Estimating photoreceptor cell survival using clinical images
Published in Healthcare & Nursing

Limitations to imaging human eyes
Your ophthalmologist's office is filled with cameras, scanners, and other devices. These instruments enable your physician to record changes in your retina, the light-sensitive tissue lining the back of your eye. One of these devices is an optical coherence tomography (OCT) system. Unlike conventional cameras, which capture information in two dimensions, OCT systems capture information in three dimensions, somewhat like an ultrasound. OCT systems produce these images by rapidly tracing a beam of light across the retina. The retina comprises layers of distinct neural cells, each with specialized functions. These layers vary by how much light they reflect to the OCT system. The system converts the pattern of reflected light into a set of images, with different layers appearing brighter or darker on the image. While OCT systems have revolutionized eye care, they still have some important limitations. Commercial OCT systems, for instance, cannot distinguish between the most important types of light-sensitive cells in the retina, the rod and cone photoreceptor cells.
Rod and cone photoreceptor cells enable complementary visual functions, are distributed across the retina in different patterns, and give rise to distinctive symptoms in retinal disease. Rod photoreceptor cells enable you to navigate from your bed to the bathroom in dim light and to notice a fast-moving objects in your peripheral vision, like an incoming baseball or a songbird in flight. Cone photoreceptor cells provide color vision. Because many cones are densely packed in the center of your retina, these cells also let you read fine print and recognize faces. Many retinal diseases lead to the death of photoreceptor cells, but rods often die at different rates than cones. To develop successful treatments for these diseases, we need to ways to quantify the relative of each photoreceptor type at different stages of disease. As a step toward this goal, we developed a computational model to estimate the surviving percentage of rod and cone photoreceptors using OCT images.1
A computational reference model for estimating photoreceptor cell survival
Our computational model combines publicly-available data,2 which describes the average density and distribution of rod and cone photoreceptor cells in humans, with OCT images from an healthy adult. The model lets us make quantitative predictions about how the loss of rod and cone photoreceptors reduces the thickness of specific retinal layers. To illustrate how the model works, we built an interactive simulation shown in Figure 1.
Interactive Simulation
Figure 1 | A computational model for estimating rod and cone photoreceptor cell survival. The optical coherence tomography system captures retinal images like the one above. After dividing the image into distinct layers, our model allows the user to change the percentage of surviving rod and cone photoreceptor cells. By adjusting the sliders, you can see how the model reduces the thickness of the layers representing the photoreceptor cells but leaves the other neural tissue unchanged. The interactive model and code are available here.
The bodies of the photoreceptors cells occupy the outer nuclear layer (ONL). By applying the same expected cell survival values to multiple images from a single OCT scan, the reference model can generate three-dimensional topographic thickness maps, as shown in Figure 2A. By averaging the thickness values in two regions of the retina (Figure 2B-C), we can translate measurements of retinal layer thicknesses into estimates of cell survival (Figure 2D). Using OCT data from a set of patients with Stargardt disease and healthy (control) individuals,3 we can estimate the degree to which rods and cones are survive in the patients (Figure 2E).
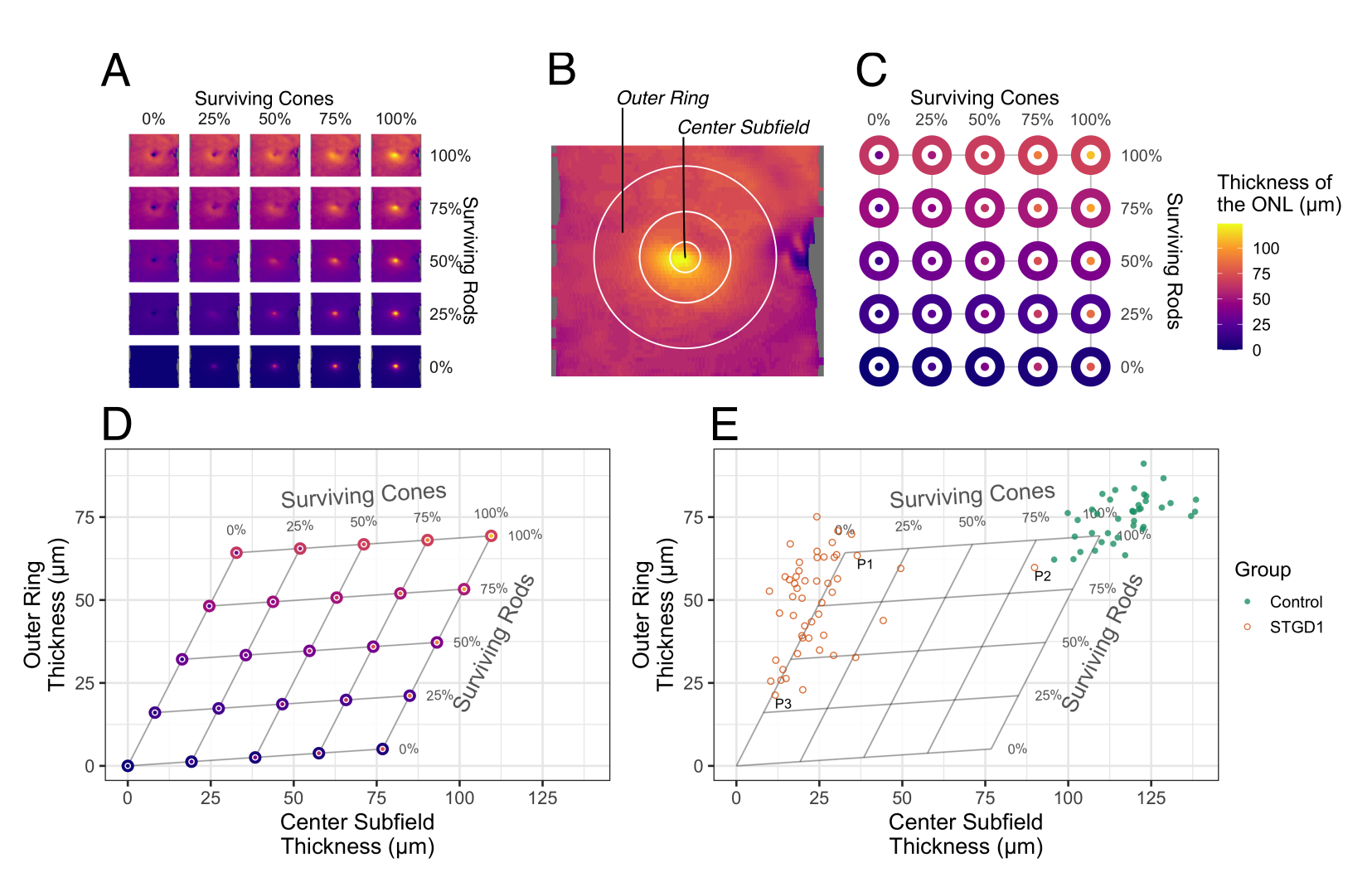
Figure 2 | The computational reference model translates layer thickness values from two regions of the retina into expected survival of rod and cone photoreceptor cells.
Using the model, we assessed data from our previous publication on a group of patients with Stargardt disease,3 one of the most common forms of inherited retinal disease in the United States. OCT images from three different patients are shown in Figure 3A, B, and C. We could also use the model to visualize the effect of disease on another OCT from a healthy eye (Figure 3A'', B'', and C'').
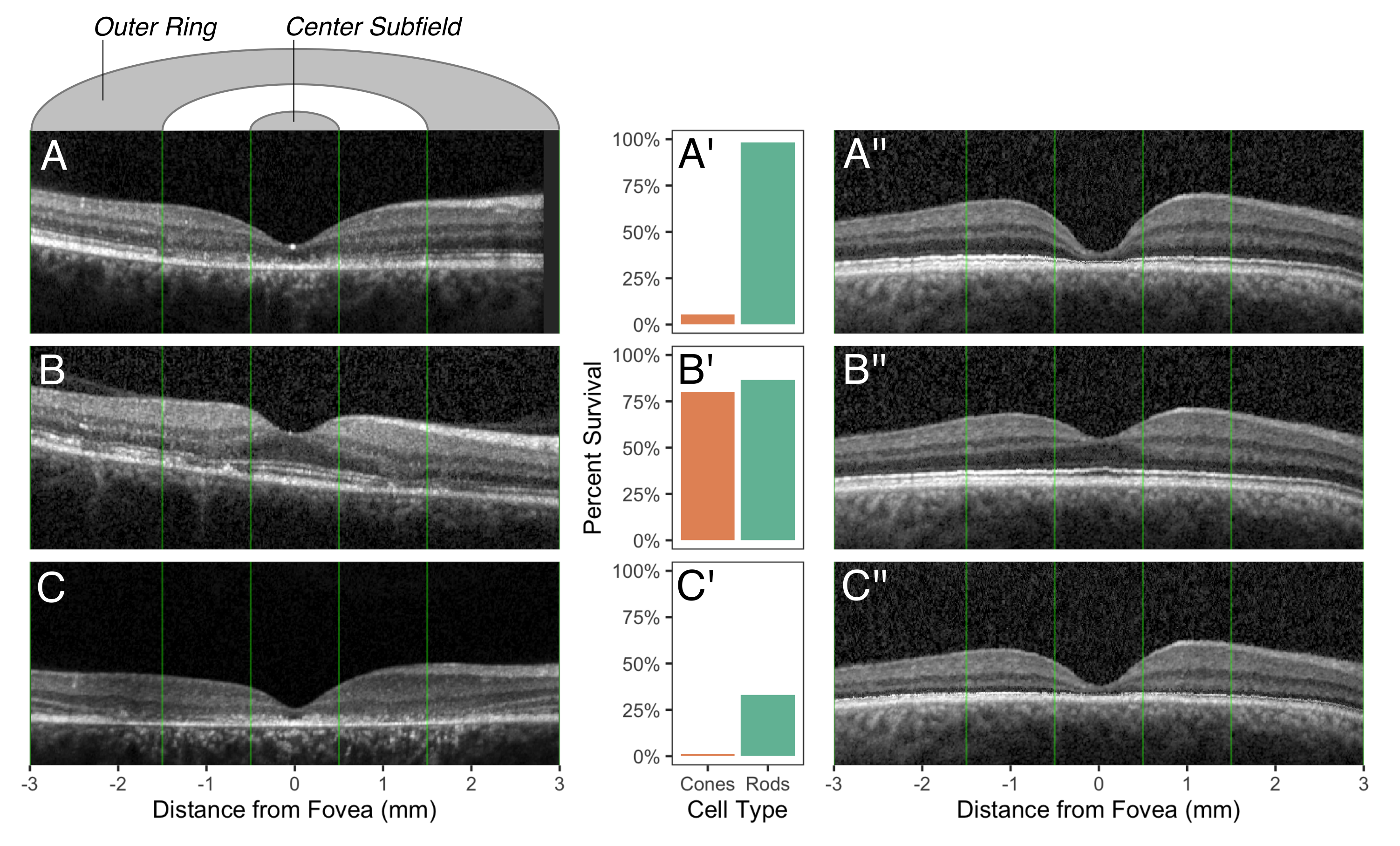
Figure 3 | OCT scans for three patients, estimates of photoreceptor survival, and corresponding images from the computational model.
Potential benefits for patients, physicians, and scientists
Our model can potentially help patients, their physicians, and researchers. As clinical trials are developed, assessing how far a patient's disease has progressed will help physicians determine what kind of treatment a patient might be eligible for. After further validation of the model, the estimates produced by our model could one day be directly computed on an OCT system and provided to physicians. Vision scientists working in computational and mathematical ophthalmology can use the reference model to visualize the outcomes of complex mathematical models.
Of course, the relationship between photoreceptor cell death and retinal layer thickness is more complicated than our model currently assumes. Our next challenge is to incorporate what is known about the variability of photoreceptor distributions and retinal layer thicknesses. This will allow us to show the uncertainty around the model-based estimates.
References
1 Whitmore, S. S. et al. Modeling rod and cone photoreceptor cell survival in vivo using optical coherence tomography. Sci Rep 13, 6896 (2023). https://doi.org:10.1038/s41598-023-33694-y
2 Curcio, C. A., Sloan, K. R., Kalina, R. E. & Hendrickson, A. E. Human photoreceptor topography. J Comp Neurol 292, 497-523 (1990). https://doi.org:10.1002/cne.902920402
3 Whitmore, S. S. et al. Analysis of retinal sublayer thicknesses and rates of change in ABCA4-associated Stargardt disease. Sci Rep 10, 16576 (2020). https://doi.org:10.1038/s41598-020-73645-5
Follow the Topic
-
Scientific Reports

An open access journal publishing original research from across all areas of the natural sciences, psychology, medicine and engineering.
Related Collections
With Collections, you can get published faster and increase your visibility.
Dementia
Publishing Model: Hybrid
Deadline: Apr 30, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in