Controlling phagosomal lipids for better antigen processing
Published in Cell & Molecular Biology
Phagocytes are immune cells that patrol our bodies looking for their next meal: pollution particles, fungi, bacteria, even cancer cells – can be eaten and destroyed through the process of phagocytosis. To do this, phagocytes surround a target with their plasma membrane which becomes sac called phagosome that pinches off and matures in part by fusing with vesicles called lysosomes that deliver digestive enzymes. Some phagocytes don’t just blindly destroy, but instead preserve small pieces of the ingested material to serve as antigens, which are critical to alert the adaptive immune system (T cells and B cells). But to achieve a balance between destruction and preservation, the digestive process needs to be tightly controlled.
To fuse or not to fuse
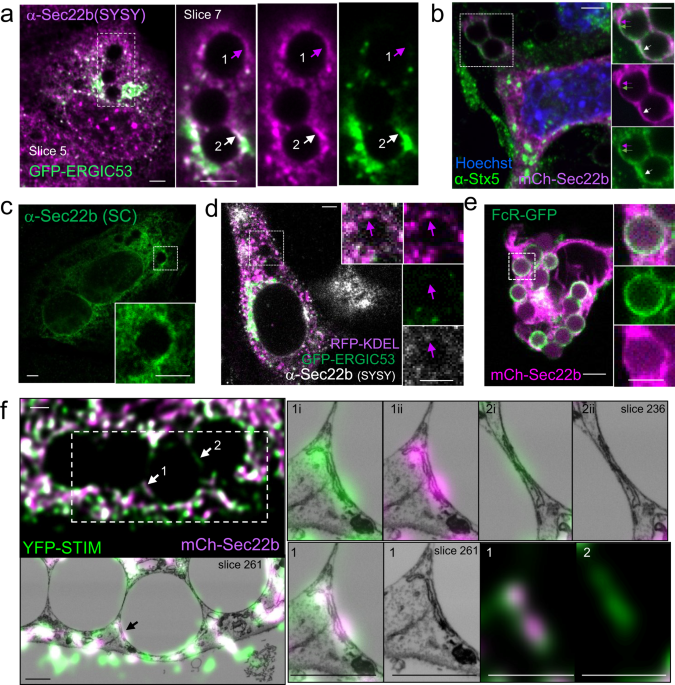
Sec22b is a protein responsible for fusion of early secretory vesicles, found also to control phagosome maturation in antigen presenting cells 1-3, though how or why remained unknown. Its role in antigen processing and presentation was also the source of some controversy 4-6. Part of the problem may be because most studies assumed that fusion of early secretory vesicles was key to Sec22b’s effect on antigen processing, until an important clue emerged. In a seminal study, Petkovic and colleagues found that, in neurons, Sec22b could refrain from driving fusion and instead act as a stable link or tether between two membranes, holding them only tens of nanometers apart, in a structure called a membrane contact site7. These non-fusing contact sites have been a hot topic in cell biology in recent years 8. Noticed more than 30 years ago, their significance wasn’t appreciated until perhaps the last decade, when people started realizing that they served as platforms for localized communication between organelles, like tunnel transfer of calcium ions, or lipid transfer across the contact site gap. Previously, I had found that contact sites anchored by the calcium regulator STIM1 promoted localized calcium hotspots around phagosomes 9, 10 that were needed to drive efficient phagosome maturation. Yet our studies also suggested STIM1 was not the only tether, so when I read the Petkovic study I thought this could be the answer to the Sec22b enigma in the antigen presentation studies: that maybe it's not fusing, it's tethering.
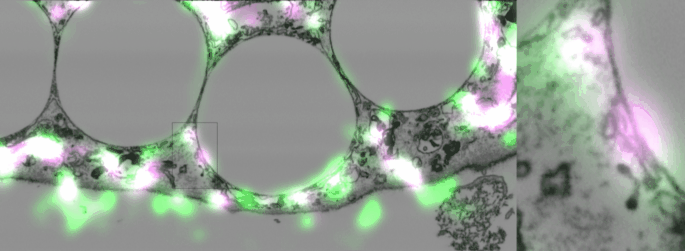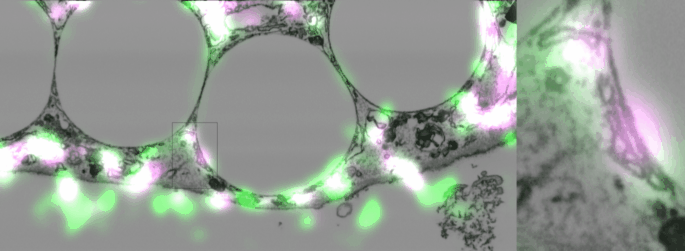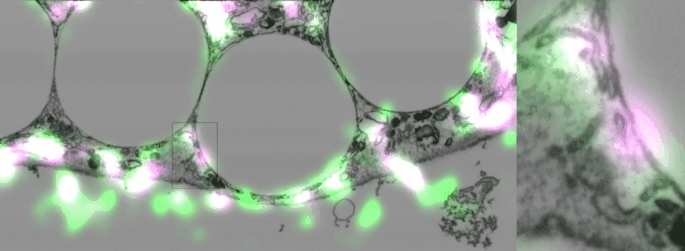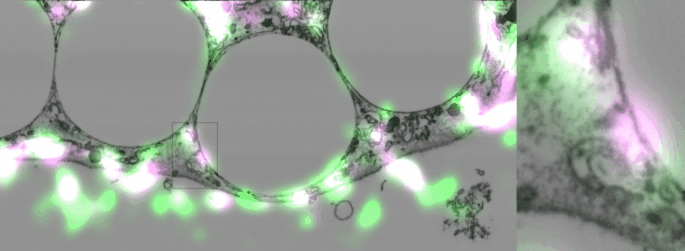
Imaging contacts in 3D
One of the difficulties of studying cellular contact sites is that they are so tiny, 10-30 nanometers, that the resolution in light microscopy is not enough to distinguish them, (though to be fair super-resolution techniques are getting there). However, electron microscopy is still the gold standard, and even so, with classic techniques using slices of 20-50 nm, two fusing organelles can appear to be just next to each other. A new focused-ion-beam electron microscope in our institute not only made it possible to take 3D slices spaced 10 nm apart but also to correlate them with fluorescent images. This technology allowed us to eliminate any doubt that the Sec22b we found around phagosomes was present in non-fusing contact sites.
No, it's not calcium
Our initial hypothesis was that Sec22b might be controlling localized calcium signaling as there were some hints in the Petkovic study that this might be the case. We were motivated by initial observations that co-expression of Sec22b had a vivid effect on the behavior of the calcium signaling regulator STIM1. This made us think were on to something, so we spent a lot of time chasing a lead that in the end just didn’t pan out because calcium signals themselves were hardly changed. We think that Sec22b might have both positive and negative effects on calcium signals, which may be related to a contact site gap that is a little smaller than what is optimal for STIM1, but we didn’t get a chance to delve too much into this. These observations in themselves are interesting for researchers focused on calcium signals and contact site biology, but it was clear that the slight changes in calcium signaling weren’t enough to explain changes in phagosome maturation.
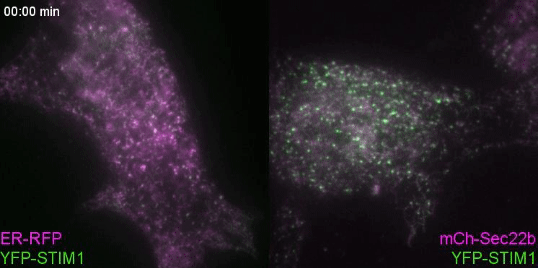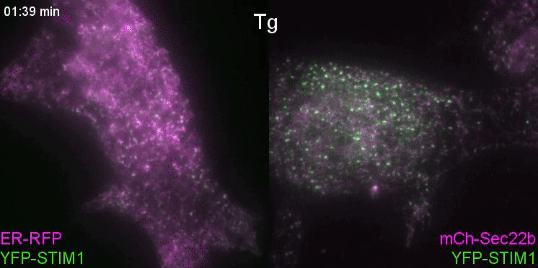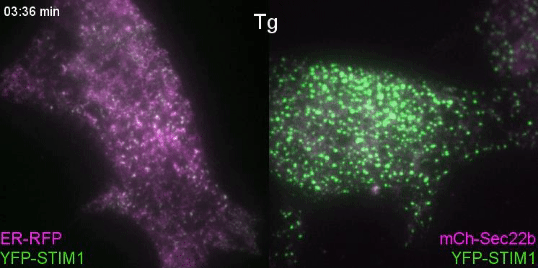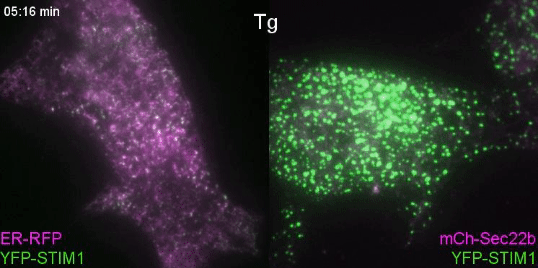
Controlling lipids for a healthier (phagosomal) digestion
Lipids transfer is another major function of membrane contact sites, so this is what we turned our attention to next. In the Petkovic study, researchers found that disrupting the yeast equivalent of Sec22b led to lipid changes at the cell membrane. This seemed like a good alternative because we knew from other studies that lipids on the surface of phagosomes were important for phagosome maturation 11, 12. Our collaborator Dr. Francesca Giordano, an expert in lipid transfer at membrane contact sites, was instrumental in helping us to hone in on the lipid transfer protein ORP8. By genetically manipulating phagocytes with ORP8 or Sec22b mutants and artificial contact site tethers, we discovered that by changing the nature and number of contacts, we could change the levels of certain lipids on phagosomes. And these changes correlated with lysosome fusion and antigen degradation levels.
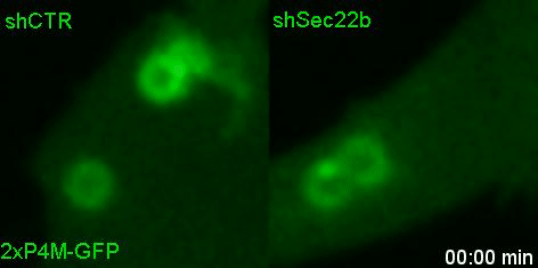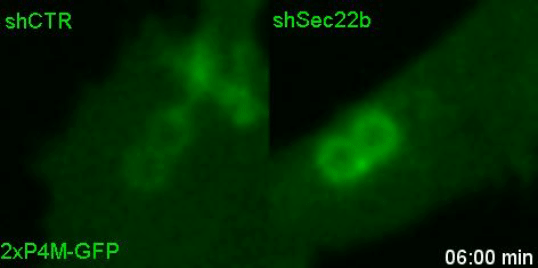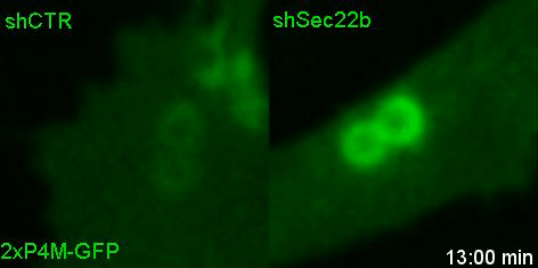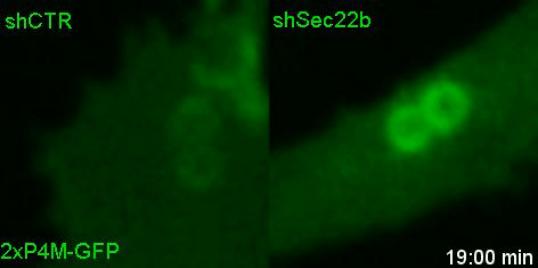
So what.... and what next?
In summary, our study adds to the understanding of contact site biology, calcium signaling, as well as the biology of phagosome maturation. From a translational perspective, contact sites and lipid transfer proteins such as ORP8 represent molecules and pathways that may be targeted in a therapeutic context, for example to get pathogen-stalled phagosomes back on the track to destruction. In addition, our study clarifies some of the mystery surrounding the function of Sec22b, by describing how it controls fusion with lysosomes. Yet one burning question that remains to be resolved is whether Sec22b is actually the mediator of fusion between phagosomes and these early secretory vesicles. This is a key issue because such vesicles are known to contain components that are critical for T cell activation, so understanding what is happening there could be very interesting for optimizing vaccines or anti-cancer immunotherapies 13, 14. We did find a very close association with these secretory vesicles lurking around the phagosomes even in the model phagocyte. We’re still working on it, but I do speculate that factors controlling Sec22b are at the heart of the balance between tethering and fusion, but all I can say with certainty for now is to stay tuned.....and to be continued....
References
- Becker, T., Volchuk, A. & Rothman, J.E. Differential use of endoplasmic reticulum membrane for phagocytosis in J774 macrophages. Proceedings of the National Academy of Sciences of the United States of America 102, 4022-4026 (2005).
- Hatsuzawa, K. et al. Sec22b is a negative regulator of phagocytosis in macrophages. Mol Biol Cell 20, 4435-4443 (2009).
- Cebrian, I. et al. Sec22b regulates phagosomal maturation and antigen crosspresentation by dendritic cells. Cell 147, 1355-1368 (2011).
- Wu, S.J. et al. A Critical Analysis of the Role of SNARE Protein SEC22B in Antigen Cross-Presentation. Cell Rep 19, 2645-2656 (2017).
- Nunes-Hasler, P. & Demaurex, N. The ER phagosome connection in the era of membrane contact sites. Biochim Biophys Acta Mol Cell Res 1864, 1513-1524 (2017).
- Montealegre, S. & van Endert, P. MHC Class I Cross-Presentation: Stage Lights on Sec22b. Trends Immunol 38, 618-621 (2017).
- Petkovic, M. et al. The SNARE Sec22b has a non-fusogenic function in plasma membrane expansion. Nat Cell Biol 16, 434-444 (2014).
- Scorrano, L. et al. Coming together to define membrane contact sites. Nat Commun 10, 1287 (2019).
- Nunes, P. et al. STIM1 juxtaposes ER to phagosomes, generating Ca(2)(+) hotspots that boost phagocytosis. Current biology : CB 22, 1990-1997 (2012).
- Nunes-Hasler, P. et al. STIM1 promotes migration, phagosomal maturation and antigen cross-presentation in dendritic cells. Nat Commun 8, 1852 (2017).
- Levin, R. et al. Multiphasic dynamics of phosphatidylinositol 4-phosphate during phagocytosis. Mol Biol Cell 28, 128-140 (2017).
- Jeschke, A. et al. Phosphatidylinositol 4-phosphate and phosphatidylinositol 3-phosphate regulate phagolysosome biogenesis. Proceedings of the National Academy of Sciences of the United States of America 112, 4636-4641 (2015).
- Lee, W. & Suresh, M. Vaccine adjuvants to engage the cross-presentation pathway. Frontiers in immunology 13, 940047 (2022).
- Baljon, J.J. & Wilson, J.T. Bioinspired vaccines to enhance MHC class-I antigen cross-presentation. Curr Opin Immunol 77, 102215 (2022).
Poster image created with #StableDiffusion and #DreamStudioAI from an original seed.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
DNA repair and human disease
Publishing Model: Hybrid
Deadline: Oct 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in