Efficient synthesis and evolution of 2’-modified nucleic acid chemistries found in FDA-approved nucleic acid therapeutics
Published in Chemistry

Nucleic acid drugs (NADs) have now begun entering the clinic and almost twenty have been approved by the FDA or EMA1. Among them are RNaseH-recruiting antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs), both of which lead to the degradation of their RNA target as well as ASOs that block certain mRNA splicing sites (splice-switching oligonucleotides). Recently, SARS-CoV-2 mRNA vaccines have joined the group. In addition, a single aptamer had been approved for clinical use.
The clinical activity of all of these NADs depends critically on one or more chemically modified nucleotides, which are generally inserted by solid-phase phosphoramidite chemistry, which is efficient but hard to scale up to large numbers of sequence or chemistry variants. Both screening and discovery of NADs might be accelerated by the development of tools for the rapid synthesis of many variants. One such tool are polymerases, which can be engineered to enable the encoded, high-fidelity synthesis of chemically divergent nucleic acid polymers (xeno-nucleic acids (XNAs))2,3. Modified XNA nucleotides can confer beneficial physicochemical and pharmacological properties like high antisense-binding affinity and resistance to nucleases to the modified oligonucleotide while retaining the natural base-pairing preferences and helical geometry4. Furthermore, the advent of mRNA vaccines has shown the potential utility and power of polymerases in the NAD manufacturing.
Polymerases are also of particular importance for NADs like nucleic acid ligands (aptamers) or catalysts that cannot be sequence-designed like ASOs but require the screening or selection of large random-sequence repertoires for discovery. For this, XNA polymerases are key enabling tools for iterative synthesis and reverse transcription of repertoires comprising modified nucleic acid chemistries.
Many of the above-mentioned nucleic acid drugs comprise nucleotides modified at the 2’-position to confer biostability, high antisense-binding affinity and favourable physicochemical, pharmacological and immunological properties4. However, the increased steric bulk of some of these modifications makes them difficult or impossible to be incorporated by natural polymerases as the large 2’-modifications exhibit steric clashes with the internal primer-template-binding funnel of the polymerase. Indeed, it has been known for some time that steric control is one of the most important mechanisms by which polymerases impart specificity and selectivity—for example, replicative DNA polymerases exclude ribonucleotides from incorporation into the genome by an active site steric gate residue5 despite the up to 100-fold higher cytoplasmic concentration of e.g., ATP compared to dATP6.
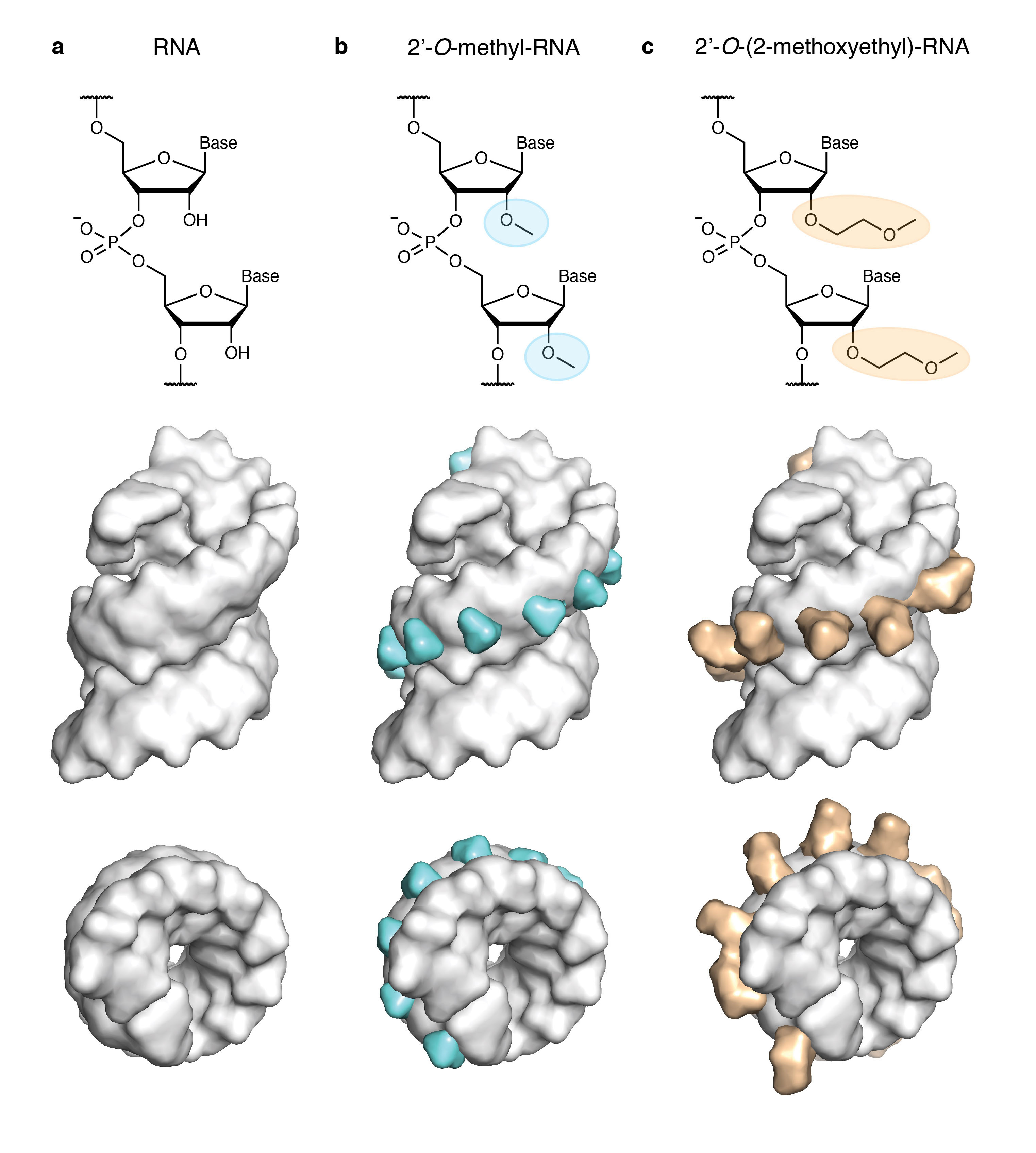
Fig. 1 The nucleic acid chemistries pertinent to this study. a) RNA, b) 2’-O-methyl-RNA (2’OMe-RNA), and c) 2’-O-(2-methoxyethyl)-RNA (MOE-RNA). Shown are chemical representations of the three structures (top row) and space-filling representations based on the X-ray structure of an MOE-RNA duplex (PDB ID 468D) viewed from the side (centre row) and the top (bottom row) with 2’OMe groups in cyan and MOE groups in orange. Note the increasing steric demand of the 2’-modification protruding into the minor groove.
One of the candidate 2’-modifications, 2’-O-methyl (2’OMe), had attracted considerable attention and polymerase engineering efforts had yielded polymerases with increasingly efficient synthesis activity for 2’OMe-RNA7–12. Another important modification used in antisense oligonucleotide therapeutics is the related but much bulkier 2’-O-(2-methoxyethyl)-RNA (MOE-RNA), which had thus far been impossible to synthesise enzymatically.
We set out to engineer high-efficiency polymerase tools enabling rapid and efficient synthesis of both 2’OMe- and MOE-RNA and unlocking in vitro evolution experiments in these nucleic acid chemistries. Building on a B-family (polB) polymerase from the hyperthermophilic archaeon Thermococcus gorgonarius previously engineered in our lab for primer-dependent RNA synthesis13,14, we used semi-rational structure-based engineering of a static polymerase model derived from the structure of the closely related DNA polymerase from T. kodakarensis15 to engineer a variant that would be capable of 2’-modified RNA synthesis. 2’-modifications like 2’OMe protrude into the minor groove, and in our model, we observed multiple steric clashes between the 2’-modifications and polymerase residues proximal to or contacting the nascent (primer) strand. Interestingly, some of these residues are part of previously-described motifs16,17 that are highly conserved in polB polymerases across all phyla. When mutated to amino acids with smaller side chains, two of these residues showed a striking synergistic increase in 2’OMe-RNA synthesis activity. Therefore, we termed these two conserved positions a nascent strand steric gate, which complements the classic steric gate.
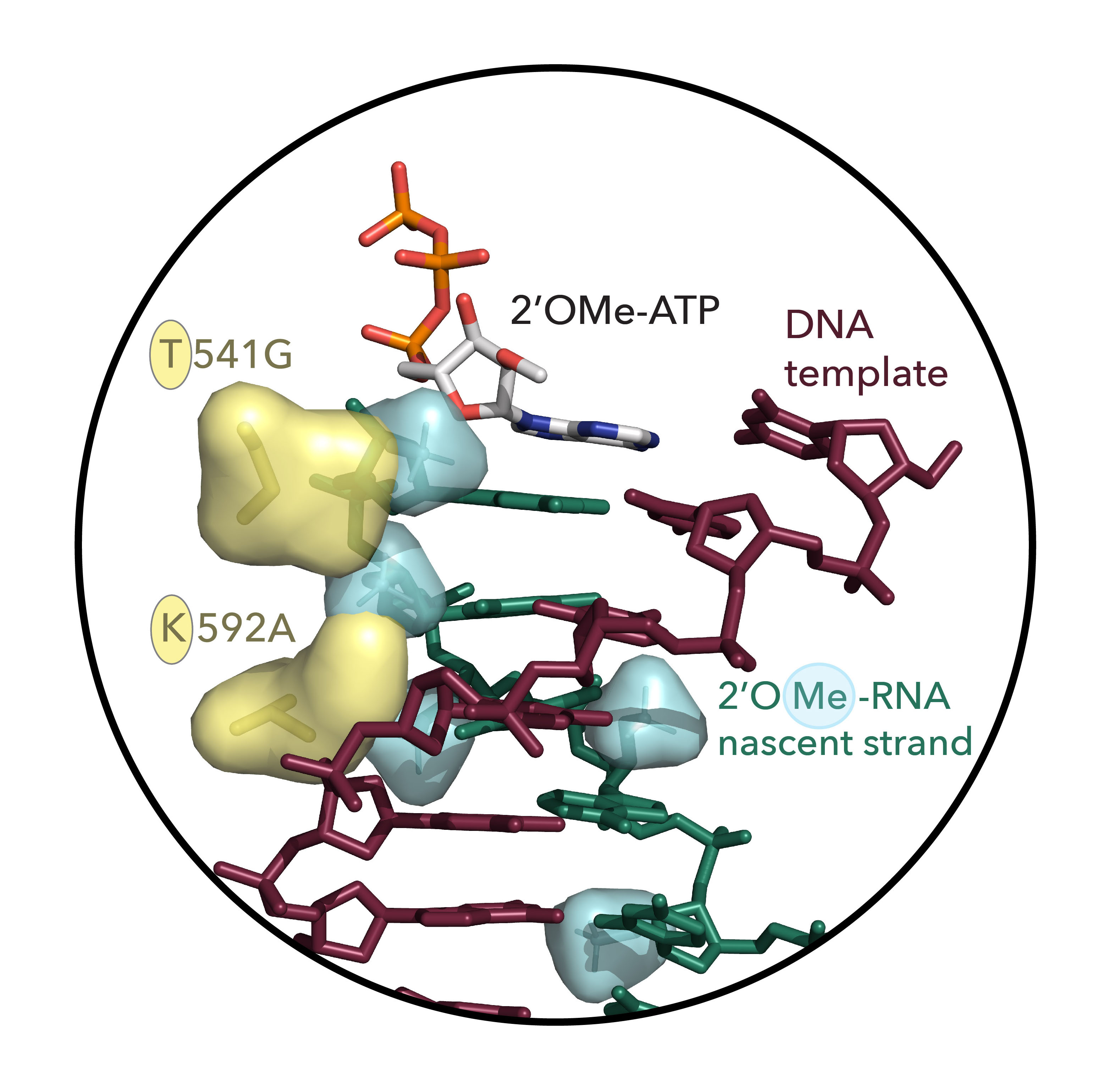
Fig. 2 The two-residue nascent strand steric gate identified in this study. Shown is a structural model of the active site of KOD DNA polymerase (PDB ID 5OMF) with DNA template strand (brown-red), active site 2’OMe-ATP, and 2’OMe-RNA nascent strand (green) with 2’-methoxy groups on nascent strand shown as space-filling envelope (cyan) and key steric gate mutations T541G and K592A displayed in yellow (sticks) with wild-type side-chain residues shown as space-filling envelope highlighting the reduction in steric bulk.
The resulting polymerase double mutant “2M” exhibits not only very efficient 2’OMe-RNA synthesis activity, but together with a previously-described 2’OMe-RNA reverse transcriptase from our lab18, enabled the in vitro evolution of the first all-2’OMe-RNA enzymes. These were selected to cleave RNAs with single-nucleotide discrimination, allowing for the allele-specific cleavage of oncogenic mutant mRNA of KRas and β-catenin, while leaving the wild-type allele intact.
Despite the much larger steric bulk of the MOE modifications, polymerase 2M also enabled efficient MOE-RNA synthesis, giving access to MOE-RNA libraries. For the first time, this opens the door to the first MOE-RNA SELEX. In order to show whether the bulky MOE-RNA modification is a viable scaffold for the elaboration of aptamers and whether it is amenable to fold productively into three-dimensional protein-binding shapes, we modified an existing 2’OMe-RNA aptamer specific for Vascular Endothelial Growth Factor (VEGF)10 with increasingly higher loads of MOE-RNA. Interestingly, a 50% substitution variant in which all 2’OMe-A, 2’OMe-C had been replaced by MOE-A, MOE-C still bound VEGF with identical affinity compared to the parent aptamer. This suggests that the first MOE-RNA aptamer is within reach.
Taken together, our study provides a synthetic tool for the efficient and rapid synthesis and evolution of the closely-related modified nucleic acid chemistries 2’OMe-RNA and MOE-RNA, which are of high interest in the field of nucleic acid therapeutics due to their biostability and high-antisense binding affinity. The 2M polymerase promises to provide a convenient synthetic route for the rapid elaboration and discovery of antisense oligonucleotides, aptamers, and nucleic acid enzymes in these chemistries approved for human use.
References
- Yamada, Y. Nucleic Acid Drugs-Current Status, Issues, and Expectations for Exosomes. Cancers 13, 5002 (2021).
- Pinheiro, V. B. et al. Synthetic genetic polymers capable of heredity and evolution. Science 336, 341–4 (2012).
- Freund, N., Fürst, M. J. L. J. & Holliger, P. New chemistries and enzymes for synthetic genetics. Curr. Opin. Biotechnol. 74, 129–136 (2022).
- Wan, W. B. & Seth, P. P. The Medicinal Chemistry of Therapeutic Oligonucleotides. J Med Chem 59, 9645–9667 (2016).
- Brown, J. A. & Suo, Z. Unlocking the Sugar “Steric Gate” of DNA Polymerases. Biochemistry 50, 1135–1142 (2011).
- Nick McElhinny, S. A. et al. Abundant ribonucleotide incorporation into DNA by yeast replicative polymerases. Proc. Natl. Acad. Sci. U. S. A. 107, 4949–4954 (2010).
- Chelliserrykattil, J. & Ellington, A. D. Evolution of a T7 RNA polymerase variant that transcribes 2’-O-methyl RNA. Nat Biotechnol 22, 1155–60 (2004).
- Ibach, J. et al. Identification of a T7 RNA polymerase variant that permits the enzymatic synthesis of fully 2’-O-methyl-modified RNA. J Biotechnol 167, 287–95 (2013).
- Meyer, A. J. et al. Transcription yield of fully 2’-modified RNA can be increased by the addition of thermostabilizing mutations to T7 RNA polymerase mutants. Nucleic Acids Res 43, 7480–8 (2015).
- Burmeister, P. E. et al. Direct in vitro selection of a 2’-O-methyl aptamer to VEGF. Chem Biol 12, 25–33 (2005).
- Chen, T. et al. Evolution of thermophilic DNA polymerases for the recognition and amplification of C2’-modified DNA. Nat Chem 8, 556–62 (2016).
- Hoshino, H., Kasahara, Y., Kuwahara, M. & Obika, S. DNA Polymerase Variants with High Processivity and Accuracy for Encoding and Decoding Locked Nucleic Acid Sequences. J Am Chem Soc 142, 21530–21537 (2020).
- Cozens, C., Pinheiro, V. B., Vaisman, A., Woodgate, R. & Holliger, P. A short adaptive path from DNA to RNA polymerases. Proc Natl Acad Sci U A 109, 8067–72 (2012).
- Cozens, C. et al. Enzymatic Synthesis of Nucleic Acids with Defined Regioisomeric 2’-5’ Linkages. Angew Chem Int Ed Engl 54, 15570–3 (2015).
- Kropp, H. M., Betz, K., Wirth, J., Diederichs, K. & Marx, A. Crystal structures of ternary complexes of archaeal B-family DNA polymerases. PLoS One 12, e0188005 (2017).
- Redrejo-Rodriguez, M. et al. Primer-Independent DNA Synthesis by a Family B DNA Polymerase from Self-Replicating Mobile Genetic Elements. Cell Rep 21, 1574–1587 (2017).
- Blasco, M. A., Mendez, J., Lazaro, J. M., Blanco, L. & Salas, M. Primer terminus stabilization at the phi 29 DNA polymerase active site. Mutational analysis of conserved motif KXY. J Biol Chem 270, 2735–40 (1995).
- Houlihan, G. et al. Discovery and evolution of RNA and XNA reverse transcriptase function and fidelity. Nat Chem 12, 683–690 (2020).
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in