Metal oxide single-component light-powered micromotors for photocatalytic degradation of nitroaromatic pollutants
Published in Materials
Nitroaromatic compounds refer to organic compounds that consist of one or more nitro groups (-NO2) that are attached to an aromatic ring. Thanks to these groups, nitroaromatic compounds have been extensively employed to produce a wide range of industrial products, including explosives, pesticides, and dyes. However, the presence of the nitro group also impedes the biodegradation of these compounds, leading to continuous environmental accumulation and heavily contaminated groundwater posing a significant threat to human health. Conventional wastewater purification methods mainly rely on biological oxidation and physical procedures (e.g., activated carbon adsorption, and nano-filtration). Nonetheless, the biological process is inadequate due to the high stability of these compounds in water, making them challenging to degrade. Physical methods only transfer pollutants instead of removing them destructively, indicating the need for further treatment. Advanced oxidation processes (AOPs) have been extensively explored to break down organic contaminants in wastewater. AOPs typically involve highly reactive oxygen species (ROS), such as hydroxyl radical (•OH) and superoxide radical (•O2-), that can oxidize hazardous chemical species in water. Still, these approaches are limited by the passive diffusion of photocatalytic materials. Furthermore, most of them require more than one component. Therefore, it is highly desirable to investigate more simple and practical strategies to facilitate the removal of pollutants.
Light-powered micro/nanomotors, micro/nano-sized materials that harness energy from a powerful, renewable, and abundant source such as light, and convert it into motion, have garnered significant interest. Previous studies have shown that active locomotion of light-powered microrobots based on photocatalytic materials can overcome the limitation of passive diffusion of photocatalysts by enhancing interactions with targeted pollutants. Usually, a noble metal coating is required to enable self-propulsion, increasing fabrication costs and complexity. Therefore, there is a need to explore low-cost and simpler microrobots for practical applications in the future.
In this study, we demonstrate a facile strategy for degrading nitroaromatic pollutants using single-component WO3 microrobots that do not require additional noble-metal coating (Fig.1). The WO3 microrobots were prepared using a simple hydrothermal reaction followed by a calcination process, making them low-cost and easy to produce. Once exposed to asymmetric light illumination, the microrobots can self-actuate in pure water without the need for fuel. This remote control and rapid response over on/off switching of UV light allows for precise control of the microrobots' motion. In addition, the addition of H2O2 enhanced the motion and diffusion of the microrobots, making them even more effective in degrading nitroaromatic compounds.
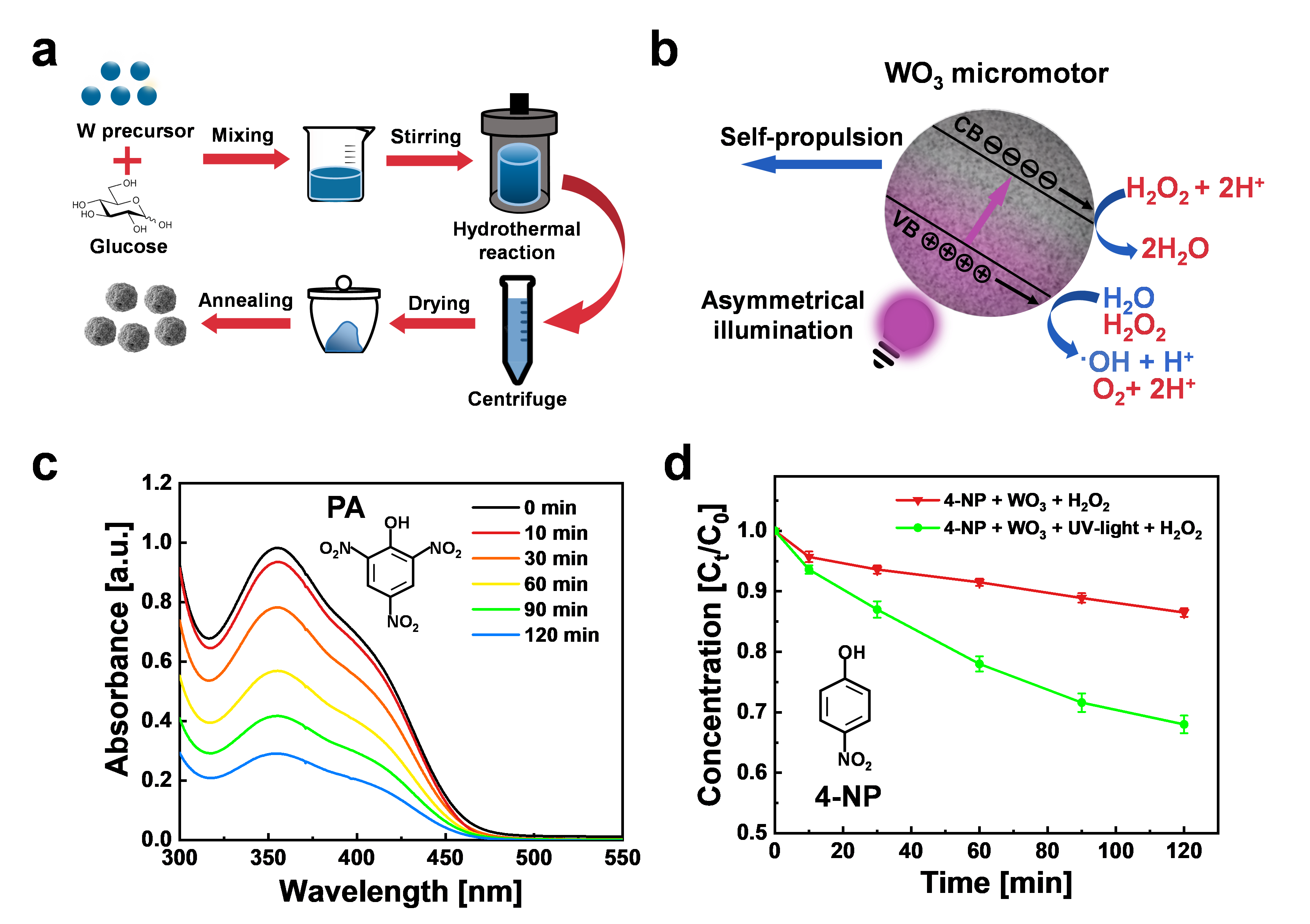 Figure 1. a Schematic illustration of the preparation of WO3 microspheres by hydrothermal and calcination processes. b Schematic illustration of the propulsion mechanism of light-powered WO3 microrobots. c UV-Vis spectra of PA after photocatalytic degradation by WO3 microrobots exposed to UV-light irradiation in 1% H2O2. d Photocatalytic degradation of 4-NP using WO3 microrobots under UV-light irradiation in 1% H2O2. Error bars represent the standard deviation, n = 3 independent replicates.
Figure 1. a Schematic illustration of the preparation of WO3 microspheres by hydrothermal and calcination processes. b Schematic illustration of the propulsion mechanism of light-powered WO3 microrobots. c UV-Vis spectra of PA after photocatalytic degradation by WO3 microrobots exposed to UV-light irradiation in 1% H2O2. d Photocatalytic degradation of 4-NP using WO3 microrobots under UV-light irradiation in 1% H2O2. Error bars represent the standard deviation, n = 3 independent replicates.
The effectiveness of these microrobots was tested on two specific pollutants, picric acid (PA) and 4-nitrophenol (4-NP), due to their hazardous nature and high stability in water. WO3 microrobots can degrade 70% of PA and 40% of 4-NP in water under light irradiation and low concentration of H2O2. In order to get further insight into the mechanism of photodegradation involving WO3 microrobots, radical experiments were also conducted to capture different ROS in the reactive medium, which demonstrated that •OH is the most effective ROS to degrade harmful organic pollutants. The reusability of WO3 microrobots was evaluated over 5 cycles of PA degradation experiments using the same microrobots. The results showed that WO3 microrobots preserved 55% degradation efficiency, indicating high photocatalytic capability.
Overall, this study provides a promising solution for environmental remediation by offering a simple and low-cost method for degrading toxic substances. The WO3 microrobots show great potential for use in micro/nanomotor systems for environmental remediation.
For further information, please read our published article in npj Clean Water, https://www.nature.com/articles/s41545-023-00235-z.
Follow the Topic
-
npj Clean Water
This journal publishes high-quality papers which report cutting-edge science, technology, application, policy and societal issues that contribute towards a more sustainable supply of clean water.
Related Collections
With Collections, you can get published faster and increase your visibility.
Resource Recovery and Carbon Mitigation in Wastewater Treatment: Advancing the Water–Energy–Carbon Nexus
Publishing Model: Open Access
Deadline: Oct 31, 2026
Advanced Sensing and Data-Driven Approaches for Water Quality Monitoring
Publishing Model: Open Access
Deadline: Oct 31, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in