Diversity of organohalide respiring bacteria and reductive dehalogenases that detoxify polybrominated diphenyl ethers in E-waste recycling sites
Published in Earth & Environment

Globally, e-waste is one of the largest and fastest-growing waste streams [1]. Regulatory disparities have long made it more economical for wealthier countries to ship e-waste to the developing world for disposal, effectively turning some poorer regions into hazardous waste dumping grounds. Recognizing the inequality inherent in this practice, many governments have sought to limit the transshipment of e-wastes. Despite efforts to enforce legislation regarding the movement of e-wastes and monitoring efforts of numerous governmental agencies and NGOs, an estimated 82.6% of global e-waste flows remain poorly documented [2]. Much of this undocumented e-waste is either dumped, openly burned, or treated and recycled by unregulated, small-scale operators. As e-waste contains high concentrations of a variety of hazardous organic compounds, such as polybrominated diphenyl ethers (PBDEs), readily leach into soil and water when e-waste is buried or are dispersed into the atmosphere when e-waste is incinerated, leaving behind a severe environmental burden and posing health risks to human populations.
Until the implementation of multiple bans in the mid- to late-2000s, polybrominated diphenyl ethers (PBDEs) were extensively used as additive flame retardants in a variety of manufactured products, ranging from clothing and household appliances to consumer electronics and construction materials. Inadvertent release, transboundary displacement, and global migration have led to the nearly ubiquitous environmental deposition of PBDEs [3]. The dangers of PBDEs became apparent after the discovery of elevated levels of PBDEs in biota, most notably in human tissues and breast milk [4-7]. Awareness of the potential for PBDEs to harm human populations and fragile ecosystems led to the listing of tetra-, penta-, hexa-, hepta-, and deca-BDEs as persistent organic pollutants (POPs) by the United Nations Stockholm Convention and subsequent bans on usage [8]. Despite these efforts to curtail further release of PBDEs, public concern about the effects of legacy PBDE contamination persists and there are still some countries and regions where PBDE usage and manufacture are either not restricted or enforcement of regulations is lax [9].
PBDEs tend to partition into anoxic environmental compartments, and were initially thought to be resistant to biotransformation due to the slow rate of natural attenuation observed under anoxic conditions. Additionally, the natural attenuation of octa-, nona-, and deca-PBDEs that was observed most frequently produced partially debrominated tetra- and penta-BDEs, which are more environmentally mobile and are highly toxic to biological populations. Long-held assumptions about anaerobic microbial degradation of PBDEs were challenged by Prof. He Jianzhong in a 2006 report of partial debromination of PBDEs mediated by an anaerobic microbial culture [10]. This first report spurred research seeking to identify anaerobes that could debrominate congeners characteristic of legacy PBDE contamination to develop sustainable approaches for in situ bioremediation. However, it was not until 10 years later that the first anaerobic bacterium to exhibit complete debromination of tetra- and penta-BDEs were isolated by Dr. Ding Chang [11], who also described a set of three reductive dehalogenases that catalyze debromination of distinct PBDE congeners. Three years after this, I isolated a tetra- and penta-BDE debrominating bacterium that employs a different set of PBDE-dehalogenases, indicating that anaerobic PBDE-debromination is an infrequent, but not unique, phenotype among anaerobic organohalide respiring bacteria (OHRB) [12]. Interestingly, both of these PBDE-debrominating OHRB were isolated from soil samples collected from sites where waste electronics and electronic equipment (e-waste) were recycled or landfilled, suggesting that the capacity for PBDE-debromination could have emerged in situ, allowing some OHRB to harvest energy for growth from anthropogenic organohalides that had leached into the soils from e-waste.
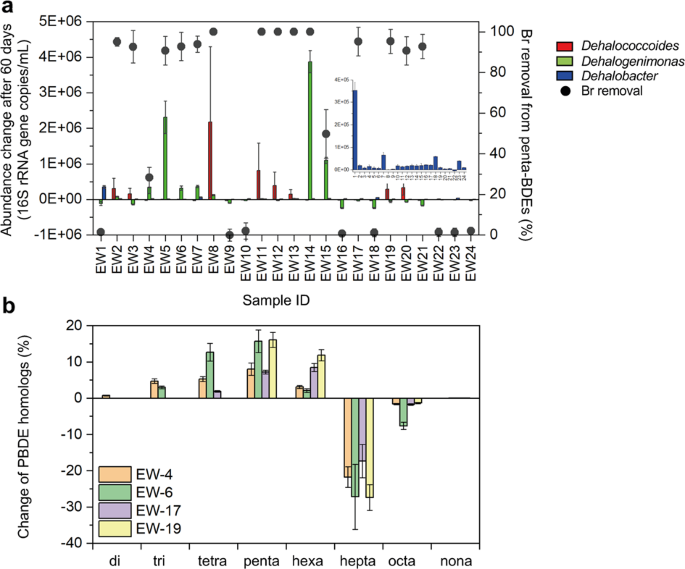
To investigate the prevalence of anaerobic PBDE-debromination among the microbial communities at e-waste sites, we established microcosms using subsurface soil samples from several geographically distinct e-waste recycling sites. Per our hypothesis that anaerobic PBDE-debromination could infrequently emerge in these communities, a small fraction of microcosms was able to partially debrominate more highly substituted PBDEs (i.e., nona- through hexa-BDEs). However, complete debromination of penta- and tetra-BDEs was observed in an unexpectedly high number of the microcosms after two months’ incubation. Following selective enrichment, some penta- and tetra-BDE debrominating microcosms could completely debrominate the same concentration of PBDEs in as little as 8 days. The presence of PBDE debromination in more than two-thirds of the microcosms prompted me to examine whether this phenotype had been overlooked in other OHRB. These experiments found previously undescribed PBDE debromination in several Dehalococcoides isolates, from which we were able to identify additional PBDE-dehalogenases. Finally, the practical application of this growing suite of PBDE-dehalogenases as biomarkers of debromination potential in environmental samples was evaluated.
The findings from this study indicate that in situ biological debromination of PBDEs is both more widespread and potentially more rapid than earlier work suggests, particularly of lesser brominated PBDEs. While the current study did not yield promising candidates for bioremediation of the most highly substituted PBDEs, the potential shown for the development of sustainable solutions to mitigate the hazards of penta- and tetra-BDEs at e-waste sites is a step in the right direction.
References:
- Bacher J, Dams Y, Duhoux T, Deng Y, Teittinen T, Mortensen LF (2020). Electronic products and obsolescence in a circular economy. European Topic Centre Waste and Materials in a Green Economy: Belgium.
- Forti V, Bald CP, Kuehr R, Bel G (2020). The Global E-waste Monitor 2020: Quantities, flows and the circular economy potential. United Nations University/United Nations Institute for Training and Research, International Telecommunication Union, and International Solid Waste Association: Bonn, Geneva and Rotterdam.
- McGrath TJ, Ball AS, Clarke BO (2017). Critical review of soil contamination by polybrominated diphenyl ethers (PBDEs) and novel brominated flame retardants (NBFRs); concentrations, sources and congener profiles. Environmental Pollution (Barking, Essex : 1987) 230: 741-757.
- de Boer J, Wester PG, Klamer HJ, Lewis WE, Boon JP (1998). Do flame retardants threaten ocean life? Nature 394: 28-29.
- Boon JP, Lewis WE, Tjoen ACMR, Allchin CR, Law RJ, De Boer J et al (2002). Levels of polybrominated diphenyl ether (PBDE) flame retardants in animals representing different trophic levels of the North Sea food Web. Environmental Science & Technology 36: 4025-4032.
- Norstrom RJ, Simon M, Moisey J, Wakeford B, Weseloh DV (2002). Geographical distribution (2000) and temporal trends (1981-2000) of brominated diphenyl ethers in Great Lakes hewing gull eggs. Environmental Science & Technology 36: 4783-4789.
- Zhu LY, Hites RA (2004). Temporal trends and spatial distributions of brominated flame retardants in archived fishes from the Great Lakes. Environmental Science & Technology 38: 2779-2784.
- United Nations Environment Programme U (2009). Stockholm convention on persistent organic pollutans (POPs): Geneva, Switzerland.
- Abbasi G, Li L, Breivik K (2019). Global Historical Stocks and Emissions of PBDEs. Environmental Science & Technology 53: 6330-6340.
- He J, Robrock KR, Alvarez-Cohen L (2006). Microbial reductive debromination of polybrominated diphenyl ethers (PBDEs). Environmental Science & Technology 40: 4429-4434.
- Ding C, Rogers MJ, Yang KL, He JZ (2017). Loss of the ssrA genome island led to partial debromination in the PBDE respiring Dehalococcoides mccartyi strain GY50. Environmental Microbiology 19: 2906-2915.
- Zhao S, Rogers MJ, Cao L, Ding C, He J (2021). Identification of reductive dehalogenases that mediate complete debromination of penta- and tetra-brominated diphenyl ethers in Dehalococcoides. Applied and Environmental Microbiology: Aem0060221.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in