Genetic and clinical landscape of breast cancers with germline BRCA1/2 variants
Published in Cancer
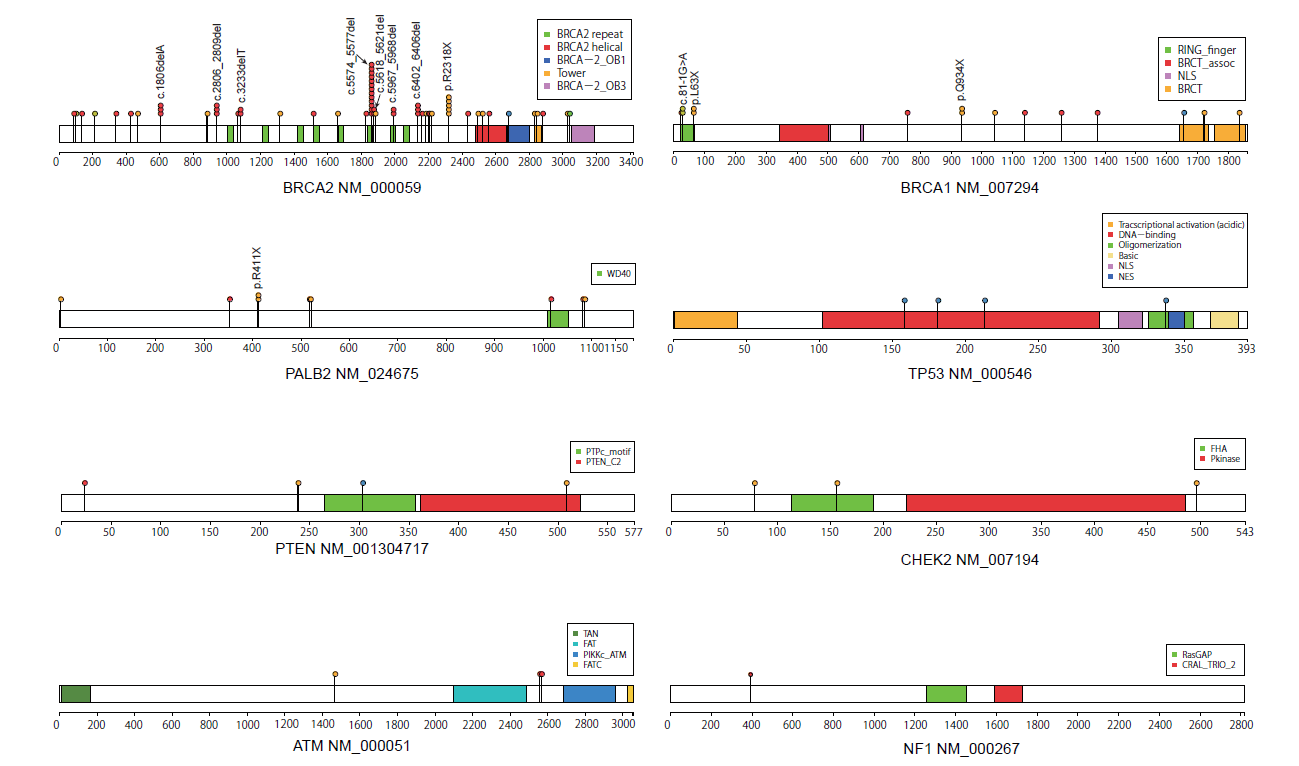
This study began with the question about the frequency of pathogenic germline variants in Japanese breast cancer patients. Previous studies reported the frequency of BRCA1/2 germline variants in high risk cohort of familial breast cancer patients, but the frequency in unselected Japanese breast cancer cohort was not reported when this study was initiated. In addition, characteristics of breast cancers that developed in patients with germline variants has not been fully elucidated. We established the biobank to collect tissue and blood samples from breast cancer patients in 2011, which enabled us to investigate these questions.
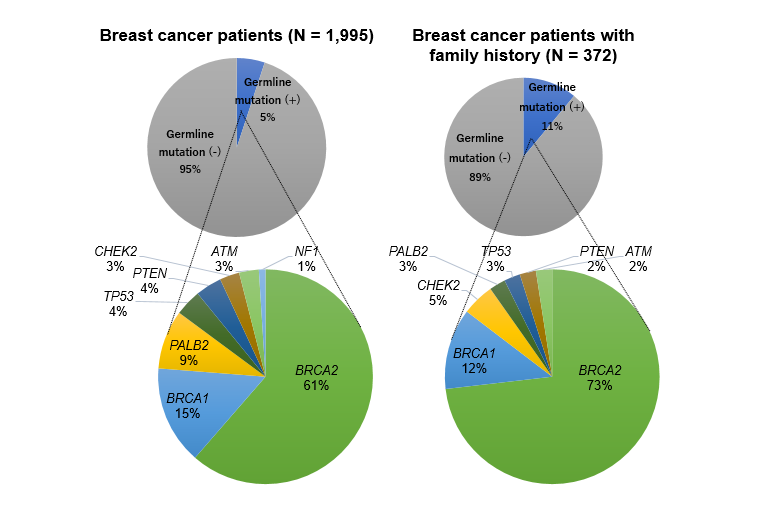
We used DNA extracted from blood cells to identify germline variants, and 11 genes implicated in hereditary breast cancer were studied by targeted-capture sequencing for 1,995 unselected Japanese women with breast cancer. Pathogenic variants were identified in 101 patients (5.1%), which included 62 BRCA2 and 15 BRCA1 mutations, which was equivalent to the previously reported frequency in the Japanese1 and Chinese cohorts2. Pathogenic variants were more frequently identified in patients with a family history of breast cancer (n = 41, 11.0%), compared with those without (n = 50, 3.4%) (P < 0.00001). Of the analyzed genes, BRCA2 was the most frequently mutated in both patients with and without a family history (see accompanying figure).
Given the higher proportion of BRCA1/2 genes, then we focused on the patients with BRCA1/2 mutations. Somatic mutations and copy number changes in the accompanying tumors were also interrogated for those patients, for whom tumor samples were available, in order to link the germline risk alleles to the associated genetic alterations. BRCA1/2 are tumor suppressor genes, and it is expected that majority of tumors that develop in cases with germline pathogenic variants lose normal alleles through LOH (loss of heterozygosity) or somatic mutation, resulting in biallelic inactivation of these genes. However, in reality, genomic sequencing of tumors with BRCA1/2 germline pathogenic variants revealed that both alleles are not always inactivated. It has been reported that about half of BRCA2 variant breast cancers and 10% of BRCA1 variant breast cancers have an intact allele3. In our study, biallelic inactivation was observed in 86% and 72% of tumors with germline BRCA1 and BRCA2 mutations, respectively. We revealed that biallelic inactivation was associated with more extensive deletions, copy neutral LOH and gain with LOH (see accompanying figure). Strikingly, TP53 and RB1 mutations were frequently observed in BRCA1- (94%) and BRCA2- (9.7%) mutated tumors with biallelic inactivation. Inactivation of TP53 and RB1 together with BRCA1 and BRCA2, respectively, involved LOH of chromosomes 17 and 13. The previous study3 showed no significant clinical differences between tumors with and without biallelic inactivation of BRCA1/2. Surprisingly, however, the patients with biallelic inactivation in our cohort showed a significantly younger onset than those without (see accompanying figure).
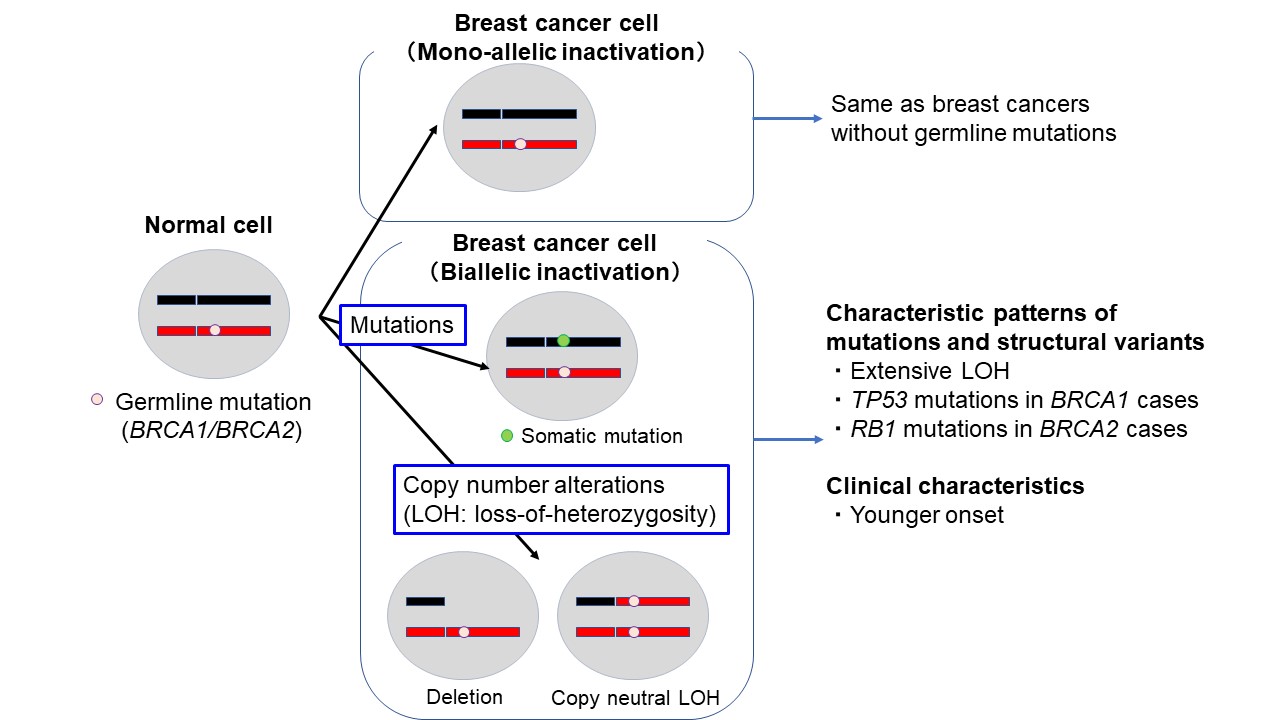
To summarize, we revealed that breast tumors with pathogenic germline BRCA1/2 variants show different genetic and clinical characteristics depending on the presence or absence of biallelic inactivation of these genes. Our data also highlights the importance of allelic status of cancer driver genes and the heterogeneity of breast cancers with germline variants. Further studies will be required to reveal the genomic landscape of breast tumors with germline variants in other breast cancer susceptibility genes.
- Momozawa, Y. et al. Germline pathogenic variants of 11 breast cancer genes in 7,051 Japanese patients and 11,241 controls. Nat Commun 9, 4083 (2018).
- Sun, J. et al. Germline Mutations in Cancer Susceptibility Genes in a Large Series of Unselected Breast Cancer Patients. Clin Cancer Res 23, 6113-6119 (2017).
- Maxwell, K.N. et al. BRCA locus-specific loss of heterozygosity in germline BRCA1 and BRCA2 carriers. Nat Commun 8, 319 (2017).
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Dec 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in