Lipopolysaccharide biosynthesis is spatially organized in the pathogen Brucella abortus
Published in Microbiology
Imagine that you are a bacterial pathogen, living most of the time inside host cells. Your interaction surface with the host cell will be the envelope. If you are a diderm bacterium, you have two membranes, the inner membrane, and the outer membrane. Between these two membranes, a periplasm in which an exoskeleton called peptidoglycan (PG for the intimate) is attached to the outer membrane and gives the shape of the bacterium. The outer membrane is thus a very important organelle because it not only makes the interaction with the "outside world”, but it also contributes to the cell shape by its linkage to the PG1. The outer membrane is thus an interesting target for future antimicrobial therapy since it is essential and accessible. Thus, there is a strong interest in studying the outer membrane of pathogens, which benefits from the wonderful fundamental research made with bacterial models like Escherichia coli.
Our favorite pathogen is Brucella abortus. It is a nasty intracellular pathogen mainly responsible for bovine brucellosis, a worldwide zoonosis. It can be grown on plates and it is genetically tractable, but in the wild, it is mainly found inside host cells. This pathogen is proposed to encounter several environments that aggress the envelope, such as an acidic pH or cationic peptides. At the same time, to benefit from its infectious cycle, B. abortus needs to grow. Unlike E. coli, B. abortus grows unipolarly, like many Rhizobiales2, which form a diversified group of alpha-proteobacteria. “Unipolarly” means that new envelope components are specifically incorporated at one pole of the small rod that B. abortus forms. This pole is called the “new pole” or “growing pole”. After a period of growth, the envelope synthesis is arrested at the new pole and new envelope material is incorporated at the midcell, prior to division. Concomitantly with division, two new poles are generated at the previous division site in each sibling cell.
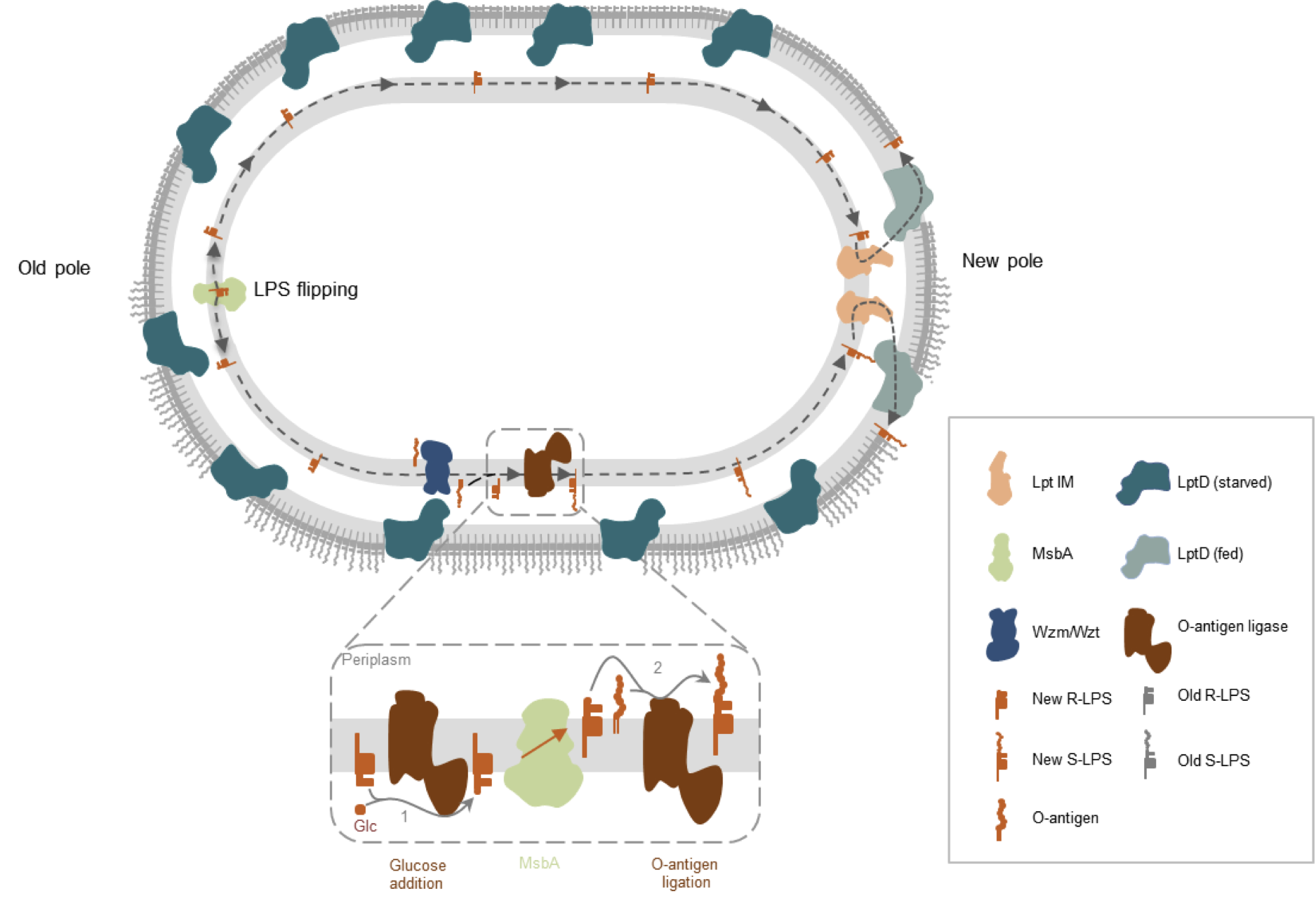
Insertion of new envelope components includes the synthesis of a complex molecule called lipopolysaccharide (LPS). As suggested by its name, it is composed of a lipidic part (the so-called “lipid A”), on which a complex polysaccharide structure, often branched and called “core”, is attached forming the rough LPS (R-LPS). On the core, a long polysaccharide called “O-chain” or “O-antigen” can be attached and is exposed to the exterior of the cell, thereby forming the smooth LPS (S-LPS). In B. abortus, the presence of the O-antigen is necessary to avoid brutal phagocytosis by macrophages, for example. Pioneering work in E. coli showed that the lipid A and the core are synthesized in the cytoplasm, and then flipped across the inner membrane thanks to MsbA. In several bacteria, the O-antigen is individually synthesized and flipped to the periplasmic leaflet of the inner membrane by another transporter, Wzm/Wzt. In the periplasm, on the outer leaflet of the inner membrane, the O-antigen can be attached to the core. The LPS molecules, with or without O-antigen, will then take a molecular lift to reach the outer leaflet of the outer membrane. This lift is composed of a complex named Lpt, for LPS transport, composed of seven proteins, four in the inner membrane, two in the outer membrane and one in the periplasm, linking the Lpt complexes in the inner and outer membrane. All these proteins are conserved and essential in B. abortus.
Two Ph.D. students of the team, Caroline Servais and Audrey Verhaeghe, collaborated to show that the inner membrane components of the LPS lift (Lpt) are associated with envelope growth sites, i.e. the new pole and the division site (Fig. 1).

Fig. 1. Localization of LptC fused to mNeonGreen (mNG-LptC, green foci). The identity of the old pole (“old”) is revealed by the presence of a red focus, corresponding to a PdhS-mCherry fusion. When bacteria start to divide, a mNG-LptC focus is detectable at the division site (“div”).
Interestingly, the MsbA flippase is mainly located at the opposite pole of the cell. We think that this arrangement would force LPS to travel at the outer leaflet of the inner membrane after its MsbA-mediated flipping before it can reach the Lpt complex. Additionally, the independently synthesized O-antigen is flipped by the ABC transporter Wzm/Wzt, which is homogeneously distributed at the IM. During this travel, R-LPS could meet the O-antigen ligase, the enzyme anchoring the O-antigen to the core of LPS, thereby becoming S-LPS. We identified this O-antigen ligase, which is a strange enzyme in Brucella spp. Indeed, it displays two active sites, one in the cytoplasm, where it is grafting glucose to the conserved structure of the core, and one in the periplasm where the O-antigen is linked to that last glucose of the core. Two activities in one enzyme, on the two sides of the inner membrane.
This work opens new interesting questions. First, as shown by a third Ph.D. student of the team, Vicky Vassen, LptD which is the major component of the Lpt system in the outer membrane, is found dispersed on the surface. This dispersed localization suggests that it could also be involved in the repair of the outer membrane if needed. Second, the particular localization of MsbA at one pole and Lpt at the opposite pole highlights an unanticipated aspect of LPS trafficking within the inner membrane that could increase the proportion of LPS with an O-antigen compared to the LPS without O-antigen exposed to the exterior. This could be a simple strategy to expose many O-antigens on the surface and efficiently avoid brutal phagocytosis by macrophages. Third, because newly synthesized PG and LPS are incorporated at the same places in the bacterial cell, it means that molecular mechanisms are efficiently coordinating the insertion of both envelope layers. Finally, at each cell cycle, new envelope incorporation occurs at the new pole, and then at the division site, unknown mechanisms, probably conserved in many Rhizobiales, are certainly involved in this spatial regulation. Interfering with these mechanisms could yield new targets to combat bacterial pathogens.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in