Explore the Research

Mdm4 supports DNA replication in a p53-independent fashion - Oncogene
Oncogene - Mdm4 supports DNA replication in a p53-independent fashion
The interaction between the tumor suppressor p53 and Mdm2 is presented in every textbook about tumor biology. However, 30 years after the discovery of Mdm2, the most potent negative regulator of p53, there is still a lack of clinically approved compounds targeting Mdm2. In particular, high-grade toxicities and limited efficacies are making it difficult to use Mdm2 inhibitors, like Nutlin-derived compounds, in the treatment of cancer patients. Part of the problem might be that we have still not obtained the full picture of biological activities displayed by the Mdm-proteins.
The discovery of Mdm4 alias MdmX added another layer of complexity to the feedback loop between p53 and Mdm2. Despite its similarity to Mdm2 and its essential role in efficient degradation of p53, this homologue lacks any detectable ubiquitin ligase activity of its own. Moreover, unlike Mdm2, its expression is not induced by p53. Its biological role is still puzzling since Mdm4 is crucial for embryonic development but gets spliced unproductively in most adult tissues (reviewed in [1]). However, several cancer entities express elevated levels of full-length Mdm4.
More and more evidence of p53-independent functions and additional binding partners of the Mdm-proteins are emerging. Our group characterized Mdm2 as a chromatin modifier [2] that prevents the accumulation of DNA/RNA-hybrids [3], thereby supporting DNA replication.
This led us to the question: What is the role that Mdm4 plays in DNA replication?
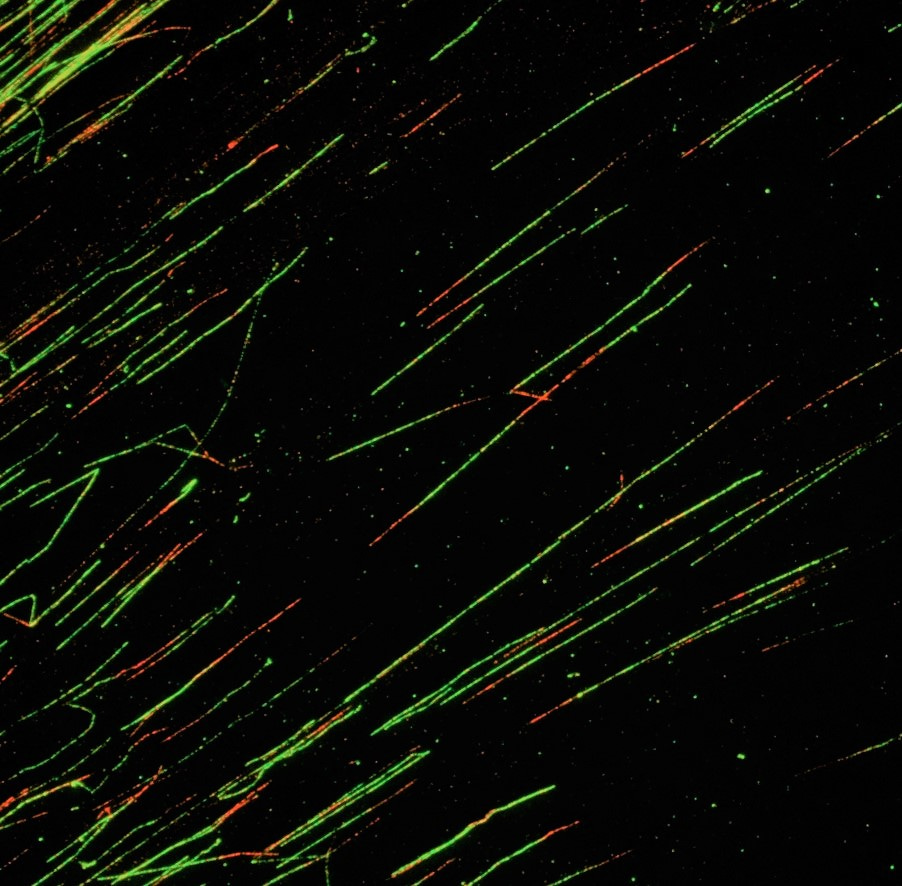
To investigate this, we studied single replication forks by conducting DNA fiber assays in cancer-derived cell lines as well as non-transformed cells lacking wildtype p53. Taking p53 out of the equation allowed us to focus on the function of Mdm4 specifically.
We found that DNA replication heavily relied on sufficient amounts of full-length Mdm4 in these cells, and we identified EZH2 and RNF2 as novel binding partners of Mdm4. How are these findings connected? In analogy to Mdm2, the absence of Mdm4 affects chromatin modifications and gives rise to the formation of DNA/RNA-hybrids – obstacles that lead to stalling replication forks. This increases the vulnerability to external replicative stressors such as gemcitabine.
Our findings suggest that it may be worthwhile to target Mdm4 not only to reactivate wildtype p53. Additionally, depleting Mdm4 leads to replicative stress and sensitizes cancer cells towards DNA damaging agents even in the absence of p53. Adult tissues, on the other hand, can be expected to tolerate the loss of Mdm4, since Mdm4 is virtually absent in these cells anyway. Thus, one would not expect much toxicity from drugs that can interfere with Mdm4 stability or activity. These considerations argue in favor of Mdm4 as a promising therapeutic target in cancer treatment, with higher specificity than its more famous brother, Mdm2.
References
1 - Marine JC, Jochemsen AG. MDMX (MDM4), a promising target for p53 reactivation therapy and beyond. Cold Spring Harb Perspect Med. 2016; 6: 1–16.
2 - Wienken M, Dickmanns A, Nemajerova A, Kramer D, Najafova Z, Weiss M et al. MDM2 Associates with Polycomb Repressor Complex 2 and Enhances Stemness-Promoting Chromatin Modifications Independent of p53. Mol Cell. 2016; 61: 68–83.
3 - Klusmann I, Wohlberedt K, Magerhans A, Teloni F, Korbel JO, Altmeyer M et al. Chromatin modifiers Mdm2 and RNF2 prevent RNA:DNA hybrids that impair DNA replication. Proc Natl Acad Sci. 2018; 115: E11311–E11320.
Follow the Topic
-
Oncogene
This journal aims to make substantial advances in our knowledge of processes that contribute to cancer by publishing outstanding research.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in