Neuron type-specific proteomics reveals distinct Shank3 proteoforms in iSPNs and dSPNs lead to striatal synaptopathy in Shank3B–/– mice
Published in Neuroscience, Protocols & Methods, and General & Internal Medicine
Background
(I) The Shank3 gene encodes multiple Shank3 protein isoforms, which are important scaffold proteins in the postsynaptic density (Figure 1). They physically connect neurotransmitter receptors, ion channels, and other membrane proteins to the actin cytoskeleton and bridge pathways involving G-protein-coupled signaling. These proteins networks are essential for synaptic formation and neuronal signal transmission.
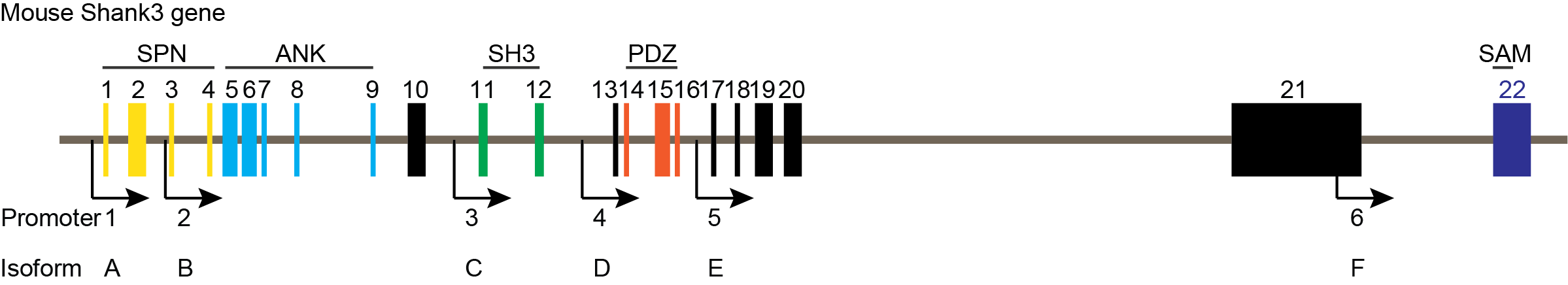
Figure 1. Mouse Shank3 gene (Gene ID: 58234) has 22 exons and six known promotors.
(II) Over half of the individuals with Phelan-McDermid syndrome (PMS) exhibit abnormalities in the SHANK3 gene. Additionally, mutations in the SHANK3 gene have been identified in approximately 1-2% of patients with autism spectrum disorder (ASD). Notably, individuals with mutations in the SHANK3 gene display a wide range of symptoms. However, the mechanism underlying this diversity is unclear.
(III) The striatum is a region of the brain that is responsible for the control of reward and movement. It contains two main types of neurons: direct spiny projection neurons (dSPNs) and indirect spiny projection neurons (iSPNs). Striatal dysfunction is commonly seen in PMS and ASD patients. However, the specific pathological contributions of dSPNs and iSPNs are still largely unknown.
Methods
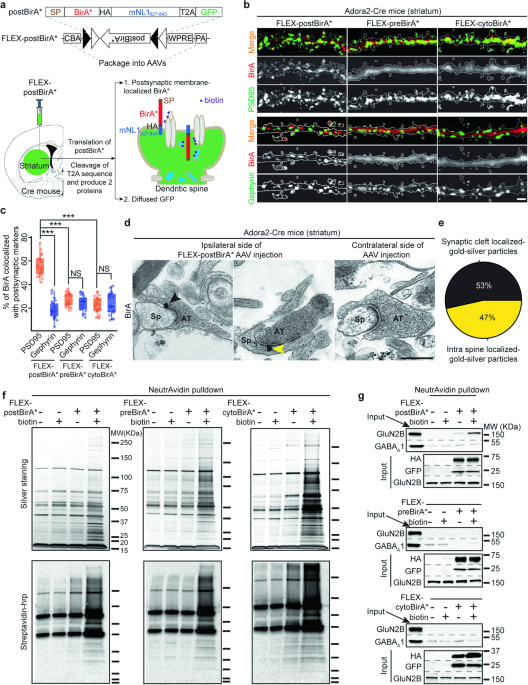
(I) Cell-type-specific proximity biotin-labeling (Figure 2): We expressed a biotin ligase, BirA*, in the iSPN (in Adora2-Cre mice) and dSPN (in Drd1-Cre mice) postsynaptic membranes. This method enables us to selectively chemically tag the postsynaptic proteome of iSPN and dSPN respectively.
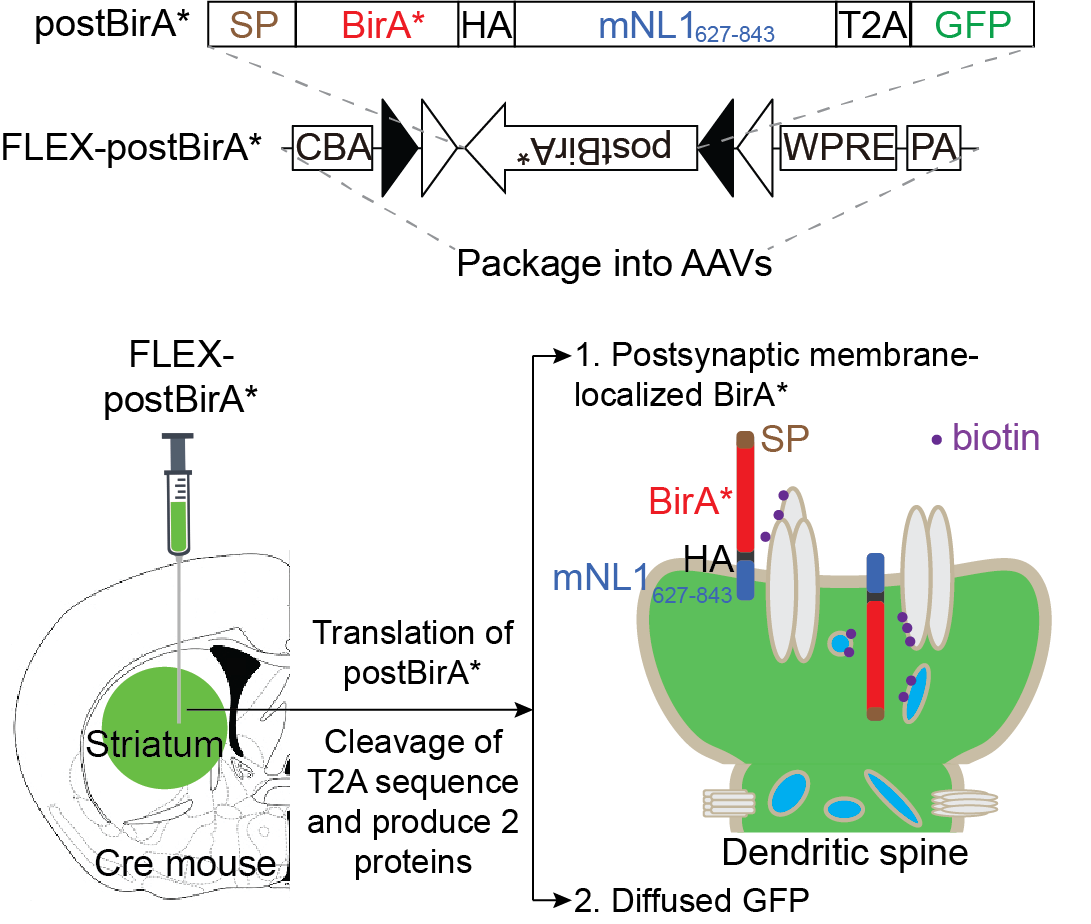
Figure 2. Cell-type-specific proximity biotin-labeling.
(II) Tandem mass tag (TMT)-based mass spectrometry (MS): We conducted a quantitative analysis of iSPNs and dSPNs postsynaptic proteomes in both wild-type (WT) and a model of ASD, Shank3B knockout (Shank3B-/-) mice.
(III) Optogenetic technique: We utilized a channelrhodopsin-based optogenetic approach to investigate corticostriatal synaptic transmission in iSPNs and dSPNs separately.
Main Results
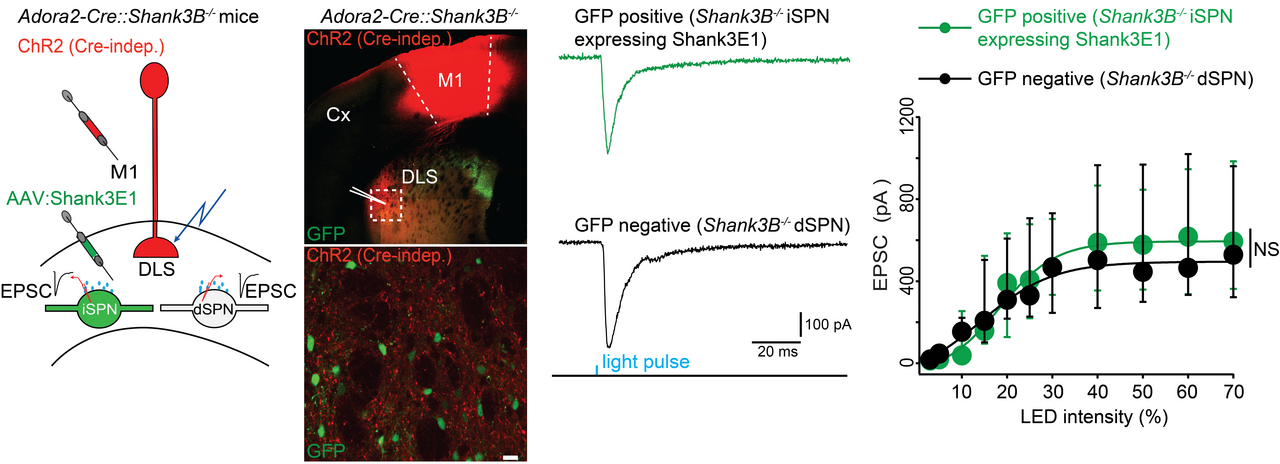
(I) The expression of Shank3 protein isoforms varies between iSPNs and dSPNs. In Shank3B-/- iSPNs, there is an almost complete loss of Shank3 proteins. Whereas in Shank3B-/- dSPNs, several Shank3 protein isoforms, such as Shank3E, are still expressed (Figure 3).
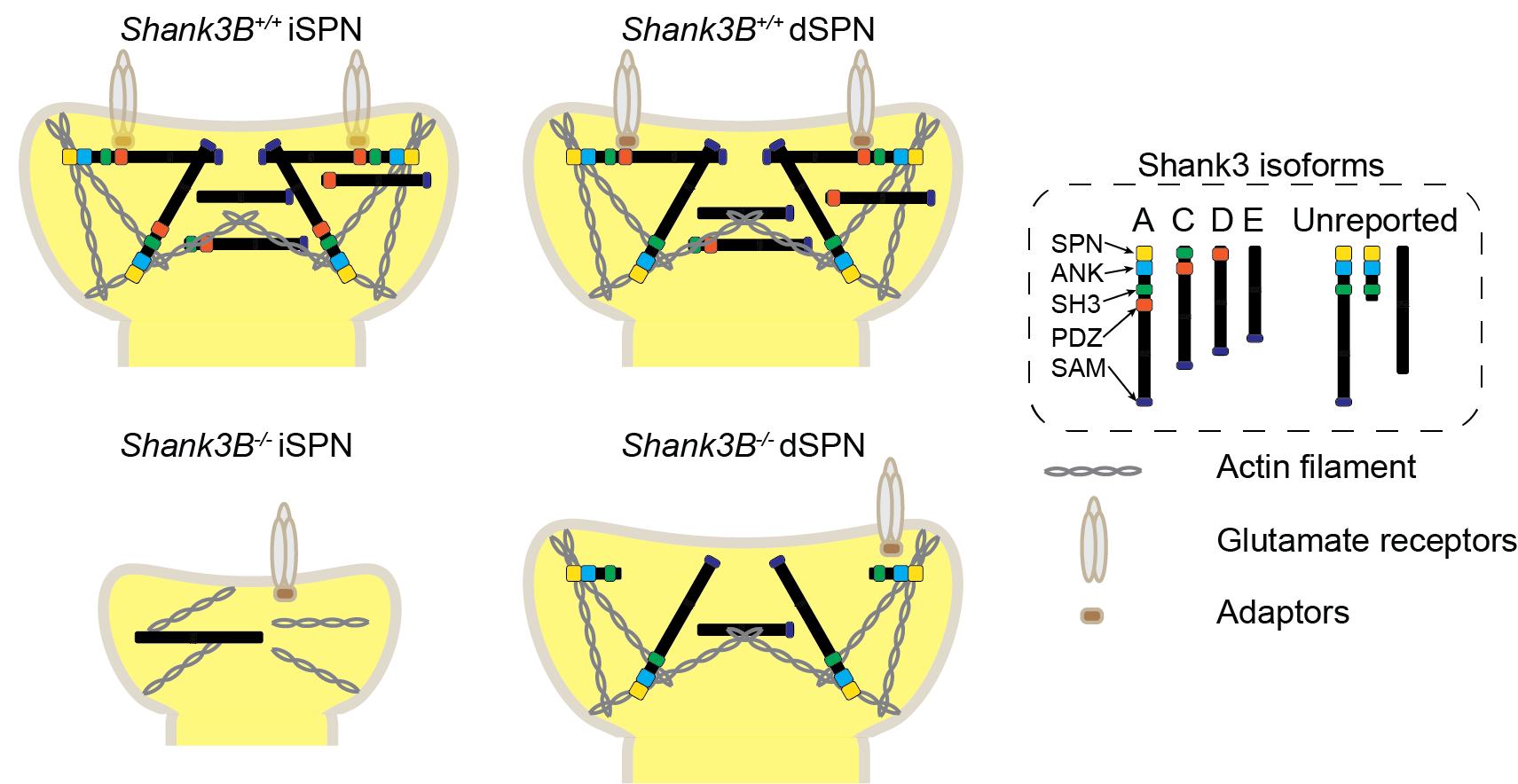
Figure 3. Working model of the molecular mechanism underlying SPN-type-specific postsynaptic compartment impairments in Shank3B-/- striatum.
(II) The postsynaptic proteome of dSPNs undergoes fewer changes than that of iSPNs in Shank3B-/- mice. Additionally, the dendritic spine morphology and synaptic function in dSPNs are less impaired compared to iSPNs in the Shank3B-/- model.
(III) Selective overexpression of Shank3E1 in Shank3B-/- iSPNs effectively rescued the deficits in dendritic spine morphology and corticostriatal synaptic transmission in iSPNs.
Insights and limitations
Our research highlights the significant impact that disrupting neuron-type-specific protein isoform expression can have on synaptopathy. Contrasting Shank3 protein isoforms across different neuron types may explain the dissimilar symptoms observed in patients with diverse Shank3 mutations. However, this study focused on two striatal neuron types and did not study human patients. Expanding our knowledge of Shank3 protein isoform expression in additional neuron types, particularly those in the human brain, will accelerate the development of pharmacological and genetic treatments for neuronal disorders like ASD in the future.
Follow the Topic
-
Molecular Psychiatry
This journal publishes work aimed at elucidating biological mechanisms underlying psychiatric disorders and their treatment, with emphasis on studies at the interface of pre-clinical and clinical research.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in