Enantiomeric excess on-the-spot
Published in Chemistry
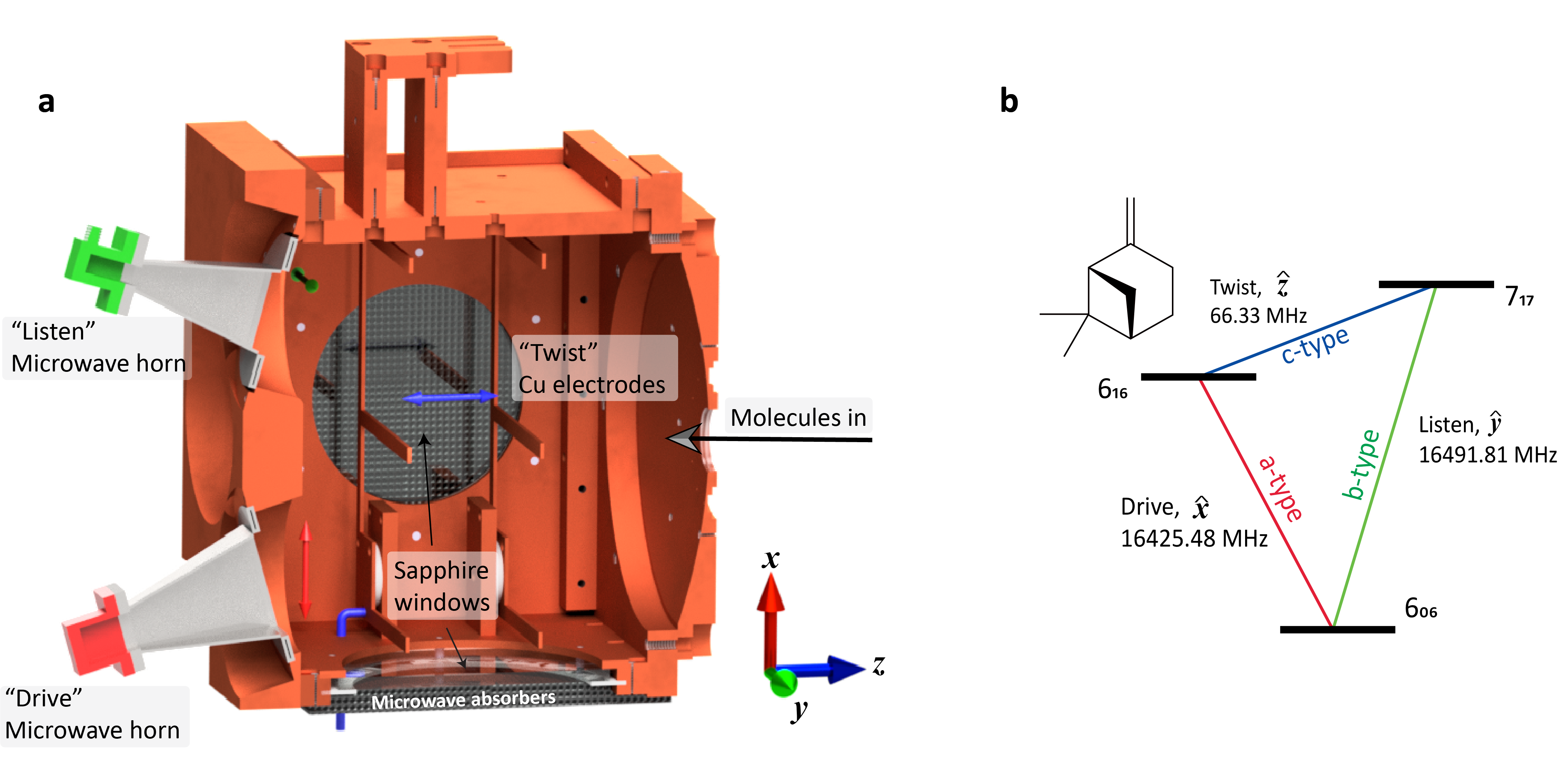
We introduce a chirality detection method using microwave spectroscopy that provides direct proof of chiral molecules in enantiomeric excess without any prior chemical knowledge of the sample. To access that information, all it takes is the acquisition of a new type of microwave spectrum, a three-wave mixing spectrum.
What is a three-wave mixing spectrum?
Three-wave mixing spectra combine two methods: chirped-pulse FT microwave spectroscopy for broadband excitation and microwave three-wave mixing for chirality detection. A three-wave mixing spectrum is a phase and polarization sensitive 2D microwave spectrum.
This complexity at the hardware level does not translate to the end result where no further analysis or expertise knowledge is required to access the chiral information. The information on the presence of species in enantiomeric excess is available on-the-spot.
Phase and polarization sensitivity allows for careful cancellation schemes that eliminate all spectroscopic lines that belong to 1) non-chiral species like ethanol or benzyl alcohol or 2) chiral species that are racemates (R and S are in equal parts). Once we get the spectrum, proof (or not) of enantiomeric excess is straightforward via the presence ( or absence) of any rotational lines.
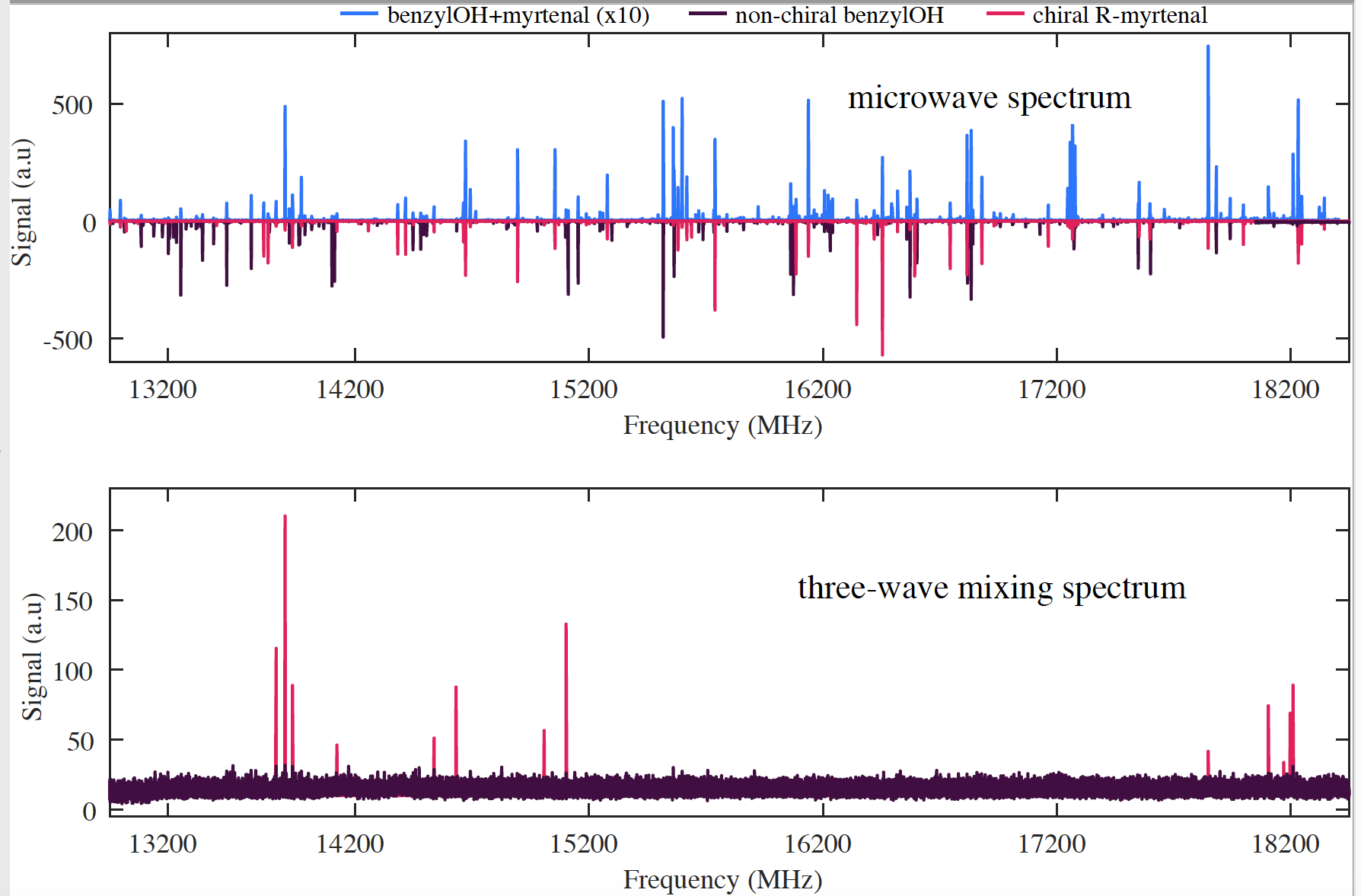
Why is this useful?
- Enantiomeric excess is a signature of life. In 2016, the first chiral molecule, propylene oxide, was detected in space [1] but current methods cannot test if molecular samples in space appear as racemates or in enantiomeric excess.
- New analytical capabilities for asymmetric synthesis and the broad search of chiral catalysts. Inside a one-pot synthesis reaction flask, there are: reactants, solvents, products, by-products, and catalysts. Since any separation or purification of the sample is unnecessary for analysis, a comparison between the three-wave mixing spectrum before and after the reaction can identify any new chiral products, in enantiomeric excess, produced.
- Access to the “unknown”. A spectrum is the most accurate molecular fingerprint, but spectral assignment -especially for structure and identification- is a difficult and time-consuming task. Three-wave mixing spectra provide proof of chiral species in excess skipping the assignment part but without compromising the rich spectral information. This allows access to chiral information of unassigned, transient, or novel exotic species.
- Spectra vs Numbers. Polarimetry has been the go-to tool for chemists interested in chiral molecules. However, for mixtures, measurements can be inconclusive, as the sum of the angles for multiple components can cancel each other out. Equal amounts of neat (-)-beta-pinene, (S)-carvone, and (R)-fenchone would have a total specific rotation of +7 degrees which carries significantly less information than a spectrum. In the paper, we show the three-wave mixing spectrum of a mixture of terpenes that contains information on the chirality, identity and structure of each species.
For more information, you can read more about our work published recently in Communications Chemistry : Koumarianou, G., Wang, I., Satterthwaite, L. et al. Assignment-free chirality detection in unknown samples via microwave three-wave mixing. Commun Chem 5, 31 (2022).
Other References
[1]McGuire, B. A. et al. Discovery of the interstellar chiral molecule propylene oxide (CH3CHCH2O).267
Science 352, 1449–1452 (2016).
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Experimental and computational methodology in structural biology
Publishing Model: Open Access
Deadline: Jul 31, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in