
Why should we care about cyanobacteria and biofilms?
Cyanobacteria play a pivotal role in our planet’s ecology – they are responsible for ~25% of carbon dioxide convertion into organic material and the concomitant oxygen release globally. Often these photosynthetic prokaryotes are found in communal assemblages known as biofilms. Despite the environmental prevalence and the industrial implications of phototrophic biofilms, the molecular mechanisms underlying cyanobacterial biofilm development were overlooked for many years. Elucidation of biofilm formation in cyanobacteria will fill the knowledge gap concerning communal behaviour of these photosynthetic prokaryotes. Moreover, such information will pave the way for mitigation of harmful cyanobacterial biofilms (e.g., those promoting biofouling on submerged objects) or promotion of beneficial biofilms (e.g., in sewage purification systems and for biomass harvesting in the biofuel industry).
How do we study cyanobacterial biofilms and what did we find?
Inequality in a cyanobacterial community. Our interest in cyanobacterial biofilms was triggered by a serendipitous finding of a knock out mutant that formed biofilms, in contrast to the planktonic wild-type. In this study we employed the biofilm-forming mutant to get a peek into the communal behaviour of these single-cell organisms. Briefly, Eli Zecharia, who recently received his PhD, demonstrated that only a quarter of the cell population expresses at high levels genes encoding proteins that enable biofilm formation, referred to as EbfG. Almost all cells, however, are assembled in the biofilm (Fig.1).
 |
Fig. 1: Only ~25% of the cells express biofilm-enabling proteins, yet the majority of the cell population is found within the biofilm. |
Alona Frenkel, a former MSc student in the lab, showed that one of these proteins, EbfG4, is localized to the cell-surface (Fig. 2a) as well as to the biofilm matrix (Fig. 2b). Likely, this protein is initially presented on the cell surface and serves as an adhisin (a cell-surface component that facilitates adhesion to other cells or to surfaces) and later is shed to be part of the extracellular matrix.
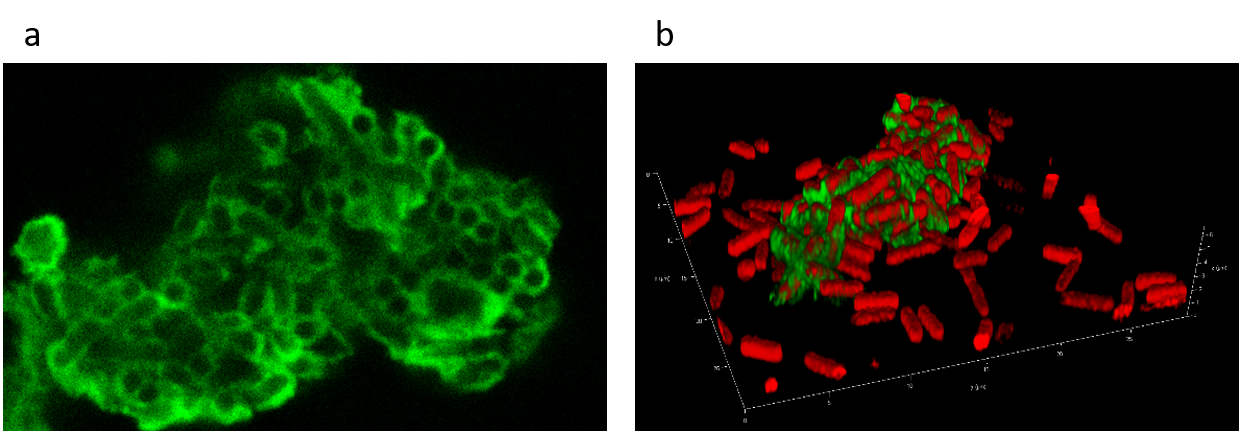 Fig. 2: Imaging by fluorescence microscopy. EbfG4 is localized to the cell surface (a) and in the extracellular matrix (b). Red – autofluorescence; Green – EbfG4.
Fig. 2: Imaging by fluorescence microscopy. EbfG4 is localized to the cell surface (a) and in the extracellular matrix (b). Red – autofluorescence; Green – EbfG4.
Following advice of my colleague, Karl Forchhammer from Tübingen University, Germany, I got in touch with Daniel Gómez-Pérez from the Eric Kemen lab at Tübingen, an expert in plant associated biofilms. Fortunately, Daniel was not only enthusiastic about implementing a bioinformatics approach to check if the EbfG proteins tend to fold as amyloids, but also followed up experimentally on his initial in silico analysis and demonstrated that EbfG1-3 form protein fibres. Together, the data suggest a beneficial ‘division of labour’ during biofilm formation, where only some of the cells allocate resources to produce matrix proteins – ‘public goods’ that support robust biofilm development by the majority of the cells (Fig. 1). It is tempting to speculate that cells at the periphery of the biofilm, which are exposed to light and are thus better energized compared to the shaded cells within the biofilm, are those that highly express the biofilm-enabling genes and provide the public goods.
Self-regulation of biofilm formation by intercellular communication. Our previous studies revealed the operation of a self-suppression mechanism that depends on an inhibitor secreted by wild-type cells, which supresses expression of the biofilm-enabling genes and dictates planktonic growth. In this study, we provide evidence for inhibitor activity at an early growth stage and its gradual accumulation along the exponential growth phase in correlation with cell density (Fig. 3). Data, however, do not support a threshold-like phenomenon known for quorum-sensing in non-photosynthetic bacteria. The chemical nature of the inhibitor is as yet enigmatic; however, these observations provide an important lead for follow up studies of cyanobacterial intercellular communication – a subject that is in its infancy.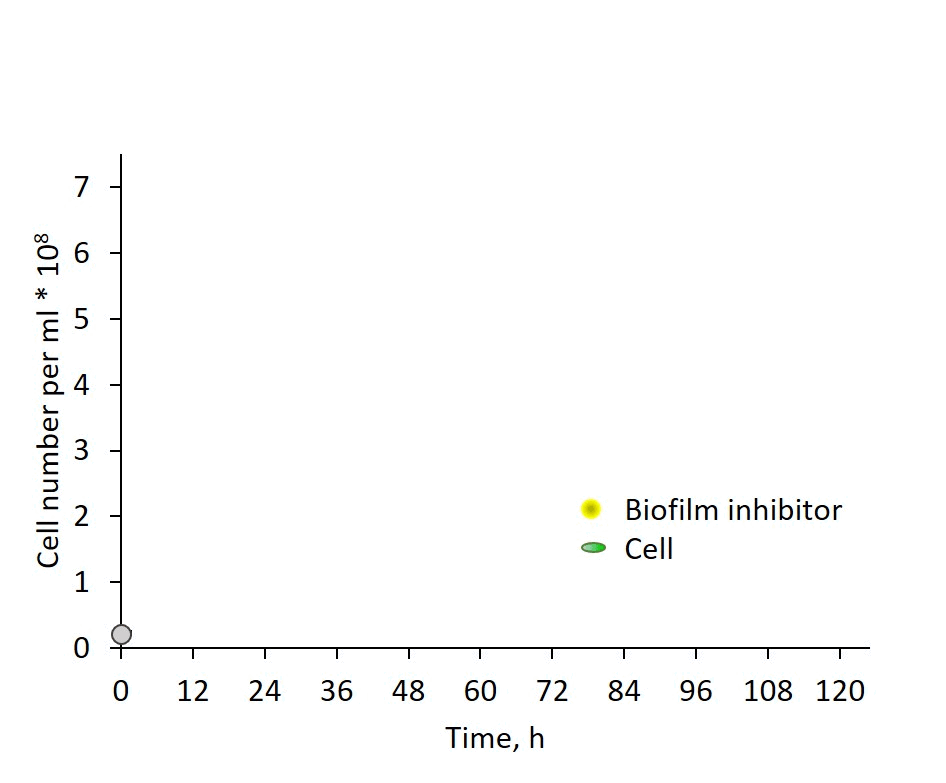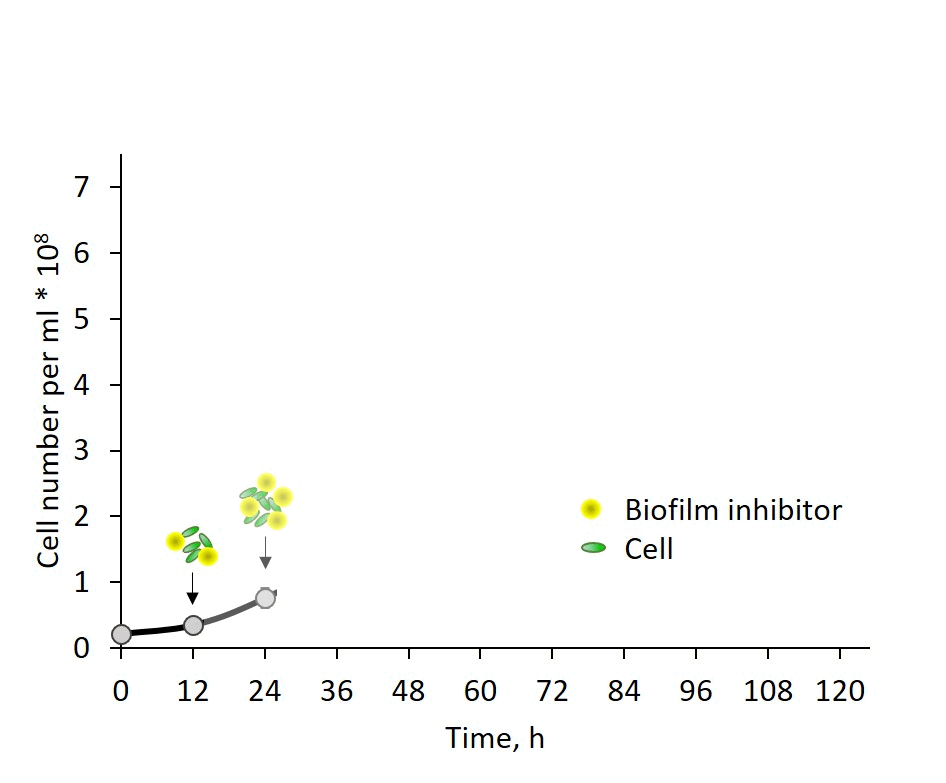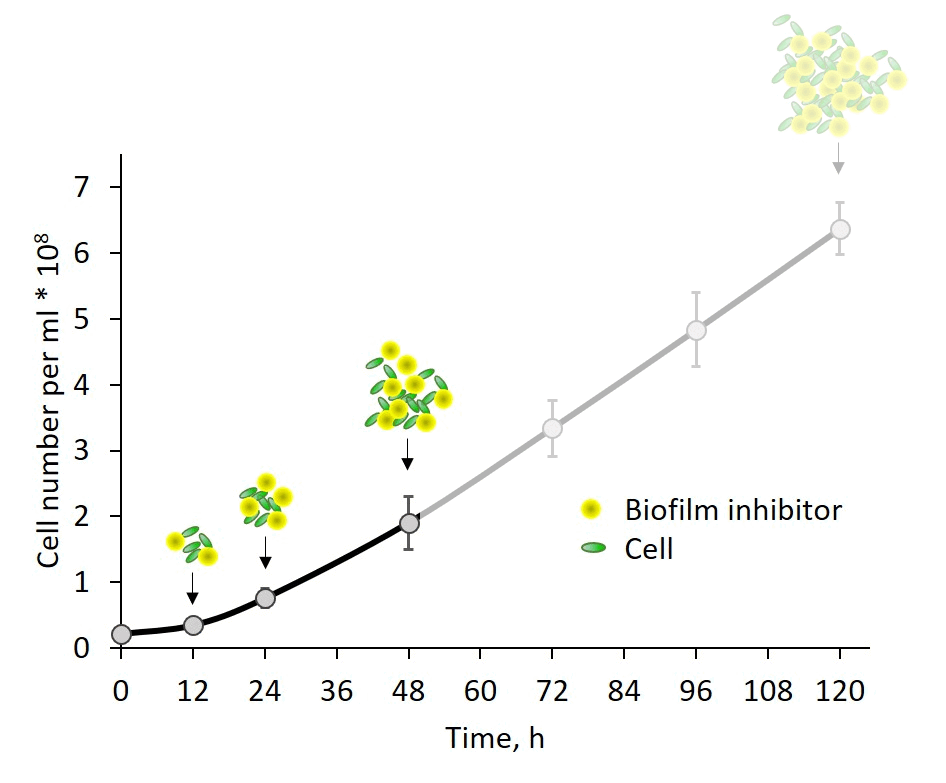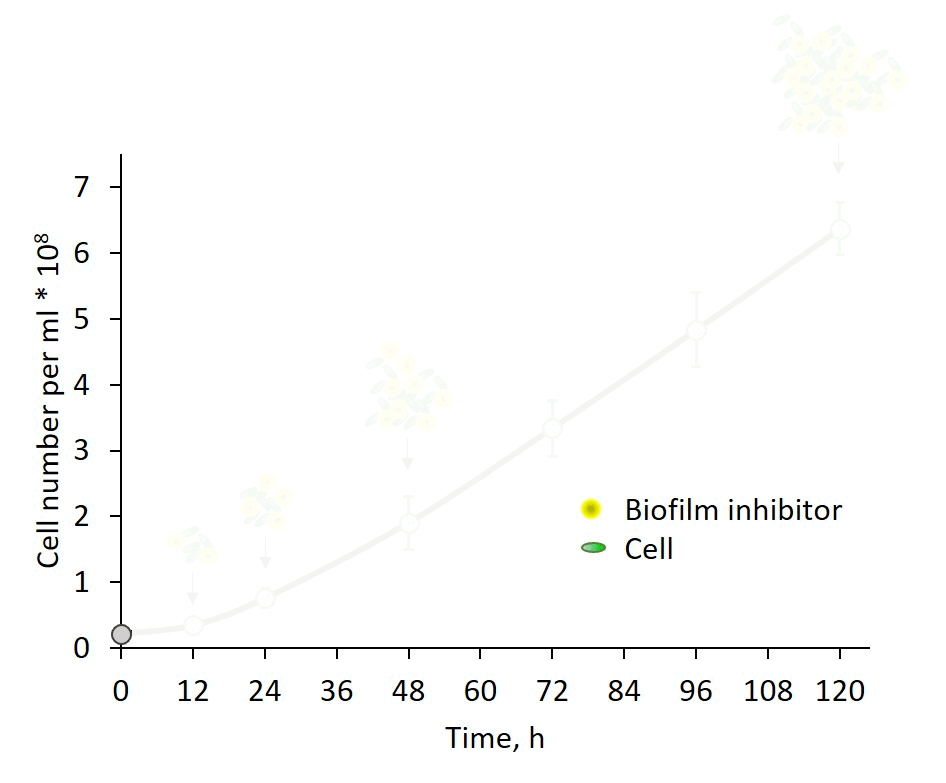
Fig. 3: Biofilm regulation by intercellular communication is manifested by inhibitor activity detectable already at very early growth stage.
Follow the Topic
-
npj Biofilms and Microbiomes
The aim of this journal is to serve as a comprehensive platform to promote biofilms and microbiomes research across a wide spectrum of scientific disciplines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Microbial endocrinology
Publishing Model: Open Access
Deadline: Oct 21, 2026
Microbiome and energy metabolism
Publishing Model: Hybrid
Deadline: Dec 06, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
Very inspiring discovery! Congratulations Rakefet and the research team!