemical fuels provides an avenue to the large-scale storage of renewable energy. Among products in CO2RR, ethanol is a high-energy-density liquid product that arouses a great deal of interest, for it commands a comparatively high market price and corresponds to a US$75 billion/year market.
Technoeconomic analysis of CO2RR systems shows that, when productivity resides below 100 mA cm-2 partial current densities, C2 electrosynthesis is less likely to have commercial impact. Unfortunately, the performance of prior reports of direct ethanol electroproduction has consistently resided below this benchmark. Until now, the best ethanol Faradaic efficiency (FE) reported was 41% when considering all prior reports documenting current densities greater than 10 mA cm-2.
To determine how to select in favour of ethanol, we needed to consider the branching between ethanol and ethylene, its main competing product. Both are derived from a shared key intermediate (HOCCH*). The key to favor the ethanol pathway is to suppress the C-O bond-breaking from HOCCH*.
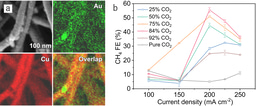
We developed catalysts by overcoating Cu with nitrogen-doped carbon (N-C/Cu) via sputter deposition. By tuning the ratio of Ar to N2 during sputter deposition, we obtained a series of N-C/Cu catalysts with different atomic percentages of nitrogen in the N-C layers. Compared to bare Cu, all these N-C/Cu catalysts exhibited enhanced selectivity towards ethanol in CO2RR. We ultimately achieved CO2-to-ethanol performance with a record FE of 52% at a partial current density of 156 mA cm-2. The corresponding cathodic energy efficiency (EE) and full cell EE for ethanol achieve values of 31% and 16% in flow cell and membrane electrode assembly systems, respectively.
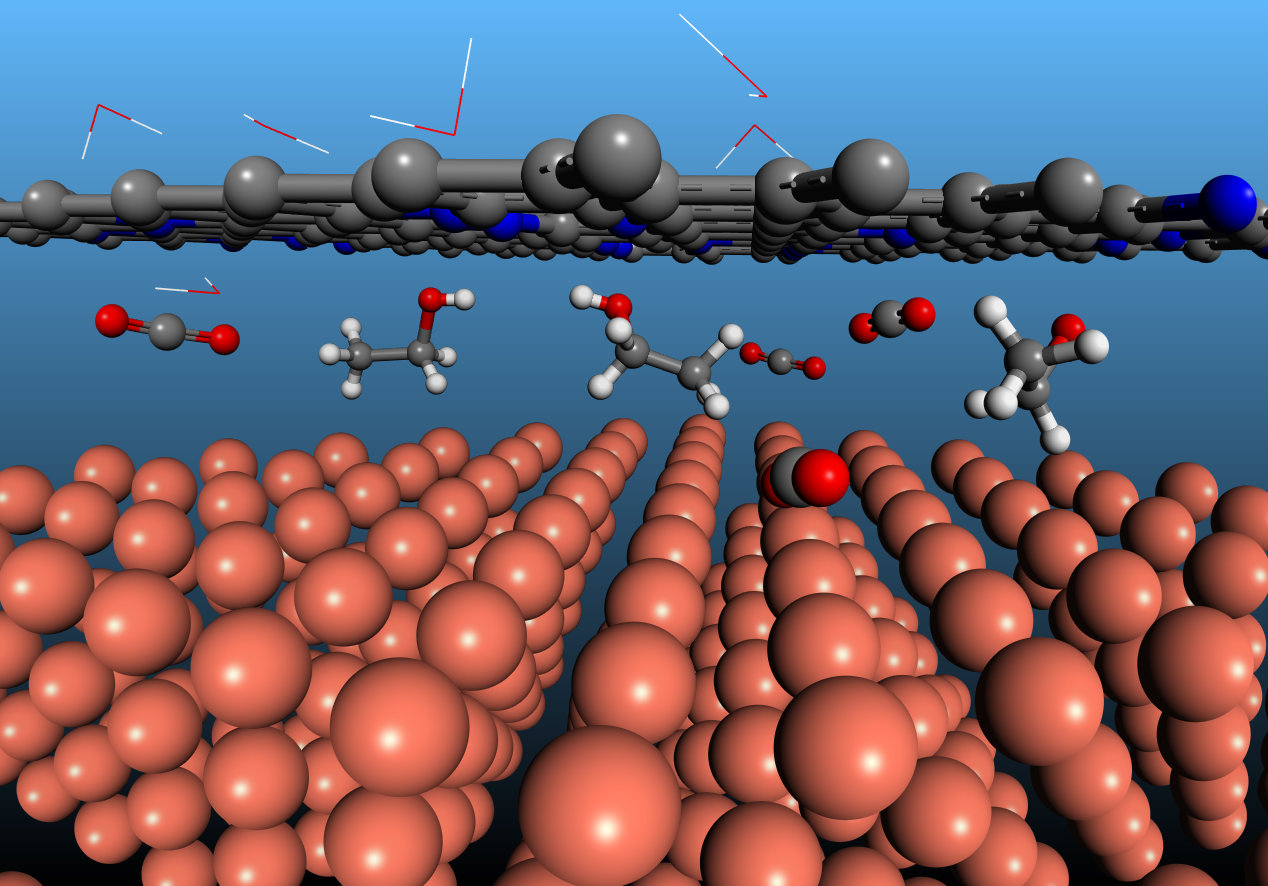
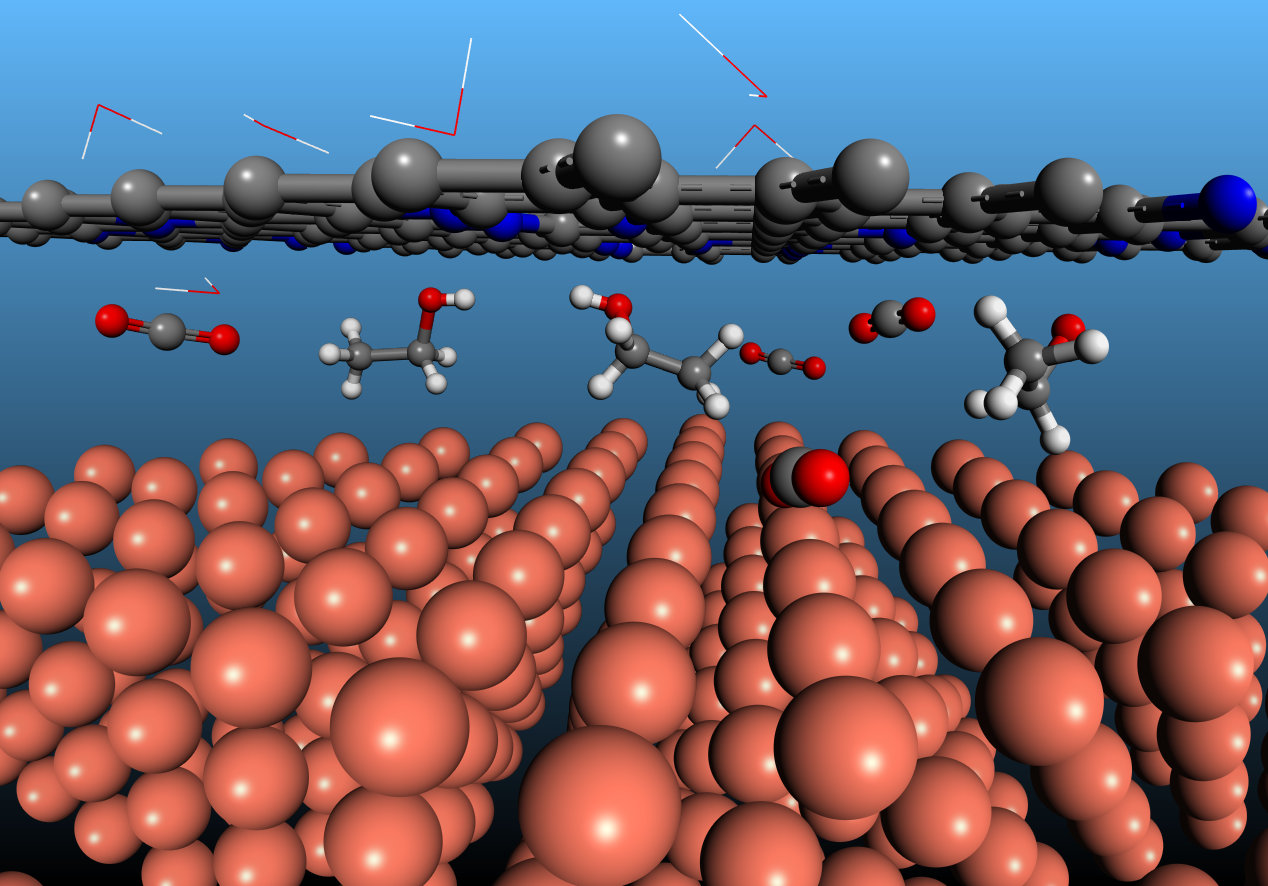
Scheme illustrating the CO2 intercalation at the N-C/Cu interface to produce ethanol. Cu, N, C, and O atoms are presented as orange, blue, grey, and red balls, respectively, and water molecules are shown as lines.
Electron microscopy revealed gaps having width typically less than 1 nm between Cu and N-C layers; these we propose serve as nanoreactors in CO2RR. With the aid of calculations and simulation, as well as in-situ Raman and X-ray absorption, we found that the confinement of the N-C layer offers strong electron-donating ability atop the Cu catalyst and facilitates C-C coupling while suppressing the deoxygenation of the intermediate HOCCH* to ethylene. It is these properties that promote selectivity to ethanol.
If you are interested in our work, you may find the full paper here: https://www.nature.com/articles/s41560-020-0607-8
Follow the Topic
-
Nature Energy
Publishing monthly, this journal is dedicated to exploring all aspects of this on-going discussion, from the generation and storage of energy, to its distribution and management, the needs and demands of the different actors, and the impacts that energy technologies and policies have on societies.
Related Collections
With Collections, you can get published faster and increase your visibility.
Microgrids and Distributed Energy Systems
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in