(NHC)Pd(II) hydride-catalyzed dehydroaromatization by olefin chain-walking isomerization and transfer-dehydrogenation
Published in Chemistry
Palladium-catalyzed dehydrogenation serves as one of the vital methods for aromatic compound synthesis. During the past decades, significant progress has been made by using endo-cyclic olefins as starting materials; inexpensive oxidant or O2 could be used to remove Pd-H generated in the catalytic cycle (Fig. 1a)1-5. However, exo-cyclic olefins are rarely employed as substrates.
In principle, a Pd-H species can simultaneously catalyze both olefin isomerization and allyl CH dehydrogenation under suitable conditions. This work found N-heterocyclic carbene (NHC) as a suitable ligand to regulate those competing reactivity. With the help of olefin additives, a modular dehydroaromatization system was established for a wide range of exo- and endo-cyclic substrates without strong oxidants and under mild physical conditions.
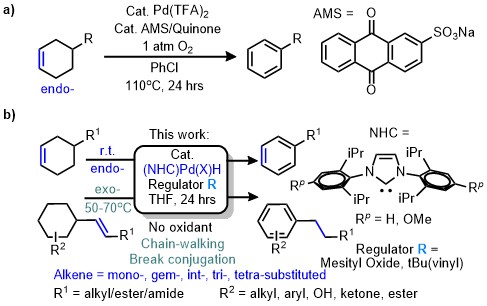
Fig. 1 Strategies in Pd-catalyzed dehydroaromatization (DHA) of cyclic alkenes. a AMS/quinone-assisted Pd(II) regeneration. b NHC-Pd(II)-H directed transfer-dehydroaromatization.
That change in active dehydrogenation species from PdX2 to (NHC)PdRX also came with good functional group tolerance. Ester, amide, pinacol ester, phenol, epoxide, etc., were tolerated under the standard neutral condition (Fig. 2a). Diverse exo-cyclic structures, like methylene, vinyl, and allyl cyclohexanes with different degrees of substitution, including tri-/tetra-substituted olefins generated by chain-walking, were all compatible (Fig. 2b).
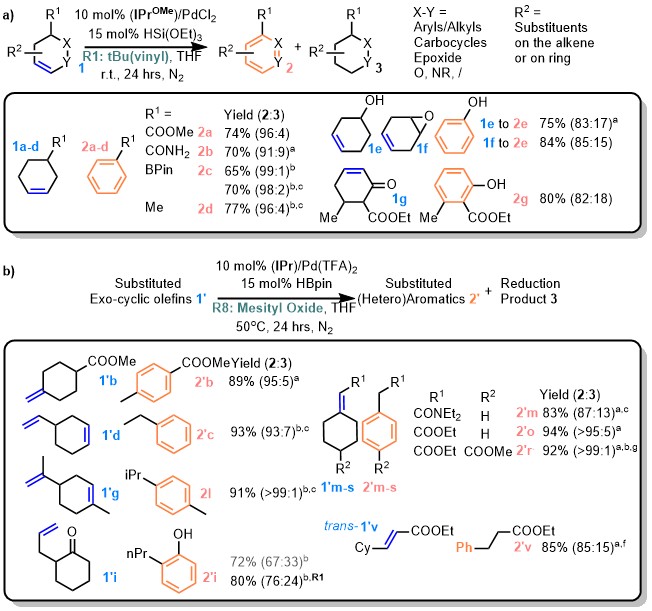
Fig. 2 Selected examples of TDHA reaction. a endocyclic olefin as substrates. b exocyclic olefins as substrates.
A suitable choice of regulator could manipulate the catalytic cycle, and the intermediates could be used for other valuable transformations. For instance, by using 1-morpholine cyclohexene as substrate, the corresponding substituted aniline or the Diels-Alder product were obtained optionally by simply changing the regulator under the standard condition (Fig. 3).
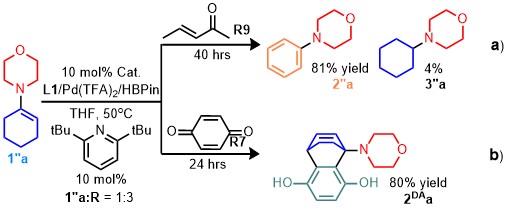
Fig. 3 Catalytic synthesis of a substituted aniline and dehydrogenative Diels-Alder product. a Synthesis of a substituted aniline. b Trapping the diene for the Diels-Alder reaction.
A highly chemoselective dehydrogenation of cyclic alkene over cyclic ketone was also achieved. If you want to know more about our work, please visit the following website: https://doi.org/10.1038/s41467-022-33163-6 .
Reference
- Wolfe, S. & Campbell, P.G.C. Allylic and homoallylic oxidation of cyclohexene by palladium(II) salts - Solvolytic behavior of possible organopalladium intermediates. J. Am. Chem. Soc. 93, 1499-1501 (1971).
- Trost, B.M. & Metzner, P.J. Reaction of olefins with palladium trifluoroacetate. J. Am. Chem. Soc. 102, 3572-3577 (1980).
- Burk, M.J. & Crabtree, R.H. Selective catalytic dehydrogenation of alkanes to alkenes. J. Am. Chem. Soc. 109, 8025-8032 (1987).
- Sheldon, R.A. & Sobczak, J.M. Catalytic oxidative dehydrogenation of cyclohexene. J. Mol. Catal. 68, 1-6 (1991).
- Iosub, A.V. & Stahl, S.S. Palladium-Catalyzed Aerobic Oxidative Dehydrogenation of Cyclohexenes to Substituted Arene Derivatives. J. Am. Chem. Soc. 137, 3454-3457 (2015).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in