Journal Club | Printing Porous Structures
Published in Chemistry
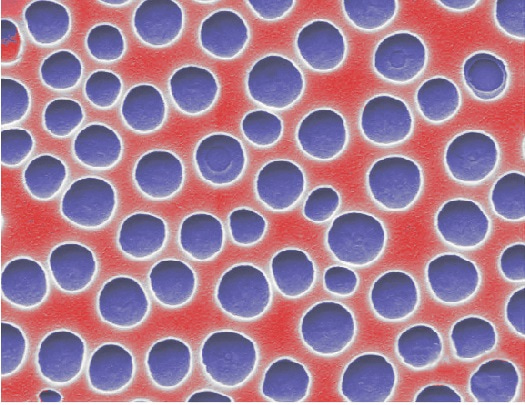
Porous materials are widely used in technologies where large surface area at small material volume is beneficial. Given the importance of these materials in many fields, research on fabrication and processing of porous materials created a great selection of different methods which enable the control of morphology of macroscopic objects on different length scales. Simultaneously, additive manufacturing and 3D printing revolutionized our potential to produce custom made and arbitrarily complex 3D objects, fast and on demand.
Implementing porosity into 3D printed objects is interesting, not least because it allows a transfer of the advantages of additive manufacturing to the fabrication of porous materials. Previously, printing of inks containing an inert template phase has been realized. Removal of the template after polymerization leaves behind a porous structure with little in-situ control on pore distribution and pore size.[1,2] Recently, polymerization induced phase separation (PIPS) – a phenomenon where an initially miscible resin of monomer and porogen phase separates during polymerization – has been successfully applied in printing porous glasses using stereolithography.[3]
In their recent communication published in Advanced Materials, Wegener and co-workers use an organic photoresist which phase separates during photopolymerization and demonstrate printing of porous polymer monoliths using 3D laser microprinting. Importantly, their photoresist which is composed of a crosslinking triacrylate, a commercial photoinitiator and a porogen can be applied in two-photon absorption based microprinting, a technique which provides better spatial resolution than previously applied stereolithography.
Since the monomer and the porogen – typically organic solvents such as Dodecyl acetate or Octadecyl acetate – are miscible in the initial state and have a homogeneous refractive index distribution, the resin does not suffer from light scattering at the excitation wavelength, a problem frequently encountered when using sacrificial templates. Washing away the unpolymerized monomer and the porogen after the printing step leaves behind air filled pores with a pore size of 50 nm, well below the size of a voxel. A significant refractive index contrast between air and the polymer and light scattering on the air-polymer interface gives the printed parts a white appearance.
Clearly, the nature of the porogen as well as the amount of porogen and monomer in the resin will affect the pore size and porosity of the printed object but the authors found an additional handle to tune the porous morphology: An increase in laser power results in a decreased porosity. This is counter intuitive at first but on a closer look, the polymerization volume in 3D microprinting is on a length scale where diffusion effects become important. Similarly, multiple exposures, which give the monomer enough time to diffuse in the pores between the exposure events, reduce the porosity. Both, increasing the laser power as well as the double exposure strategy allow the authors to write gradient structures with porosities ranging from 27% to 3%. Concomitantly with tuning the porosity, the scattering coefficient can be tuned and light scattering completely vanishes for high exposure doses or low porosity, rendering the material transparent.
The possibility to control the porosity of polymer materials in combination with microprinting greatly expands our scope of fabrication techniques for porous materials. Using the exposure time as an additional handle to tune the porosity in photopolymerization based 3D printing as well as controlling porosity and light scattering gradients in polymer materials opens the way to interesting applications.
The original article can be found here:
Mayer, F. Ryklin, D. Wacker, I. Curticean, R. Calkovsky, M. Niemeyer, A. Dong, A. Levkin, P. A. Gerthsen, D. Schroder, R. R. Wegener, M. 3D Two-Photon Microprinting of Nanoporous Architectures. Adv. Mater. (2020), 2002044 DOI: 10.1002/adma.202002044
References:
[1] Johnson, D. W. Sherborne, C. Didsbury, M. P. Pateman, C. Cameron, N. R. Claeyssens, F. Macrostructuring of Emulsion‐templated Porous Polymers by 3D Laser Patterning. Adv. Mater. 25 (2013) 3178 DOI: 10.1002/adma.201300552
[2] Mu, X. Bertron, T. Dunn, C. Qiao, H. Wu, J. Zhao, Z. Sladana, C. Qi, H. J. Porous polymeric materials by 3D printing of photocurable resin. Mater. Horiz. 4 (2017) 442 DOI: 10.1039/C7MH00084G
[3] Moore, D. G. Barbera, L. Masania, K. Studart, A. R. Three-dimensional printing of multicomponent glasses using phase-separating resins. Nat. Mater. 19 (2020) 212 DOI: 10.1038/s41563-019-0525-y
Image credit: Johannes Kreutzer / DOI: 10.1038/srep15947
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
Excellent study!!