Highly stable coherent nanoprecipitates via diffusion-dominated solute uptake and interstitial ordering
Published in Materials
Light-weight aluminum (Al) alloys are extensively applied in industrial fields, known as the second most widespread metal after iron (including steel). However, the Achilles' Heel of commercial Al alloys is that they are limited to low-temperature applications < ~150 °C, far from the critical requirement of 300-400 °C service by modern industrial development. It is thus urgent and critical to develop high temperature-resistant Al alloys by proposing new strategies for microstructural tuning towards enhanced thermal stability.
Basically, the Al alloys are strengthened by nanoprecipitates that strongly pin dislocations and effectively block their movement. The development of high temperature-resistant Al alloys is, to some extension, equivalent to produce heat-resistant nanoprecipitates, for which a high thermal stability and a large volume fraction are both necessarily required, but they are usually exclusive in Al alloys. High-solubility and fast-diffusing solutes (such as Cu, Zn, and Si) do constitute nanoprecipitates with a large volume fraction (> ~1.0 vol.%) but with insufficient thermal stability. On the contrary, slow-diffusing solutes (such as Sc, Ti, and Zr) can lead to nanoprecipitates with high stability but in inadequate volume fraction (< 0.3 vol.%). The solutes added within Al alloys could be simply divided into two kinds, as representatively demonstrated in Fig. 1. We then put forward the design of great-volume, highly stable nanoprecipitates by coupling the two kinds of solutes, i.e. the high-solubility solutes to produce nanoprecipitates in a high volume fraction while the slow-diffusing solutes to stabilize these nanoprecipitates.
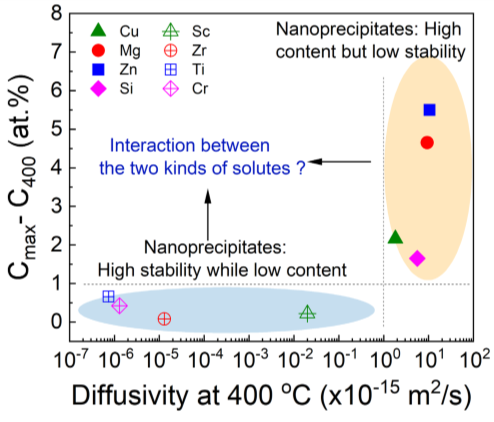
Fig. 1 Comparison of the excess solid solubility (= Cmax - C400, the difference between the maximum solid solubility and the solid solubility at 400 °C that represents the volume fraction of possibly-formed nanoprecipitates) and diffusivity at 400 °C (representing thermal stability of the formed nanoprecipitates) among some representative solutes in the Al alloys
We have achieved this coupling in Sc-microalloyed Al-Cu-based alloys through interfacial segregation. High-solubility Cu solute is to create high-volume θ′-Al2Cu nanoprecipitates in the Al-Cu-based alloys. The plate-like θ′-Al2Cu phase is coherent with the matrix at the plate interfaces. The slow-diffusing Sc solute tends to segregate at the θ′/matrix interfaces to stabilize the nanoprecipitates by (i) reducing the interfacial energy or the driving force for particle coarsening (from the thermodynamic point of view), and (ii) isolating the nanoprecipitates from Cu entrance or suffocating the nanoprecipitates (from the kinetic point of view). This interfacial coupling, although stabilizing the θ′ nanoprecipitates up to ~300 °C, becomes unstable at higher temperatures, since the segregated solutes are apt to concentrate into intermetallic particles (such as Al3Sc in the Al-Cu-Sc alloys). A new coupling strategy should be advanced to reach 350 °C- or 400 °C-level heat-resistant nanoprecipitates.
Here in this paper, we demonstrated an interstitial solute ordering strategy in Sc-microalloyed Al-Cu-Mg-Ag alloys, which is actualized through an in-situ phase transformation. The commercial Al-Cu-Mg-Ag alloys are well known for their excellent thermal stability at temperatures up to 200 °C, where Cu atoms are used to form {111}-orientated Ω nanoprecipitates rather than {100}-orientated θ′ ones, with Mg and Ag atoms segregated at the coherent plate interfaces. At the second procedure of an ad hoc two-stage aging treatment in our work, the slow-diffusing Sc atoms intruded into the Ω nanoprecipitates gradually and occupied the interstitial sites periodically. The diffusion-controlled Sc uptake and interstitial ordering built up a sublattice to strengthen the crystal structure of the Ω phase. That means the previously formed Ω nanoprecipitates were in situ transformed into more stable new ones with an intensified structure, which we define as V phase. A direct coupling between the fast-diffusing Cu and slow-diffusing Sc solutes became a reality within the nanoprecipitates. The V nanoprecipitates, inheriting a great volume fraction from the precursor Ω ones and holding a super-high stability boosted by the Sc-ordering sublattice, enable the Al alloys to reach an unprecedent creep resistance as well as exceptional tensile strength (~ 100 MPa) at 400 ºC.
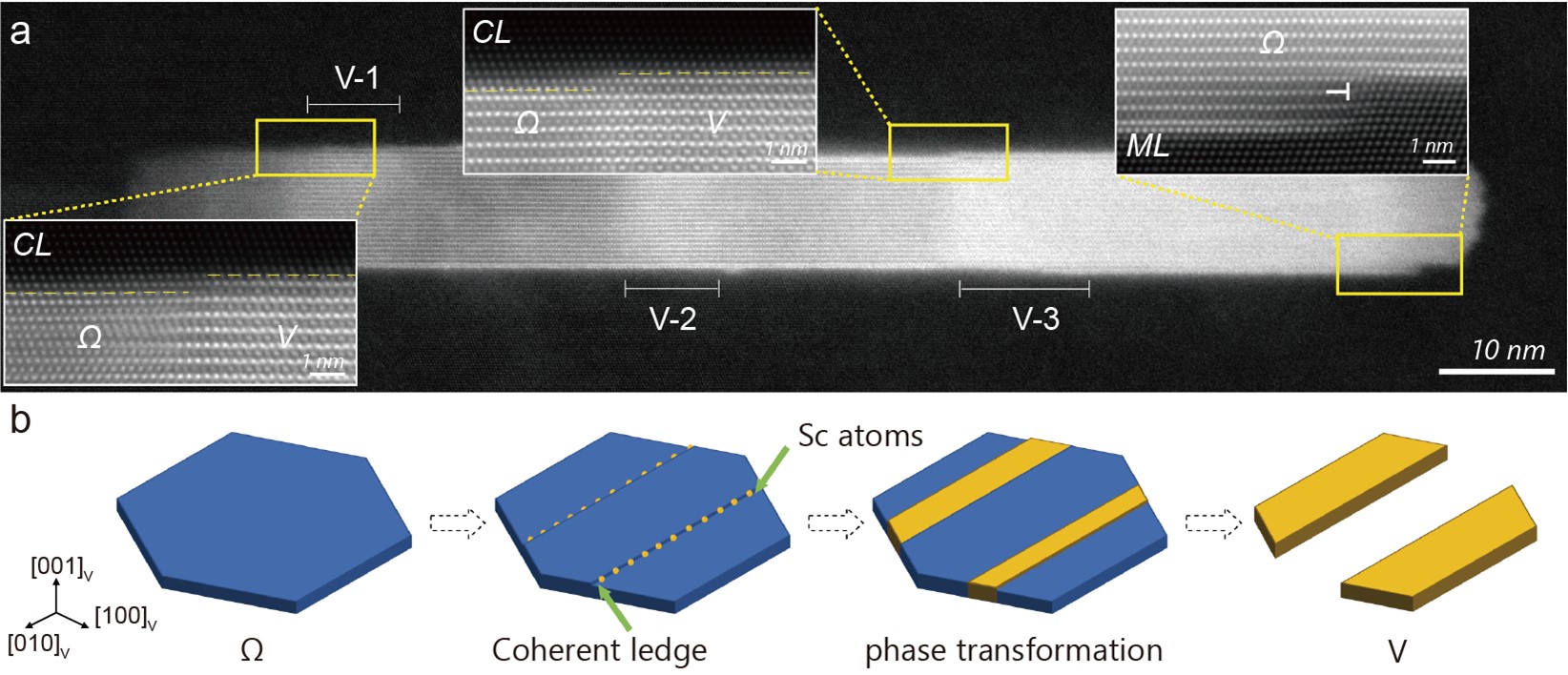
In analyzing the detailed mechanisms for Ω-to-V in situ phase transformation, we found unexpected phenomena at the atomic length scale: it is the coherent growth ledges (on the coherent plate interfaces of Ω nanoprecipitates) that provide diffusion channels for the Sc uptake. Figure 2(a) shows experimental evidences that multiple V phases were simultaneously formed within a large Ω nanoprecipitate, all triggered by coherent ledges. Since the proposal of the concept of growth ledge in the early 1960’s, justifiably credited to Aaronson, ledge movement has been exclusively recognized as a growth mechanism in solid-solid phase transformations, especially in precipitation. Here in the present Ω-to-V phase transformation, the coherent ledge opens another function that is to trigger Sc diffusion by providing a specific diffusion channel. The sketches in Fig. 2(b) illustrate the ledge-promoted in site phase transformation, where the V formation induced by Sc ordering competes with Sc-unreached Ω dissolution.
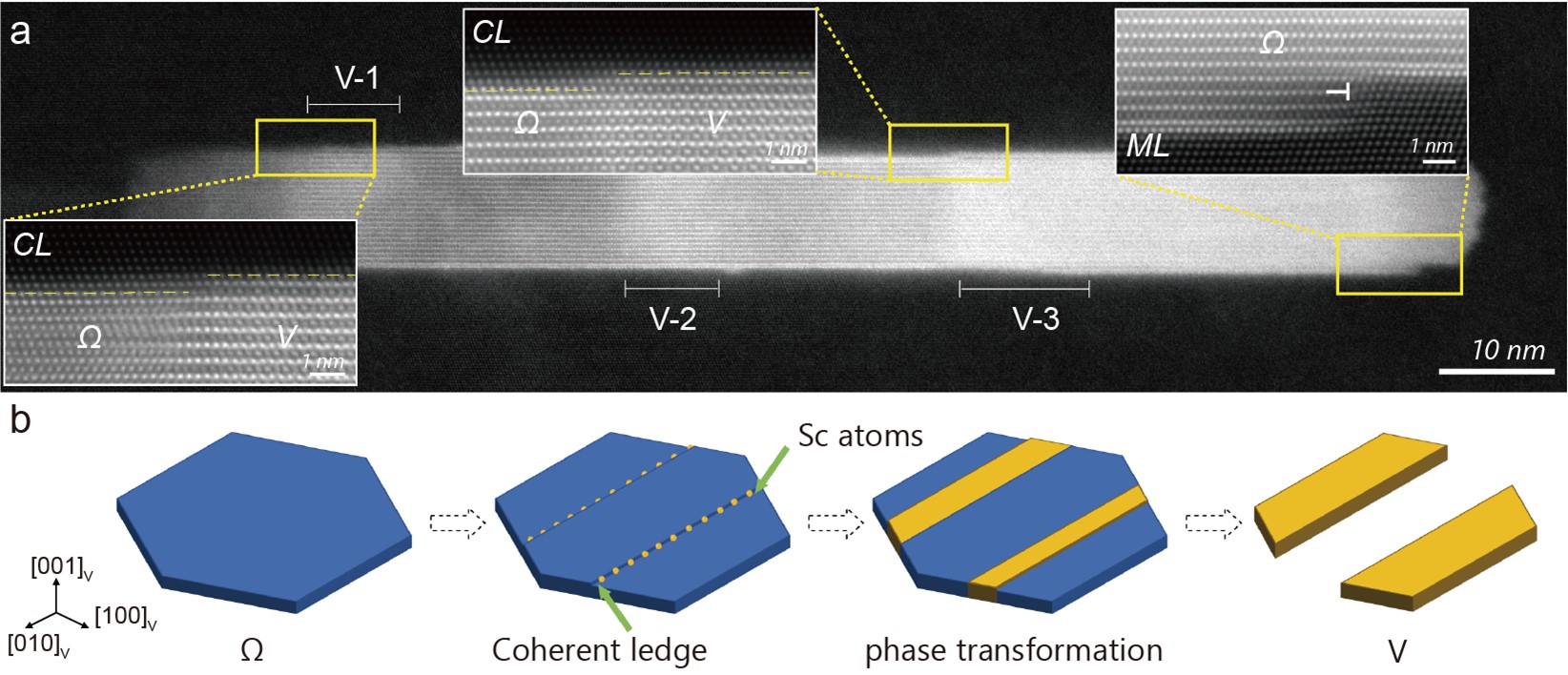
Fig. 2 (a) A representative HAADF image showing multiple V phases (named V-1, V-2, and V-3) simultaneously formed within a large Ω nanoprecipitate, all associated with coherent ledges (CLs); (b) Sketches to show the CL-aided Sc diffusion into Ω and the in-situ phase transformation from Ω to V, with a competition between V formation and Sc-unreached Ω dissolution.
In summary, the highlights in our work mainly include: (i) unusually ledge-triggered Sc uptake to assemble fast-diffusing Cu and slow-diffusing Sc solutes; (ii) interstitial Sc ordering to create highly stable, and high-volume V nanoprecipitates; and (iii) enable the Al alloys to reach an unprecedent creep resistance and exceptional tensile strength (~ 100 MPa) at 400 ºC. We also explained in this work why the template-like phase transformation happens in the Al-Cu-Mg-Ag alloys (not in Al-Cu alloys), what are the prerequisite for this unseen phase transformation, how the coherent ledges could aid the Sc uptake, etc. For more details, please check out our paper “Highly stable coherent nanoprecipitates via diffusion-dominated solute uptake and interstitial ordering” in Nature Materials.
Follow the Topic
-
Nature Materials
A monthly multi-disciplinary journal that brings together cutting-edge research across the entire spectrum of materials science and engineering, including applied and fundamental aspects of the synthesis/processing, structure/composition, properties and performance of materials.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in