Critical role of water structure around interlayer ions for ion storage in layered double hydroxides
Published in Materials

Layered materials have found various applications in areas such as water purification and energy storage. Various layered materials containing water have been identified. These include conventional clays and layered metal oxides and hydroxides. In a nanoconfinement, ions are typically hydrated and hydration structures determine the ion storage ability. Yet, studying the interplay between the ion configuration and hydration structure in the ion-storage mechanism has proven to be challenging. Even more challenging is analysing the changes in hydration structures that occur in a nanoconfinement during the ion transport process.
Our group hypothesized that the hydration structure determines the intermolecular interactions acting on the interlayer species and thus determines the "hardness" of the layered material itself. The quartz crystal microbalance with energy dissipation monitoring (QCM-D) method is an in situ measurement tool which has been commonly utilised for studying the dynamic deformation behaviours of water-containing soft matter accompanying viscoelastic changes (that is, hardness) in association with the mass transfer. The change in viscoelasticity and mass is simultaneously monitored by energy dissipation and frequency change, respectively. Therefore, we believe that the QCM-D method is a powerful tool to study the hydration structure during the ion transport process in layered materials.
In this study, layered double hydroxides (LDHs), a class of anionic clay minerals, were utilised as a model material due to their compatibility with the existence of hydrated ions and the rigid layer-stacking structure. First, two LDHs with different interlayer filling densities of ions were prepared with modulated host charge densities (Figure 1a). Next, the adsorption isotherm of dilute nitrate for these LDHs was analysed. A 24% decrease in the filling density increased the storage capacity by 300%. On this basis, the QCM-D method was studied to prove the dynamic structural change of LDHs upon nitrate storage. Both the LDHs exhibited nonlinear change in viscoelasticity by hardening at the beginning of the reaction and then softening as the reaction progressed (Figure 1b). The change was discreet for weakly charged LDHs. Finally, the results were combined with multimodal ex situ experimental results and theoretical calculations. The low filling density effectively facilitates the 2D hydrogen-bond networking structure in the interlayer water around nitrate ions along with minimal change in the layered structure, which leads to the high storage capacity, while the high filling density facilitates the 3D networking structure, requiring large expansion of interlayer distance and destabilising the LDHs.
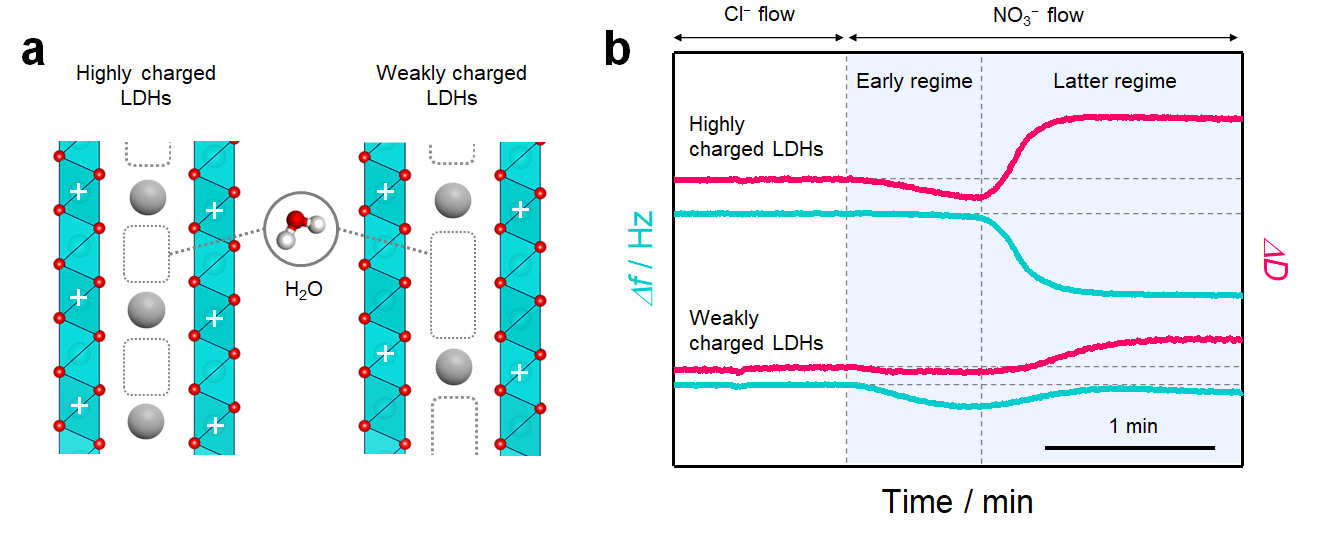
Figure. 1 | (a) Schematic of the interlayer structure in layered materials with different host charge densities. In the interlayer space, water molecules are incorporated into the pores that are not filled with ions. (b) QCM-D profiles for LDHs with different host charge densities showing the change in the frequency (Δf) and dissipation (ΔD).
Recently, understanding the water structure has been of great importance for not only ion storage layered metal oxides/hydroxides, but also van der Waals materials, MXenes, and conventional clays. Our results demonstrate that the ion configuration in the interlayer confinement enables precise control of the dynamic structural change in water, considerably improving the ion storage ability of the material. We expect that further tuning of the host materials will offer a new framework for the optimisation of ion storage-related functionalities. The result was recently published in Nature Communications titled "Critical role of water structure around interlayer ions for ion storage in layered double hydroxides."
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in