β-Hydroxylation of α-amino-β-hydroxylbutanoyl-glycyluridine catalyzed by a nonheme hydroxylase ensures the maturation of caprazamycin
Published in Chemistry
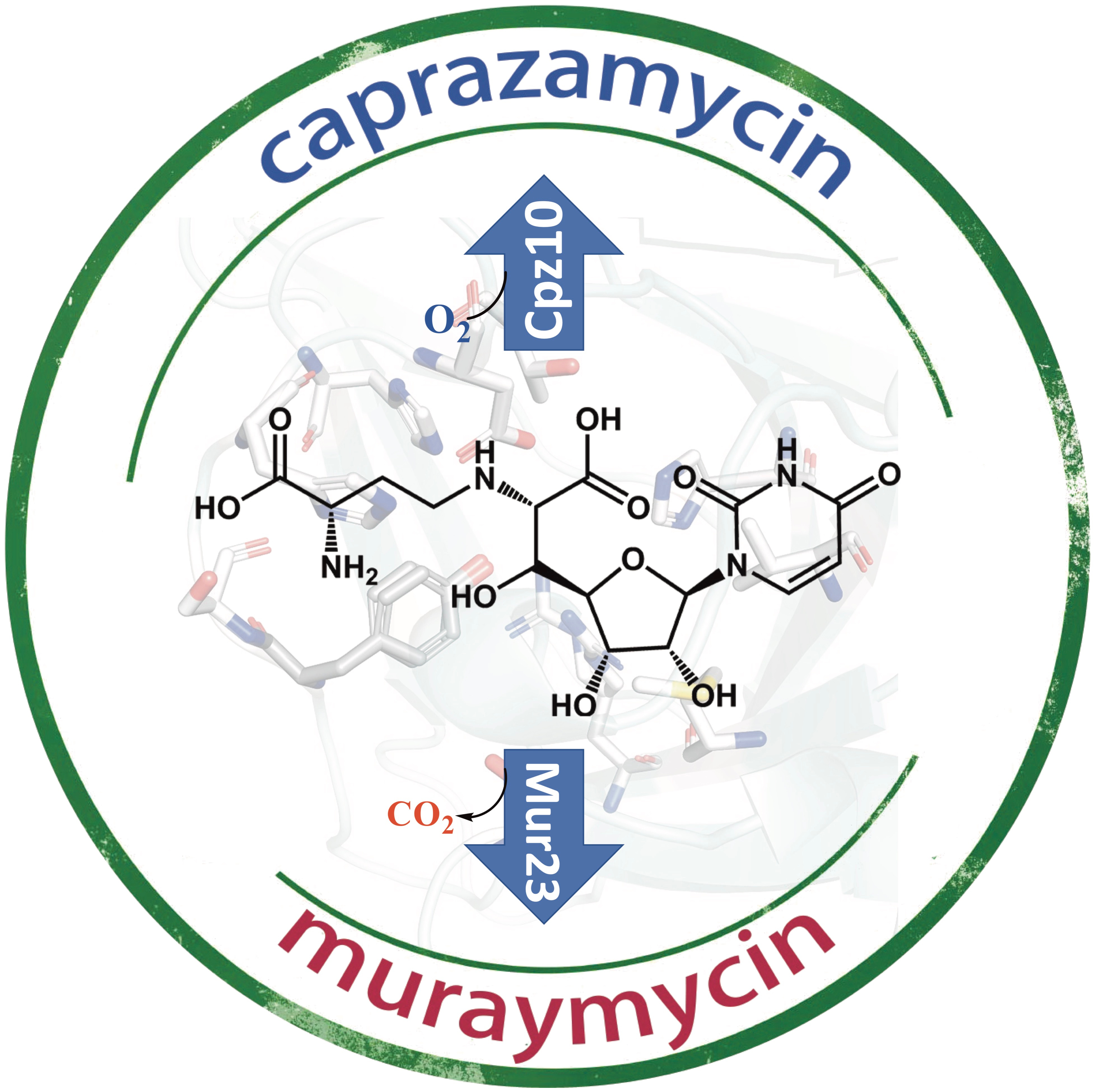
Caprazamycin is a nucleoside antibiotic that inhibits phospho-N-acetylmuramyl-pentapeptide translocase (MraY). The biosynthesis of nucleoside antibiotics has been studied but is still far from completion. The present study characterized enzymes Cpz10, Cpz15, Cpz27, Mur17, Mur23 out of caprazamycin/muraymycin biosynthetic gene cluster, particularly the nonheme αKG-dependent enzyme Cpz10. Cpz15 is a β-hydroxylase converting uridine mono-phosphate to uridine 5′ aldehyde, then incorporating with threonine by Mur17 (Cpz14) to form 5′-C-glycyluridine. Cpz10 hydroxylates synthetic 11 to 12 in vitro. Major product 13 derived from mutant Δcpz10 is phosphorylated by Cpz27. β-Hydroxylation of 11 by Cpz10 permits the maturation of caprazamycin, but decarboxylation of 11 by Mur23 oriented to muraymycin formation. Cpz10 recruits two iron atoms to activate dioxygen with regio-/stereo-specificity and commit electron/charge transfer, respectively. The chemo-physical interrogations should greatly advance our understanding of caprazamycin biosynthesis, which is conducive to pathway/protein engineering for developing more effective nucleoside antibiotics.
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Experimental and computational methodology in structural biology
Publishing Model: Open Access
Deadline: Jul 31, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in