Highly conducting single-molecule topological insulators based on mono- and di-radical cations
Published in Chemistry
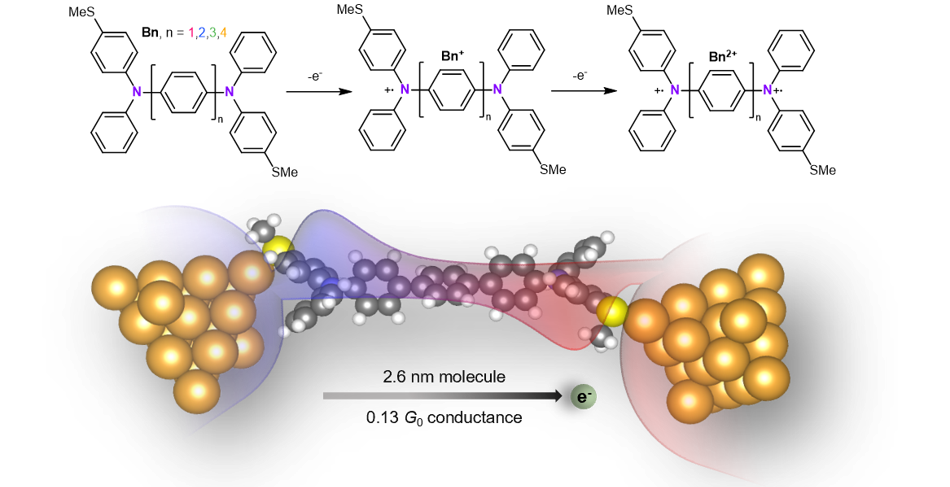
An abstract figure of this work. Top: the two-step oxidation of the bis(triarylamines) molecular series. Bottom: the geometry of the highest conducting trimer (n=3) molecule in the molecular junction. Red and blue regions are artistic depictions on the coupling between the two edge states.
Designing long and highly conducting molecular wires that function as 1D topological insulators has been a challenge from a theoretical and experimental perspective [Nature 396, 60-63 (1998); Science 300, 1384-1389 (2003); Science 320, 1482-1486 (2008); Science 323, 1193-1197 (2009)]. Although molecular wires with weak length-dependent conductance decay [Nature Nanotechnol. 6, 517-523 (2011); Nature Commun. 6, 1-8 (2015)] have been proposed, a reversed conductance decay [Phys. Rev. B 75, 245407 (2007)] is a key signature of a 1D topological insulator. A common proposal to achieve such wires is to design molecular wires with diradical resonance structures, following the Su-Schrieffer-Heeger (SSH) model [Phys. Rev. Lett., 42, 1698 (1979); Rev. Mod. Phys., 60, 781 (1988); Nature Commun., 7, 1-6 (2016); Nano Lett., 18, 7298-7304 (2018); Nature Nanotechnol., 15, 437-443 (2020);]. While this has been well established theoretically, experimental realizations are lacking.
Here, we design, synthesize and characterize a reliable redox active molecular wire series using bis(triarylamines), and show that they exhibit a robust reversed conductance decay with length upon successive chemical oxidation, realizing the SSH model. We carry out single-molecule junction conductance measurements, DFT+NEGF calculations, and structural analysis on the neutral, monocation, and dication states of these wires to provide a comprehensive picture of this nonclassical behavior. Importantly, we show that the negative conductance decay for the monocation series is much higher than that of dication series when measured at a low bias as would not be expected for a classical SSH wire. Furthermore, our results demonstrate a new strategy to design high-conducting molecular wires.
Another critical finding of this work is the ultrahigh conductance (greater than 0.1 G0) that we measure for the 2.6 nm long dication trimer in this series, 5400-fold greater than the neutral form. Such an exceptional enhancement of conductance upon oxidation has not, to the best of our knowledge, been observed before. It suggests a potential application for the wire as a building block for highly conducting molecular-based materials. Interestingly, for oxidized tetramer, the conductance gets lower than that of the oxidized trimer, showing a transition from negative conductance decay to positive conductance decay. We attribute this transition to the decoupling of the topological radical states on the edges on the molecular length as its length increases, suggesting the distance limit of the edge state coupling in oligophenylene based molecular systems.
Read more about our work in Nature Chemistry:
Contributing Authors:
Liang Li, Jonathan Z. Low, Jan Wilhelm, Guanming Liao, Suman Gunasekaran, Claudia R. Prindle, Rachel L. Starr, Dorothea Golze, Colin Nuckolls, Michael L. Steigerwald, Ferdinand Evers, Luis M. Campos, Xiaodong Yin, and Latha Venkataraman
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in