Is diet-induced hyperinsulinemia and inflammation linked to kidney cancer risk and mortality?
Published in Cancer
Sushma Nepal, Fred K. Tabung
Background on dietary patterns, hyperinsulinemia, inflammation, and cancer
A dietary pattern is defined as the quantities, varieties, combinations, and frequency of foods 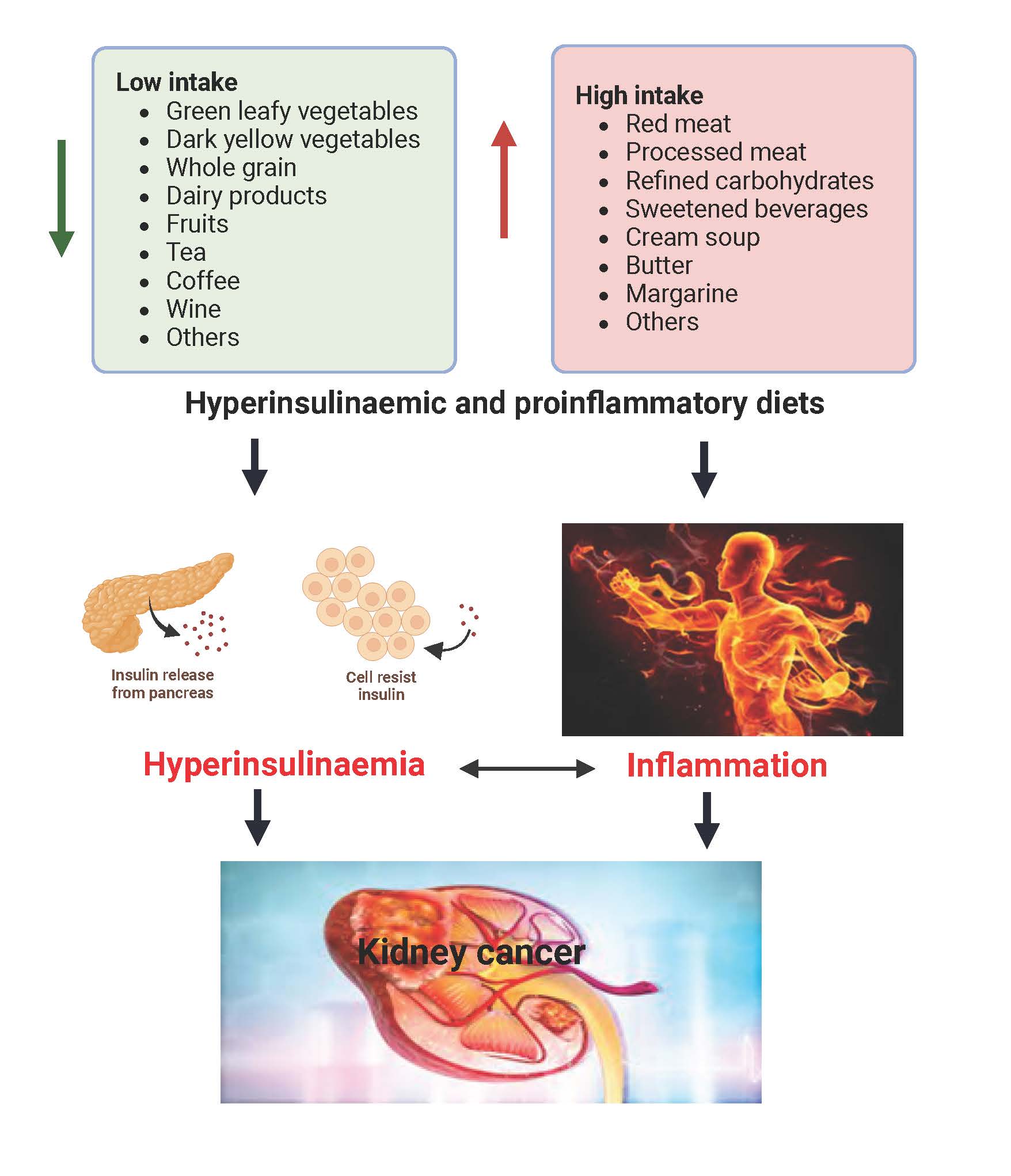 and drinks, as well as eating behaviors, rather than on single foods or nutrients. The dietary pattern approach to nutrition research has advantages over the study of single nutrients or single foods because it accounts for the complex interactions among nutrients in foods and among foods in meals as well as eating behaviors. Diet may impact cancer and other chronic diseases via its regulation of chronic insulin hypersecretion and chronic systemic inflammation. The Empirical Dietary Pattern for Hyperinsulinemia (EDIH) and the Empirical Dietary Inflammatory Pattern (EDIP) are two dietary indices that assess the ability of the diet to contribute to insulin hypersecretion and inflammation, respectively. A higher EDIH score represents a more hyperinsulinemic diet, and a higher EDIP score indicates a more pro-inflammatory diet, both of which are indicative of lower dietary quality. Hyperinsulinemia leads to an excess of insulin in blood and insulin is a growth factor that can promote cell proliferation and inhibit cell death, conditions favorable for cancer to develop and thrive. Similarly, chronic inflammation can contribute to the initiation, growth, and spread of cancer cells. Diet plays a significant role in regulating hyperinsulinemia and inflammation within the body. Previous studies have demonstrated the role of dietary patterns in cancer risk and mortality, as well as in various other chronic diseases, including diabetes and cardiovascular diseases. However, there is a lack of evidence specifically on the association between EDIH and EDIP with kidney cancer risk and mortality. We also calculated the scores for Healthy Eating Index (HEI-2015), a measure of adherence to the Dietary Guidelines for Americans (DGA), which we used as a standard of care dietary pattern to compare our results. In this study, we investigated the association of EDIH and EDIP, and HEI-2015, with the risk of kidney cancer development, kidney cancer mortality, and all-cause mortality.
and drinks, as well as eating behaviors, rather than on single foods or nutrients. The dietary pattern approach to nutrition research has advantages over the study of single nutrients or single foods because it accounts for the complex interactions among nutrients in foods and among foods in meals as well as eating behaviors. Diet may impact cancer and other chronic diseases via its regulation of chronic insulin hypersecretion and chronic systemic inflammation. The Empirical Dietary Pattern for Hyperinsulinemia (EDIH) and the Empirical Dietary Inflammatory Pattern (EDIP) are two dietary indices that assess the ability of the diet to contribute to insulin hypersecretion and inflammation, respectively. A higher EDIH score represents a more hyperinsulinemic diet, and a higher EDIP score indicates a more pro-inflammatory diet, both of which are indicative of lower dietary quality. Hyperinsulinemia leads to an excess of insulin in blood and insulin is a growth factor that can promote cell proliferation and inhibit cell death, conditions favorable for cancer to develop and thrive. Similarly, chronic inflammation can contribute to the initiation, growth, and spread of cancer cells. Diet plays a significant role in regulating hyperinsulinemia and inflammation within the body. Previous studies have demonstrated the role of dietary patterns in cancer risk and mortality, as well as in various other chronic diseases, including diabetes and cardiovascular diseases. However, there is a lack of evidence specifically on the association between EDIH and EDIP with kidney cancer risk and mortality. We also calculated the scores for Healthy Eating Index (HEI-2015), a measure of adherence to the Dietary Guidelines for Americans (DGA), which we used as a standard of care dietary pattern to compare our results. In this study, we investigated the association of EDIH and EDIP, and HEI-2015, with the risk of kidney cancer development, kidney cancer mortality, and all-cause mortality.
How did we answer our study question?
We utilized the baseline food frequency questionnaires (FFQ) from 115,830 women aged 50 to 79 who were participating in the Women's Health Initiative (WHI), to calculate the dietary scores. We excluded participants who reported prevalent cancer at baseline (except non-melanoma skin cancer), individuals with extreme energy intake, those with extreme BMI scores, and anyone missing dietary or outcome data. We also removed kidney cancer cases diagnosed within the first four years, resulting in 115,830 women for the kidney cancer risk analysis, 117,870 for mortality due to kidney cancer, and 115,918 for all-cause mortality analysis.
The timeframe for the occurrence of cancer or death was calculated from the study entry date to the date when an event was reported on a follow-up questionnaire. The study followed participants for new cases of kidney cancer, deaths caused by kidney cancer, and deaths from all causes from baseline 1993-98 until the participant was diagnosed, died, lost to follow-up, or remained alive at the end of the study in March 2019. Reports of kidney cancer diagnoses or deaths were verified centrally by a team of trained physicians at the Fred Hutchinson Cancer Center in Seattle, WA, USA. Additional verification of deaths was obtained via the National Death Index. We classified participants into fifths based on each dietary index score and employed Cox regression analyses adjusted for multiple potential confounding variables to determine risk of kidney cancer, mortality specific to kidney cancer, and overall mortality, using hazard ratios. For example, those in the highest fifth were expected to be consuming the most hyperinsulinemic or most pro-inflammatory diet and therefore at highest risk of the study outcomes, compared with those classified in the lowest fifth.
What were our findings?
Over a median follow up time of 19.9 years, there were 429 participants diagnosed with kidney cancer, and the relative risk of kidney cancer development was increased by 77% for women classified in the highest EDIH quintile (most hyperinsulinemic diet) compared to the lowest quintile. Over the same time period, 41,333 participants died of all causes including 113 from kidney cancer, and for kidney cancer-specific mortality, there was an 85% higher risk of dying from kidney cancer among those consuming the most hyperinsulinemic diets (EDIH quintile 5) compared to those with low EDIH diets. In contrast, the EDIP score did not show a significant association with these outcomes except total mortality.
In summary, higher EDIH scores, indicating greater insulinemic potential of the diet or a low-quality diet, was linked to an increased risk of developing kidney cancer, and of dying from kidney cancer as well as other causes of death. Also, a high-quality diet as measured by the HEI-2015 score was associated with a lower risk of kidney cancer development and all-cause mortality.
Take-home message
The foods we consume on a habitual basis (i.e., the foods that the body “sees” on a regular basis) play a significant role in our susceptibility to chronic diseases and overall health. Highly processed foods with a lot of added sugar are frequently consumed by all demographic subgroups from children to adults. This pattern contributes to a higher EDIH score which is associated with an increased risk of developing chronic conditions like kidney cancer, as well as higher mortality rates and reduced survival. Consequently, embracing a dietary pattern with a high ability to lower chronic insulin hypersecretion (low EDIH diet) will be crucial for promoting healthier and longer lives. A low-EDIH dietary pattern has high intakes of in green-leafy vegetables, dark-yellow vegetables, whole-fat dairy, fatty fish, whole grains, coffee and moderate wine; and low intakes of red meat, processed meat, sugar-sweetened beverages, artificially sweetened beverages, butter, margarine, and refined grains.
Follow the Topic
-
British Journal of Cancer
This journal is devoted to publishing cutting edge discovery, translational and clinical cancer research across the broad spectrum of oncology.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in