The question of what society should do – or not do – to address climate change featured prominently in the recent 2019 Canadian federal election. As always, this issue could be boiled down to concerns about economics: namely, are carbon taxes “worth it”? Canada, a geographically diverse country blessed in some places with abundant renewable (hydroelectric) energy but long reliant in others on non-renewable (petroleum-based) resources, has wrestled for some time with the idea of balancing environmental responsibility and economic activity. For example, in adjusting Canada’s emission targets from those mandated by the Kyoto Protocol to ones set out in the less strenuous Copenhagen Accord, then Environment Minister Jim Prentice cited concerns over ‘industrial competitiveness’ caused by more significant reductions.[1]
In this context, to promote the widespread adoption of sustainable energy or manufacturing technologies, the cost of the materials involved must be carefully considered. With respect to solar energy capture and conversion, dye-sensitized solar cells are an example of an alternative to silicon panel solar cells that are made from inexpensive, abundant semiconductor materials with low fabrication costs. The catch is that the electrode surface must be ‘sensitized’ towards light energy collection using a suitable light-absorbing dye. ‘Photosensitizers’ constructed from coordination complexes of precious metals like ruthenium and iridium are particularly useful in this respect, as such dyes can be designed to exhibit the long-lived charge-transfer (CT) excited states needed for efficient charge injection or for building up suitable concentrations of reactive excited states for synthesis. So, while significant strides have been made in reducing the costs associated with electrode materials, substrates and redox-mediators, the current state-of-the-art in dye performance still requires the use of scarcer, more expensive, transition metals. Replacing these with dyes based on iron, “the least expensive and most widely used metal” according to the 2019 USGS Mineral Commodity Summary [2], is therefore a tantalizing goal. Years of research, however, have delivered only a handful of iron coordination complexes with any sort of detectable excited state lifetimes.
Our group is interested in how ligand design can be used control the photophysical properties and reactivity of late transition metal coordination complexes. In particular, we have focused on benzannulated N-heterocyclic ligands that can defy conventional understanding of how extending a ligand’s pi-system might impact absorption/emission.[3,4] During these investigations, we found that installing ligands which pair benzannulated heterocycles with amido donors on iron(II) formed deep green coordination complexes with absorption profiles covering from the ultraviolet to the edge of the visible.
In collaboration with UManitoba colleagues Johan van Lierop and Rebecca Davis, we discovered the green colour is a result of the molecules representing an intermediate case of the ‘HOMO inversion’ model proposed computationally by Elena Jakubikova.[5] Intrigued by the panchromatic absorption of these Fe(II) complexes (but anticipating picosecond or shorter excited state lifetimes), we connected with experts in ultrafast spectroscopy, Clemens Burda and Charles Kolodziej at the Center for Chemical Dynamics of Case Western Reserve University, thanks to the help of Wenfang Sun at nearby North Dakota State University.
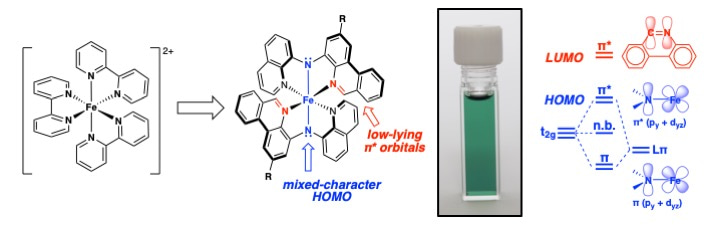
As it turned out, we didn’t need ultrafast resolution! The ligand design imbued these Fe(II) complexes with exceptionally long-lived (for Fe) nanosecond CT states. The ‘MLCT’-type nature of the excited state generated by photo-induced charge transfer from filled orbitals of mixed iron-amido character into energetically accessible, vacant heterocycle-based acceptor orbitals should be appropriate for electron injection into n-type semiconductors, complementing recently reported Fe(III) complexes with similarly long-lived LMCT excited states.[6] In addition, the panchromatic absorption of these complexes should enable a greater proportion of the solar spectrum to be harvested. Accordingly, we are now investigating the ability of these complexes to sensitize semiconductor surfaces in DSSC-type devices and to facilitate photo-induced electron transfer in homogeneous solutions.
You can read the full story here:
https://www.nature.com/articles/s41557-019-0357-z
[1] https://policyoptions.irpp.org/magazines/canadas-water-challenges/a-conversation-with-jim-prentice/ ; accessed November 14th, 2019.
[2] https://www.usgs.gov/centers/nmic/mineral-commodity-summaries ; accessed November 14th, 2019.
[3] Mondal, R., Lozada, I. B., Davis, R. L., Williams, J. A. G. & Herbert, D. E. Site-Selective Benzannulation of N-Heterocycles in Bidentate Ligands Leads to Blue-Shifted Emission from [(P^N)Cu]2(μ-X)2 Dimers. Inorg. Chem. 57, 4966-4978 (2018).
[4] Mandapati, P. et al. Luminescent Platinum(II) Complexes of N^N–^N Amido Ligands with Benzannulated N-Heterocyclic Donor Arms: Quinolines Offer Unexpectedly Deeper Red Phosphorescence than Phenanthridines. Inorg. Chem. 58, 14808-14817 (2019).
[5] Mukherjee, S., Torres, D. E. & Jakubikova, E. HOMO inversion as a strategy for improving the light-absorption properties of Fe(II) chromophores. Chem. Sci. 8, 8115–8126 (2017).
[6] Kjær, K. S. et al. Luminescence and reactivity of a charge-transfer excited iron complex with nanosecond lifetime. Science 363, 249-253 (2019).
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in